Tutorial on Work Function by Dr. Rudy Schlaf Work Function in Metals: Figure 1 Shows a Schematic Energy Diagram of a Metal
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Study of Velocity Distribution of Thermionic Electrons with Reference to Triode Valve
Bulletin of Physics Projects, 1, 2016, 33-36 Study of Velocity Distribution of Thermionic electrons with reference to Triode Valve Anupam Bharadwaj, Arunabh Bhattacharyya and Akhil Ch. Das # Department of Physics, B. Borooah College, Ulubari, Guwahati-7, Assam, India # E-mail address: [email protected] Abstract Velocity distribution of the elements of a thermodynamic system at a given temperature is an important phenomenon. This phenomenon is studied critically by several physicists with reference to different physical systems. Most of these studies are carried on with reference to gaseous thermodynamic systems. Basically velocity distribution of gas molecules follows either Maxwell-Boltzmann or Fermi-Dirac or Bose-Einstein Statistics. At the same time thermionic emission is an important phenomenon especially in electronics. Vacuum devices in electronics are based on this phenomenon. Different devices with different techniques use this phenomenon for their working. Keeping all these in mind we decided to study the velocity distribution of the thermionic electrons emitted by the cathode of a triode valve. From our work we have found the velocity distribution of the thermionic electrons to be Maxwellian. 1. Introduction process of electron emission by the process of increase of Metals especially conductors have large number of temperature of a metal is called thermionic emission. The free electrons which are basically valence electrons. amount of thermal energy given to the free electrons is Though they are called free electrons, at room temperature used up in two ways- (i) to overcome the surface barrier (around 300K) they are free only to move inside the metal and (ii) to give a velocity (kinetic energy) to the emitted with random velocities. -

Quantum Mechanics Electromotive Force
Quantum Mechanics_Electromotive force . Electromotive force, also called emf[1] (denoted and measured in volts), is the voltage developed by any source of electrical energy such as a batteryor dynamo.[2] The word "force" in this case is not used to mean mechanical force, measured in newtons, but a potential, or energy per unit of charge, measured involts. In electromagnetic induction, emf can be defined around a closed loop as the electromagnetic workthat would be transferred to a unit of charge if it travels once around that loop.[3] (While the charge travels around the loop, it can simultaneously lose the energy via resistance into thermal energy.) For a time-varying magnetic flux impinging a loop, theElectric potential scalar field is not defined due to circulating electric vector field, but nevertheless an emf does work that can be measured as a virtual electric potential around that loop.[4] In a two-terminal device (such as an electrochemical cell or electromagnetic generator), the emf can be measured as the open-circuit potential difference across the two terminals. The potential difference thus created drives current flow if an external circuit is attached to the source of emf. When current flows, however, the potential difference across the terminals is no longer equal to the emf, but will be smaller because of the voltage drop within the device due to its internal resistance. Devices that can provide emf includeelectrochemical cells, thermoelectric devices, solar cells and photodiodes, electrical generators,transformers, and even Van de Graaff generators.[4][5] In nature, emf is generated whenever magnetic field fluctuations occur through a surface. -

Alkamax Application Note.Pdf
OLEDs are an attractive and promising candidate for the next generation of displays and light sources (Tang, 2002). OLEDs have the potential to achieve high performance, as well as being ecologically clean. However, there are still some development issues, and the following targets need to be achieved (Bardsley, 2001). Z Lower the operating voltage—to reduce power consumption, allowing cheaper driving circuitry Z Increase luminosity—especially important for light source applications Z Facilitate a top-emission structure—a solution to small aperture of back-emission AMOLEDs Z Improve production yield These targets can be achieved (Scott, 2003) through improvements in the metal-organic interface and charge injection. SAES Getters’ Alkali Metal Dispensers (AMDs) are widely used to dope photonic surfaces and are applicable for use in OLED applications. This application note discusses use of AMDs in fabrication of alkali metal cathode layers and doped organic electron transport layers. OLEDs OLEDs are a type of LED with organic layers sandwiched between the anode and cathode (Figure 1). Holes are injected from the anode and electrons are injected from the cathode. They are then recombined in an organic layer where light emission takes place. Cathode Unfortunately, fabrication processes Electron transport layer are far from ideal. Defects at the Emission layer cathode-emission interface and at the Hole transport layer anode-emission interface inhibit Anode charge transfer. Researchers have added semiconducting organic transport layers to improve electron Figure 1 transfer and mobility from the cathode to the emission layer. However, device current levels are still too high for large-size OLED applications. SAES Getters S.p.A. -

11.5 FD Richardson Emission
Thermoionic Emission of Electrons: Richardson effect Masatsugu Sei Suzuki Department of Physics, SUNY at Binghamton (Date: October 05, 2016) 1. Introduction After the discovery of electron in 1897, the British physicist Owen Willans Richardson began work on the topic that he later called "thermionic emission". He received a Nobel Prize in Physics in 1928 "for his work on the thermionic phenomenon and especially for the discovery of the law named after him". The minimum amount of energy needed for an electron to leave a surface is called the work function. The work function is characteristic of the material and for most metals is on the order of several eV. Thermionic currents can be increased by decreasing the work function. This often- desired goal can be achieved by applying various oxide coatings to the wire. In 1901 Richardson published the results of his experiments: the current from a heated wire seemed to depend exponentially on the temperature of the wire with a mathematical form similar to the Arrhenius equation. J AT 2 exp( ) kBT where J is the emission current density, T is the temperature of the metal, W is the work function of the metal, kB is the Boltzmann constant, and AG is constant. 2. Richardson’s law We consider free electrons inside a metal. The kinetic energy of electron is given by 1 ( p 2 p 2 p 2 ) p 2m x y z Fig. The work function of a metal represents the height of the surface barrier over and above the Fermi level. Suppose that these electrons escape from the metal along the positive x direction. -

Determination of Filament Work Function in Vacuum by Paul Lulai
Determination of Filament Work Function in Vacuum by Paul Lulai John L Vossen Memorial Award Recipient October 2001 Objective: The objective of this experiment is to experimentally determine the work function (f) of a filament. This experiment will also demonstrate that work function is a property of the surface of the sample and has very little to do with the bulk of the sample. In this process, students will use data to determine an equation that models the relationship between temperature and resistance of a filament. Students will also construct a system for conducting research in vacuum. A Review of Work Function and Thermionic Emission: The energy required to remove an electron from the Fermi-level of a metal and free it from the influence of that metal is a property of the metal itself. This property is known as the metal’s work function (f) (Oxford Paperback Reference, 1990). Before the advent of transistors, vacuum tubes were used to amplify signals. It requires less energy to remove an electron from a vacuum tube’s filament if the work function of the filament is low. As current flows through a circuit of two dissimilar metals heat is absorbed at one junction and given up at the other. It has recently been discovered that if the metals used have low work functions this process (known as the Peltier Effect) can occur at an efficiency of 70-80%. This process shows promise for uses ranging from cooling electronics to more industrial cooling processes. How well electrons can be removed from a heated filament depends on that filaments work function. -

Chapter 4: Bonding in Solids and Electronic Properties
Chapter 4: Bonding in Solids and Electronic Properties Free electron theory Consider free electrons in a metal – an electron gas. •regards a metal as a box in which electrons are free to move. •assumes nuclei stay fixed on their lattice sites surrounded by core electrons, while the valence electrons move freely through the solid. •Ignoring the core electrons, one can treat the outer electrons with a quantum mechanical description. •Taking just one electron, the problem is reduced to a particle in a box. Electron is confined to a line of length a. Schrodinger equation 2 2 2 d d 2me E 2 (E V ) 2 2 2me dx dx Electron is not allowed outside the box, so the potential is ∞ outside the box. Energy is quantized, with quantum numbers n. 2 2 2 n2h2 h2 n n n In 1d: E In 3d: E a b c 2 8m a2 b2 c2 8mea e 1 2 2 2 2 Each set of quantum numbers n , n , h n n n a b a b c E 2 2 2 and nc will give rise to an energy level. 8me a b c •In three dimensions, there are multiple combination of energy levels that will give the same energy, whereas in one-dimension n and –n are equal in energy. 2 2 2 2 2 2 na/a nb/b nc/c na /a + nb /b + nc /c 6 6 6 108 The number of states with the 2 2 10 108 same energy is known as the degeneracy. 2 10 2 108 10 2 2 108 When dealing with a crystal with ~1020 atoms, it becomes difficult to work out all the possible combinations. -
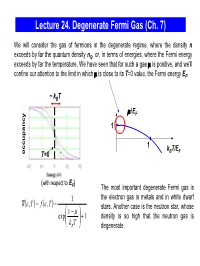
Lecture 24. Degenerate Fermi Gas (Ch
Lecture 24. Degenerate Fermi Gas (Ch. 7) We will consider the gas of fermions in the degenerate regime, where the density n exceeds by far the quantum density nQ, or, in terms of energies, where the Fermi energy exceeds by far the temperature. We have seen that for such a gas μ is positive, and we’ll confine our attention to the limit in which μ is close to its T=0 value, the Fermi energy EF. ~ kBT μ/EF 1 1 kBT/EF occupancy T=0 (with respect to E ) F The most important degenerate Fermi gas is 1 the electron gas in metals and in white dwarf nε()(),, T= f ε T = stars. Another case is the neutron star, whose ε⎛ − μ⎞ exp⎜ ⎟ +1 density is so high that the neutron gas is ⎝kB T⎠ degenerate. Degenerate Fermi Gas in Metals empty states ε We consider the mobile electrons in the conduction EF conduction band which can participate in the charge transport. The band energy is measured from the bottom of the conduction 0 band. When the metal atoms are brought together, valence their outer electrons break away and can move freely band through the solid. In good metals with the concentration ~ 1 electron/ion, the density of electrons in the electron states electron states conduction band n ~ 1 electron per (0.2 nm)3 ~ 1029 in an isolated in metal electrons/m3 . atom The electrons are prevented from escaping from the metal by the net Coulomb attraction to the positive ions; the energy required for an electron to escape (the work function) is typically a few eV. -

Chapter 13 Ideal Fermi
Chapter 13 Ideal Fermi gas The properties of an ideal Fermi gas are strongly determined by the Pauli principle. We shall consider the limit: k T µ,βµ 1, B � � which defines the degenerate Fermi gas. In this limit, the quantum mechanical nature of the system becomes especially important, and the system has little to do with the classical ideal gas. Since this chapter is devoted to fermions, we shall omit in the following the subscript ( ) that we used for the fermionic statistical quantities in the previous chapter. − 13.1 Equation of state Consider a gas ofN non-interacting fermions, e.g., electrons, whose one-particle wave- functionsϕ r(�r) are plane-waves. In this case, a complete set of quantum numbersr is given, for instance, by the three cartesian components of the wave vector �k and thez spin projectionm s of an electron: r (k , k , k , m ). ≡ x y z s Spin-independent Hamiltonians. We will consider only spin independent Hamiltonian operator of the type ˆ 3 H= �k ck† ck + d r V(r)c r†cr , �k � where thefirst and the second terms are respectively the kinetic and th potential energy. The summation over the statesr (whenever it has to be performed) can then be reduced to the summation over states with different wavevectork(p=¯hk): ... (2s + 1) ..., ⇒ r � �k where the summation over the spin quantum numberm s = s, s+1, . , s has been taken into account by the prefactor (2s + 1). − − 159 160 CHAPTER 13. IDEAL FERMI GAS Wavefunctions in a box. We as- sume that the electrons are in a vol- ume defined by a cube with sidesL x, Ly,L z and volumeV=L xLyLz. -

Core-Level Photoemission and Work-Function Investigation of Na on Cu(110) C
University of Rhode Island DigitalCommons@URI Physics Faculty Publications Physics 1993 Core-Level Photoemission and Work-Function Investigation of Na on Cu(110) C. Su X. Shi See next page for additional authors Follow this and additional works at: https://digitalcommons.uri.edu/phys_facpubs Terms of Use All rights reserved under copyright. Citation/Publisher Attribution Su, C., Shi, X., Tang, D., Heskett, D., & Tsuei, K.-D. (1993). Core-level photoemission and work-function investigation of Na on Cu(110). Physical Review B, 48(16), 12146-12150. doi: 10.1103/PhysRevB.48.12146 Available at: http://dx.doi.org/10.1103/PhysRevB.48.12146 This Article is brought to you for free and open access by the Physics at DigitalCommons@URI. It has been accepted for inclusion in Physics Faculty Publications by an authorized administrator of DigitalCommons@URI. For more information, please contact [email protected]. Authors C. Su, X. Shi, D. Tang, David R. Heskett, and K. -D. Tsuei This article is available at DigitalCommons@URI: https://digitalcommons.uri.edu/phys_facpubs/229 PHYSICAL REVIEW B VOLUME 48, NUMBER 16 15 OCTOBER 1993-II Core-level photoemission and work-function investigation of Na on Cu(110) C. Su, X. Shi, D. Tang, and D. Heskett Department ofPhysics, University ofRhode Island, Kingston, Rhode Island 02881 K.-D. Tsuei Department ofPhysics, Brookhaven National Laboratory, Upton, New York 11973 (Received 19 November 1992; revised manuscript received 12 April 1993) Core-level photoemission, low-energy electron diffraction (LEED), and work-function change mea- surements have been carried out to study the coverage dependence of Na/Cu(110) at room temperature. -

Work Function and Process Integration Issues of Metal
WORK FUNCTION AND PROCESS INTEGRATION ISSUES OF METAL GATE MATERIALS IN CMOS TECHNOLOGY REN CHI NATIONAL UNIVERSITY OF SINGAPORE 2006 WORK FUNCTION AND PROCESS INTEGRATION ISSUES OF METAL GATE MATERIALS IN CMOS TECHNOLOGY REN CHI B. Sci. (Peking University, P. R. China) 2002 A THESIS SUBMITTED FOR THE DEGREE OF DOCTOR OF PHILOSOPHY DEPARTMENT OF ELECTRICAL AND COMPUTER ENGINEERING NATIONAL UNIVERSITY OF SINGAPORE OCTOBER 2006 _____________________________________________________________________ ACKNOWLEGEMENTS First of all, I would like to express my sincere thanks to my advisors, Prof. Chan Siu Hung and Prof. Kwong Dim-Lee, who provided me with invaluable guidance, encouragement, knowledge, freedom and all kinds of support during my graduate study at NUS. I am extremely grateful to Prof. Chan not only for his patience and painstaking efforts in helping me in my research but also for his kindness and understanding personally, which has accompanied me over the past four years. He is not only an experienced advisor for me but also an elder who makes me feel peaceful and blessed. I also greatly appreciate Prof. Kwong from the bottom of my heart for his knowledge, expertise and foresight in the field of semiconductor technology, which has helped me to avoid many detours in my research work. I do believe that I will be immeasurably benefited from his wisdom and professional advice throughout my career and my life. I would also like to thank Prof. Kwong for all the opportunities provided in developing my potential and personality, especially the opportunity to join the Institute of Microelectronics, Singapore to work with and learn from so many experts in a much wider stage. -

Thermoelectric Phenomena
Journal of Materials Education Vol. 36 (5-6): 175 - 186 (2014) THERMOELECTRIC PHENOMENA Witold Brostow a, Gregory Granowski a, Nathalie Hnatchuk a, Jeff Sharp b and John B. White a, b a Laboratory of Advanced Polymers & Optimized Materials (LAPOM), Department of Materials Science and Engineering and Department of Physics, University of North Texas, 3940 North Elm Street, Denton, TX 76207, USA; http://www.unt.edu/LAPOM/; [email protected], [email protected], [email protected]; [email protected] b Marlow Industries, Inc., 10451 Vista Park Road, Dallas, TX 75238-1645, USA; Jeffrey.Sharp@ii- vi.com ABSTRACT Potential usefulness of thermoelectric (TE) effects is hard to overestimate—while at the present time the uses of those effects are limited to niche applications. Possibly because of this situation, coverage of TE effects, TE materials and TE devices in instruction in Materials Science and Engineering is largely perfunctory and limited to discussions of thermocouples. We begin a series of review articles to remedy this situation. In the present article we discuss the Seebeck effect, the Peltier effect, and the role of these effects in semiconductor materials and in the electronics industry. The Seebeck effect allows creation of a voltage on the basis of the temperature difference. The twin Peltier effect allows cooling or heating when two materials are connected to an electric current. Potential consequences of these phenomena are staggering. If a dramatic improvement in thermoelectric cooling device efficiency were achieved, the resulting elimination of liquid coolants in refrigerators would stop one contribution to both global warming and destruction of the Earth's ozone layer. -

Schottky Barrier Height Dependence on the Metal Work Function for P-Type Si Schottky Diodes
Schottky Barrier Height Dependence on the Metal Work Function for p-type Si Schottky Diodes G¨uven C¸ ankayaa and Nazım Uc¸arb a Gaziosmanpas¸a University, Faculty of Sciences and Arts, Physics Department, Tokat, Turkey b S¨uleyman Demirel University, Faculty of Sciences and Arts, Physics Department, Isparta, Turkey Reprint requests to Prof. N. U.; E-mail: [email protected] Z. Naturforsch. 59a, 795 – 798 (2004); received July 5, 2004 We investigated Schottky barrier diodes of 9 metals (Mn, Cd, Al, Bi, Pb, Sn, Sb, Fe, and Ni) hav- ing different metal work functions to p-type Si using current-voltage characteristics. Most Schottky contacts show good characteristics with an ideality factor range from 1.057 to 1.831. Based on our measurements for p-type Si, the barrier heights and metal work functions show a linear relationship of current-voltage characteristics at room temperature with a slope (S = φb/φm) of 0.162, even though the Fermi level is partially pinned. From this linear dependency, the density of interface states was determined to be about 4.5 · 1013 1/eV per cm2, and the average pinning position of the Fermi level as 0.661 eV below the conduction band. Key words: Schottky Diodes; Barrier Height; Series Resistance; Work Function; Miedema Electronegativity. 1. Introduction ear dependence should exist between the φb and the φm. Corresponding to this, SB heights of various el- The investigation of rectifying metal-semiconductor emental metals, including Au, Ti, Pt, Ni, Cr, have contacts (Schottky diodes) is of current interest for been reported [14 – 16].