IJMM July 07.Indd
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

“Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics Against Gram
ORIGINAL ARTICLE A Comparative In Vitro Sensitivity Study of “Ceftriaxone– Sulbactam–EDTA” and Various Antibiotics against Gram- negative Bacterial Isolates from Intensive Care Unit Sweta Singh1, Chinmoy Sahu2, Sangram Singh Patel3, Abhay Singh4, Nidhi Yaduvanshi5 ABSTRACT Introduction: A rapid increase in multidrug-resistant (MDR) strains is being seen across the globe especially in the Southeast Asian region, including India. Carbapenems and colistin form the mainstay of treatment against gram-negative pathogens, especially extended-spectrum beta-lactamase (ESBL)- and metallo-beta-lactamse (MBL)-producing isolates. However, due to increased resistance to carbapenems and toxicity of colistin, especially in intensive care units (ICUs), carbapenem-sparing antibiotics like ceftriaxone–sulbactam–EDTA (CSE) combination needs to be evaluated. Materials and methods: Bacterial isolates cultured from various clinical samples from all ICUs for a period of 9 months were evaluated. Bacterial identification was performed by matrix assisted laser desorption ionization time of flight mass spectrometry (MALDI-TOF MS) and antibiotic susceptibility testing were performed by disk diffusion and E test method. Antibiogram of various antibiotics was noted. Extended-spectrum beta-lactamase- and MBL-producing bacteria were identified by phenotypic methods. Antibiotic sensitivity results of CSE were compared with the comparator drugs like colistin, carbapenems, and tigecycline in Enterobacteriaceae, Acinetobacter spp., and Pseudomonas spp. along with ESBL and MBL producers. Results: A total of 2,760 samples of blood, cerebrospinal fluid (CSF), respiratory samples, tissue, and pus were collected from ICUs with maximum isolates from pus (37%) followed by respiratory samples (31%) and blood (27%). Escherichia coli and Klebsiella pneumoniae were the predominant gram-negative pathogens accounting for 56% of the isolates followed by Acinetobacter spp. -

Cefoxitin Versus Piperacillin– Tazobactam As Surgical Antibiotic Prophylaxis in Patients Undergoing Pancreatoduodenectomy: Protocol for a Randomised Controlled Trial
Open access Protocol BMJ Open: first published as 10.1136/bmjopen-2020-048398 on 4 March 2021. Downloaded from Cefoxitin versus piperacillin– tazobactam as surgical antibiotic prophylaxis in patients undergoing pancreatoduodenectomy: protocol for a randomised controlled trial Nicole M Nevarez ,1 Brian C Brajcich,2 Jason Liu,2,3 Ryan Ellis,2 Clifford Y Ko,2 Henry A Pitt,4 Michael I D'Angelica,5 Adam C Yopp1 To cite: Nevarez NM, ABSTRACT Strengths and limitations of this study Brajcich BC, Liu J, et al. Introduction Although antibiotic prophylaxis is Cefoxitin versus piperacillin– established in reducing postoperative surgical site tazobactam as surgical ► A major strength of this study is the multi- infections (SSIs), the optimal antibiotic for prophylaxis in antibiotic prophylaxis institutional, double- arm, randomised controlled in patients undergoing pancreatoduodenectomy (PD) remains unclear. The study trial design. objective is to evaluate if administration of piperacillin– pancreatoduodenectomy: ► A limitation of this study is that all perioperative care protocol for a randomised tazobactam as antibiotic prophylaxis results in decreased is at the discretion of the operating surgeon and is controlled trial. BMJ Open 30- day SSI rate compared with cefoxitin in patients not standardised. 2021;11:e048398. doi:10.1136/ undergoing elective PD. ► All data will be collected through the American bmjopen-2020-048398 Methods and analysis This study will be a multi- College of Surgeons National Surgical Quality ► Prepublication history for institution, double- arm, non- blinded randomised controlled Improvement Program, which is a strength for its this paper is available online. superiority trial. Adults ≥18 years consented to undergo PD ease of use but a limitation due to the variety of data To view these files, please visit for all indications who present to institutions participating included. -

Cefoperazone Sodium
Disclaimer While these labels are the most current Drug Control Authority (DCA) approved versions of content, these are not necessarily reflective of final printed labeling currently in distribution LPD Title : Cefoperazone Sodium LPD Date : 07 February 2017 Country : Malaysia Reference : CDS version 5.0 dated 07 January 2017 Reason : Addition of adverse drug reactions (ADRs) Haemorrhage and Coagulopathy to Section 4.8 Undesirable effects, and also, addition of serious hemorrhage and coagulopathy to the warning about vitamin K deficiency in Section 4.4 Special warnings and precautions for use Addition of ADRs Anaphylactic reaction and Anaphylactic shock to Section 4.8 Undesirable effects Addition of ADR Dermatitis exfoliative to Section 4.8 Undesirable Effects and a warning statement on severe cutaneous adverse reactions (SCARs) to Section 4.4, Special warnings and precautions for use 1. NAME OF THE MEDICINAL PRODUCT CEFOBID 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Cefoperazone sodium is a semisynthetic broad-spectrum cephalosporin antibiotic for parenteral use only. Cefoperazone contains 34 mg sodium (1.5 mEq) per gram. Cefoperazone is a white crystalline powder which is freely soluble in water. The pH of a 25% aqueous solution is 5.0 to 6.5 and the solution is colorless to straw-yellow, depending on the concentration. The empirical formula is C25H26N9NaO8S2. 3. PHARMACEUTICAL FORM Cefoperazone sodium is available in vials containing 1 and 2 grams. Not For Distribution PFIZER CONFIDENTIAL Not all product strengths or pack sizes -

Lactamase-Producing Antibiotic-Resistant Escherichia Coli and Klebsiella Pneumoniae in Ready-To-Eat Street Foods
antibiotics Article Prevalence and Characterization of Extended-Spectrum β-Lactamase-Producing Antibiotic-Resistant Escherichia coli and Klebsiella pneumoniae in Ready-to-Eat Street Foods Shobha Giri 1 , Vaishnavi Kudva 1, Kalidas Shetty 2 and Veena Shetty 1,* 1 Department of Microbiology, KS Hegde Medical Academy, Nitte (Deemed to be University), Mangalore 575018, India; [email protected] (S.G.); [email protected] (V.K.) 2 Department of Plant Sciences, North Dakota State University, Fargo, ND 58102, USA; [email protected] * Correspondence: [email protected] Abstract: As the global urban populations increase with rapid migration from rural areas, ready-to- eat (RTE) street foods are posing food safety challenges where street foods are prepared with less structured food safety guidelines in small and roadside outlets. The increased presence of extended- spectrum-β-lactamase (ESBL) producing bacteria in street foods is a significant risk for human health because of its epidemiological significance. Escherichia coli and Klebsiella pneumoniae have become important and dangerous foodborne pathogens globally for their relevance to antibiotic resistance. The present study was undertaken to evaluate the potential burden of antibiotic-resistant E. coli and K. pneumoniae contaminating RTE street foods and to assess the microbiological quality of foods in a Citation: Giri, S.; Kudva, V.; Shetty, typical emerging and growing urban suburb of India where RTE street foods are rapidly establishing K.; Shetty, V. Prevalence and with public health implications. A total of 100 RTE food samples were collected of which, 22.88% Characterization of were E. coli and 27.12% K. pneumoniae. The prevalence of ESBL-producing E. -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -

Comparative Effects of Cefpirome (Hr 810) and Other Cephalosporins on Experimentally Induced Pneumonia in Mice
VOL. XXXIX NO. 7 THE JOURNAL OF ANTIBIOTICS 971 COMPARATIVE EFFECTS OF CEFPIROME (HR 810) AND OTHER CEPHALOSPORINS ON EXPERIMENTALLY INDUCED PNEUMONIA IN MICE N. KLESEL, D. ISERT, M. LIMBERT, G. SEIBERT, I. WINKLER and E. SCHRINNER Hoechst Aktiengesellschaft, Dept. of Chemotherapy, 6230 Frankfurt a. M. 80, FRG (Received for publication March 24, 1986) The chemotherapeutic efficacy of cefpirome (HR 810), a new polar aminothiazolyl- cephalosporin and that of ceftazidime, cefotaxime, cefoperazone, latamoxef and cefodizime were examined against experimental pneumonia caused by Klebsiella pneammlliae DT-S in mice. When compared in terms of MIC values against the infecting organism and the phar- macokinetic pattern, cefpirome showed equal activity and a similar pharmacokinetic behavior to ceftazidime and cefotaxime in mice. Trials to assess the bactericidial activity in vhro, however, showed that cefpirome displayed a more marked bactericidal effect in pneumonic mice than the other cephalosporins tested. Only cefodizime, a cephalosporin with extremely high and prolonged blood and tissue levels in experimental animals exerted chemotherapeutic effects similar to cefpirome. After cefpirome or cefodizime medication (50 mg/kg), the viable counts in the lungs of experimental animals fell steadily to 1/10,000 of the pretreatment level and, in contrast to the reference compounds, no regrowth of the challenge organisms could be observed with both drugs. Moreover, with ED.-,,,,sranging from 1.1 to 59.1 mg/kg in treatment studies, cefpirome as well as cefodizime were two to ten times more effective than ceftazidime and cefotaxime, whereas cefoperazone and latamoxef were considerably less effective. Cefpirome (3-((2,3-cyclopenteno-I-pyridium)methyl)-7-(2-syn-methoximino-2-(2-aminothiazol-4- yl)acetamido)ceph-3-em-4-carboxylate, HR 810) is a new semi-synthetic parenteral cephalosporin antibiotic with a broad spectrum of antibacterial activity in vitro and in rivo. -

Severe Sepsis Tipsheet
Severe Sepsis Tipsheet SEVERE SEPSIS A, B, and C of the following must be met within 6 hours of each other. A. Documentation of a suspected infection. There may be reference to “possible infection,” “suspect infection,” “rule out infection,” or similar documentation. B. TWO or more manifestations of systemic infection according to the Systemic Inflammatory Response Syndrome (SIRS) criteria, which are: • Temperature > 38.3 C or < 36.0 C (> 100.9 F or < 96.8 F) • Heart rate (pulse) > 90 • Respiration > 20 per minute • White blood cell count > 12,000 or < 4,000 or > 10% bands C. Organ dysfunction, evidenced by any one of the following: • Systolic blood pressure (SBP) < 90 mmHg, or mean arterial pressure < 65 mmHg, or a systolic blood pressure decrease of more than 40 mmHg. Physician/APN/PA documentation must be present in the medical record indicating a > 40 mmHg decrease in SBP has occurred and is related to infection, severe sepsis or septic shock and not other causes. • Acute respiratory failure as evidenced by a new need for invasive or non-invasive mechanical ventilation. To use acute respiratory failure as a sign of organ dysfunction there must be: • Documentation of acute respiratory failure AND • Documentation the patient is on mechanical ventilation • Invasive mechanical ventilation requires an endotracheal or tracheostomy tube. Non-invasive mechanical ventilation may be referred to as BiPAP or CPAP. • New need for mechanical ventilation indicates the patient was not using the same type of mechanical ventilation prior to the current acute respiratory failure. • Use the time at which there is documentation the patient has both acute respiratory failure and is on mechanical ventilation. -
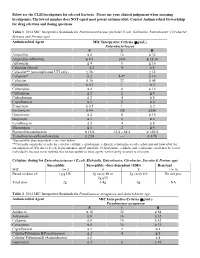
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

Fosfomycin Resistance in Escherichia Coli Isolates from South Korea and in Vitro Activity of Fosfomycin Alone and in Combination with Other Antibiotics
antibiotics Article Fosfomycin Resistance in Escherichia coli Isolates from South Korea and in vitro Activity of Fosfomycin Alone and in Combination with Other Antibiotics Hyeri Seok 1 , Ji Young Choi 2, Yu Mi Wi 3, Dae Won Park 1, Kyong Ran Peck 4 and Kwan Soo Ko 2,* 1 Division of Infectious Diseases, Department of Medicine, Korea University Ansan Hospital, Korea University College of Medicine, Ansan 15355, Korea; [email protected] (H.S.); [email protected] (D.W.P.) 2 Department of Microbiology, Sungkyunkwan University School of Medicine, Suwon 16419, Korea; [email protected] 3 Division of Infectious Diseases, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon 51353, Korea; [email protected] 4 Division of Infectious Diseases, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul 06351, Korea; [email protected] * Correspondence: [email protected]; Tel.: +82-31-299-6223 Received: 17 February 2020; Accepted: 3 March 2020; Published: 6 March 2020 Abstract: We investigated fosfomycin susceptibility in Escherichia coli clinical isolates from South Korea, including community-onset, hospital-onset, and long-term care facility (LTCF)-onset isolates. The resistance mechanisms and genotypes of fosfomycin-resistant isolates were also identified. Finally, the in vitro efficacy of combinations of fosfomycin with other antibiotics were examined in susceptible or extended spectrum β-lactamase (ESBL)-producing E. coli isolates. The fosfomycin resistance rate was 6.7% and was significantly higher in LTCF-onset isolates than community-onset and hospital-onset isolates. Twenty-one sequence types (STs) were identified among 19 fosfomycin-resistant E. coli isolates, showing diverse genotypes. fosA3 was found in only two isolates, and diverse genetic variations were identified in three genes associated with fosfomycin resistance, namely, GlpT, UhpT, and MurA. -

Piperacillin/Tazobactam Drug Class1 Antibiotic – Penicillin with Β-Lactamase Inhibitor
Monographs for Commonly Administered Intravenous Medications in Home and Community Care Piperacillin/Tazobactam Drug Class1 Antibiotic – penicillin with β-lactamase inhibitor Spectrum1 Refer to product monograph for complete spectrum For β-lactamase producing bacteria strains (e.g., Haemophilus influenza, Escherichia coli, and Staphylococcus aureus). Also to susceptible Acinetobacter species, Klebsiella pneumonia, Pseudomonas aeruginosa often combined with aminoglycoside). Cross Sensitivities / Allergies1 Cross sensitivities with penicillin and possibly cephalosporin and/or beta-lactam inhibitors Indications1,2 Intra-abdominal Respiratory tract Skin and skin structure Septicemia Gynecological Urinary tract Other conditions based on culture and sensitivity results Outpatient Considerations1 For patients with a documented allergy to penicillin, cephalosporin or beta-lactam inhibitor, the first dose should be administered in a hospital or clinic setting. Must be able to access laboratory monitoring (either at outpatient laboratory or by arranging in-home lab) if using an interacting oral medication (see Potential Drug Interactions section) Prescribing Considerations At time of ordering please provide the following to the pharmacist: and Dosage in Adults1,2 Height, weight Most recent serum creatinine with date obtained Indication (infection being treated) Usually dosed every 6 to 8 hours. Available as 2 g/0.25 g, 3 g/0.375 g, 4 g/0.5 g piperacillin/tazobactam (dose selected based on indication) Maximum 18 g/ 2.25 g piperacillin/tazobactam daily Dose and administration interval require adjustment for renal impairment Errors have occurred due to unusual ordering as combined dose of piperacillin and tazobactam. Order may be expressed only as piperacillin component or as a total of piperacillin + tazobactam (8:1 ratio). -

Time-Series Analysis of the Relationship of Antimicrobial Use and Hand Hygiene Promotion with the Incidence of Healthcare-Associated Infections
The Journal of Antibiotics (2012) 65, 311–316 & 2012 Japan Antibiotics Research Association All rights reserved 0021-8820/12 www.nature.com/ja ORIGINAL ARTICLE Time-series analysis of the relationship of antimicrobial use and hand hygiene promotion with the incidence of healthcare-associated infections Yuan-Ti Lee1,2,3, Shiuan-Chih Chen1,2,4, Meng-Chih Lee1,2,4, Hung-Chang Hung1,2,5, Huey-Jen Huang3,6, Hui-Chih Lin1,7, Der-Jinn Wu1,2,3 and Shih-Ming Tsao1,2,3 We analyzed the effect of antimicrobial use and implementation of a hand hygiene program on the incidence of healthcare- associated infections (HAIs) and healthcare-associated methicillin-resistant Staphylococcus aureus (HA-MRSA) infections at the Chung Shan Medical University Hospital (Taichung, Taiwan). Monthly data were retrospectively reviewed from January 2004 to December 2010. Use of antimicrobials and alcohol-based hand cleaner were separately regressed against the incidences of HAIs and HA-MRSA infections. Infection incidence was expressed as persons per 1000 patient days (PDs), monthly use of i.v. antibiotics was expressed as defined daily doses per 1000 PDs and monthly alcohol-based hand cleaner use was expressed as bottle per 1000 PDs. Multivariate analysis indicated that use of hand cleaner was associated with reduced incidence of HAIs (P ¼ 0.0001) and HA-MRSA infections (Po0.0001). Time-series analysis indicated that increased use of hand cleaner was significantly associated with significant decreases in the incidences of HAIs and HA-MRSA infections. Total antibiotic use had no significant effect on HAIs, but was associated with more HA-MRSA infections.