Uranyl Acetate Safety
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Extraction Processing of Concentrated Solutions of Uranyl Nitrate with High Impurities Content V.M
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Electronic archive of Tomsk Polytechnic University Сhemistry UDC 546.791.02.238:66.061.51 EXTRACTION PROCESSING OF CONCENTRATED SOLUTIONS OF URANYL NITRATE WITH HIGH IMPURITIES CONTENT V.M. Korotkevich, V.V.Lazarchuck, T.G. Shikerun, V.I. Shamin, N.A. Mikhailova, F.A. Dorda Federal Unitary Enterprise Siberian Group of Chemical Enterprises, Seversk Еmail: [email protected] Process flowsheet of recycling uranium concentrated solutions with its purification from insoluble impurities of iron, silicon, molybde num, calcium oxides and hydroxides and soluble impurities with application of centrifugal extractors cascade has been developed and suggested for commercial introduction. The process was carried out at extractant saturation (30 % tributyl phosphate in hydrocarbon diluent) in extraction assembly lower than a limiting level (85...95 g/l) and in wash assembly – at limiting saturation (up to 120 g/l). As a result the waste uranium content in watertail solutions 0,01...0,04 g/l and minimal content of impurities in reextractors is provided. Perspective program on development of nuclear technology are also: reliability of hermetization, possi power engineering proposes to involve wide range of raw bility of remote service, resistance to corrosive attack of materials from uranium ores to secondary uranium ma structural materials, duration of overhaul period etc. terials into processing at atomic enterprises. In this case In Russia the centrifugal extractor of ECТseries the problem of reprocessing concentrated uranium so with continuous extraction of solid phase [3], which may lutions with high impurity content occurs. -
Nitrate (UO2 (NO)) 4
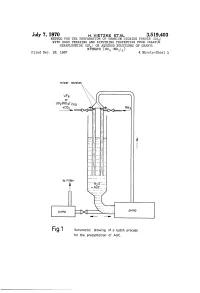
July 7, 1970 H. VETZKE ETA 3,519,403 METHOD FOR THE PREPARATION OF URANIUM DIOXIDE POWDER (UO) WITH GOOD PRESSING AND SINTERING PROPERTIES FROM URANIUM HEXAFLUORIDE (UF) OR AQUEOUS SOLUTIONS OF URANYL Fied Dec. 18, 1967 NiTRATE (UO2 (NO)) 4. Sheets-Sheet l mixer nozzles --race to fitter Fig.1 Schematic drawing of a batch process for the precipitation of AUC. July 7, 1970 H. VETZKE EA 3,519,403 METHOD FOR THE PREPARATION OF URANIUM DIOXIDE POWDER (UO) WITH GOOD PRESSING AND SINTERING PROPERTIES FROM RANIUM HEXAFLUORIDE (UF) OR AQUEOUS SOLUTIONS OF URANYL Filed Dec. 8, 1967 NiTRATE (UO2 (NO)) 4. Sheets-Sheet 3. mixer nozzles UF6 Of U02 (NO3)2ad tC92--X Ex-Ns / to filter cooling precipitation Vessel vessel Fig. 2 Schematic drawing of a continuous process for the precipitation of AUC July 7, 1970 H, VETZKE ETAL 3,519,403 METHOD FOR THE PREPARATION OF URANIUM DIOXIDE POWDER (UO) WITH GOOD PRESSING AND SINTERING PROPERTIES FROM URANIUM HEXAFLUORIDE (UF) OR AQUEOUS SOLUTIONS OF URANYL Filed Dec. 18, 1967 NiTRATE (UO (NO)2) 4. Sheets-Sheet 3 Off gas Off gas 487\m Nx UO2 powder XXX.& X.X S& steam / H2 Fig. 3 Ot Schematic drawing of a fluidized bed furnace for the reduction of AUC to U02, July 7, 1970 H. VEZKE ETAL 3,519,403 METHOD FOR THE PREPARATION OF URANIUM DIOXIDE POWDER (UO) WITH GOOD PRESSING AND SINTERING PROPERTIES FROM URANIUM HEXAFLUORIDE (UF) OR AQUEOUS SOLUTIONS OF URANYL NITRATE (UO2 (NO)a) Filed Dec. 18, 1967 4. Sheets-Sheet 1 -- Offgas Step 1: Decomposition, reduction and pyrohydrolysis i Step 2: Pyrohydrolysis Step 3: Controlled oxidation an o- are as an un- Fig. -

Sources, Effects and Risks of Ionizing Radiation
SOURCES, EFFECTS AND RISKS OF IONIZING RADIATION United Nations Scientific Committee on the Effects of Atomic Radiation UNSCEAR 2016 Report to the General Assembly, with Scientific Annexes UNITED NATIONS New York, 2017 NOTE The report of the Committee without its annexes appears as Official Records of the General Assembly, Seventy-first Session, Supplement No. 46 and corrigendum (A/71/46 and Corr.1). The report reproduced here includes the corrections of the corrigendum. The designations employed and the presentation of material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. The country names used in this document are, in most cases, those that were in use at the time the data were collected or the text prepared. In other cases, however, the names have been updated, where this was possible and appropriate, to reflect political changes. UNITED NATIONS PUBLICATION Sales No. E.17.IX.1 ISBN: 978-92-1-142316-7 eISBN: 978-92-1-060002-6 © United Nations, January 2017. All rights reserved, worldwide. This publication has not been formally edited. Information on uniform resource locators and links to Internet sites contained in the present publication are provided for the convenience of the reader and are correct at the time of issue. The United Nations takes no responsibility for the continued accuracy of that information or for the content of any external website. -

Uranium-Free X Solution
www.nature.com/scientificreports OPEN Uranium‑free X solution: a new generation contrast agent for biological samples ultrastructure Aldo Moscardini1,6, Sebastiano Di Pietro 2,6, Giovanni Signore3*, Paola Parlanti4, Melissa Santi5, Mauro Gemmi5 & Valentina Cappello5* Biological samples are mainly composed of elements with a low atomic number which show a relatively low electron scattering power. For Transmission Electron Microscopy analysis, biological samples are generally embedded in resins, which allow thin sectioning of the specimen. Embedding resins are also composed by light atoms, thus the contrast diference between the biological sample and the surrounding resin is minimal. Due to that reason in the last decades, several staining solutions and approaches, performed with heavy metal salts, have been developed with the purpose of enhancing both the intrinsic sample contrast and the diferences between the sample and resin. The best staining was achieved with the uranyl acetate (UA) solution, which has been the election method for the study of morphology in biological samples. More recently several alternatives for UA have been proposed to get rid of its radiogenic issues, but to date none of these solutions has achieved efciencies comparable to UA. In this work, we propose a diferent staining solution (X Solution or X SOL), characterized by lanthanide polyoxometalates (LnPOMs) as heavy atoms source, which could be used alternatively to UA in negative staining (NS), in en bloc staining, and post sectioning staining (PSS) of biological samples. Furthermore, we show an extensive chemical characterization of the LnPOM species present in the solution and the detailed work for its fnal formulation, which brought remarkable results, and even better performances than UA. -

Circular of the Bureau of Standards No
t9?2 fcixt .Wii * S/ DEPARTMENT OF COMMERCE BUREAU OF STANDARDS S. W. STRATTON, Director CIRCULAR OF THE BUREAU OF STANDARDS No. Ill [Issued, March 11, 1921] RECOMMENDED SPECIFICATION FOR FLAT INTERIOR LITHOPONE PAINT, WHITE AND LIGHT TINTS PREPARED AND RECOMMENDED BY THE U. S. INTERDEPARTMENTAL COM- MITTEE ON PAINT SPECIFICATION STANDARDIZATION, JANUARY 21, 1921. P. H. WALKER, BUREAU OF STANDARDS, CHAIRMAN; J. W. GINDER, TREAS- URY DEPARTMENT, SECRETARY [This committee was appointed at the suggestion of the Secretary of Commerce, and consisted of repre- sentatives of the War, Navy, Agriculture, Interior, Post Office, Treasury, and Commerce Departments, the Panama Canal, and the Educational Bureau of the Paint Manufacturers* Association of the United States. The committee submitted a preliminary draft of the specification to a large number of representa- tives of the paint manufacturers, and gave careful consideration to the replies received.] CONTENTS Page 1. General 1 2 . Sampling 2 3 . Laboratory examination 3 4 . Analysis of pigment 5 5 . Reagents 7 1. GENERAL This specification covers ready-mixed lithopone paints, fre- quently known as flat, washable wall paint, in white and a variety of light tints. Paints under this specification are not intended for outside exposure; they shall dry to dead flat opaque coats that will adhere well to wood, metal, and plaster, stand washing with soap and water, and show no material change in color on exposure to light. The paint shall be purchased by volume (231 cubic inches to the gallon). 34615°—21 T 2 Circular of the Bureau of Standards (a) Pigment.—The pigment shall consist of: Maximum Minimum Per cent Per cent Lithopone 80 Zinc oxide 10 Tinting and extending pigments 10 Material soluble in water 0.8 Note.— he lithopone used must contain not less than 26 per cent of zinc sulphide and must not darken on exposure. -

United States Pmao" ICC Patented June 30, '1959 1 2 Adsorption on Manganese Dioxide, but It May on the 2,892,677 Other Hand He Left in the Solution
r. 2,892,677 United States Pmao" ICC Patented June 30, '1959 1 2 adsorption on manganese dioxide, but it may on the 2,892,677 other hand he left in the solution. If it is removed, a certain amount of manganese nitrate is formed from the ' SEPARATION OF URANIUM FROM THORIUM manganese dioxide in the solution and this nitrate reacts AND PROTACTINIUM with the sodium diethyldithiocarbamate to give manga William Kenneth Rodgerson Musgrave, Durham, Eng ' nese diethyldithiocarbamate, which must subsequently be land, assignor, by mesne assignments, to the United separated from the uranium. ,, . .States of America as represented by the United States Whether or not the protactinium has been removed, Atomic Energy Commission the solution is brought to a pH of between 2 and 3, for No Drawing. Application November 27, 1946 10 example by the addition of ammonia. The sodium di Serial No. 712,722 ethyldithiocarbamate is then dissolved in a solvent in which the subsequently formed uranyl diethyldithiocar '8 Claims. (Cl. 23—14.5) bamate is also soluble. Such a solvent is, for example, amyl acetate or methyl isobutyl ketone, the former being This invention relates to the separation of uranium 15 preferable for the reasons which will be indicated below. from thorium and protactinium. A mixture of these The sodium diethyldithiocarbamate is dissolved in amyl elements is obtained, for example, as the result of irradia acetate to form a solution containing 0.25% of the tion by neutrons of so-called thorium carbonate, which former, and this solution is then shaken up with the is a mixture of thorium oxide and thorium carbonate. -

Solvent Extraction of Uranium with Acetylacetone and Tri-N-Butyl
DAEHAN HWAHAK HWOEJEE (Journal of the Korean Chemical Society) Vol. 24, No. 3, 1980 Printed in the Republic of Korea 아세 틸아세톤과 트리부틸인산의 도데칸용액 에 의한 우라늄의 용매추출 裴奎善-鄰奇碩 韓國核燃料開發公團 (1980. 2. 27 接受 ) Solvent Extraction of Uranium with Acetylacetone and Tri-n-Butyl Phosphate in “-Dodecane Kyu Sun Bai and Key Suck Jung Korea Nuclear Fuel Development Institute, Daeduck 300-32, Korea (Received Feb. 27, 1980) 요 악 . 도데칸에 녹인 아세틸아세톤과 트리부틸인산으로 묽은 질산우라닐수용액에서 우라늄 (VI) 을 추출했다 . pHl 이상에서 이 혼합추출제의 상승적 효과가 관측되었다 . 추출되는 화학종은 1:2:1 및 1:2:2 우라닐 -아세털 아세톤 -트리부털 인산착물이다 . 이들 반응의 추출정수들을 측정하였다 . ABSTRACT. Uranium (VI) was extracted from dilute aqueous solutions of uranyl nitrate with acetylacetone and tri-n-butyl phosphate in n-dodecane. Synergistic effect was observed with the mixed reagents above pH 1, The species extracted are the 1:2:1 and the 1:2:2 uranyl-AA-TBP complexes. The extraction constants for these reactions have been determined. by crystallization of the salt as UO2(NO3)2 INTRODUCTION •6H2O. HAA and TBP were dissolved in do Acetylacetone (HAA) is a simple -diketone. decane and uranyl nitrate in water. Equal A weak acid with pKa=8. 2% HAA has long volumes of organic and aqueous phases were been used as a chelating agent for many equilibrated in 100 mZ separatory funnels by metals.2 It is quite soluble in water and its vigorous shaking for several minutes. Equili brium temperature was 25 °C or ambient. pH distribution coefficient is E=5. 95 for benzene/ water system.3 Because of its solubility in measurements were made by using a Corning water, HAA has not been so widely used as, Model 130 pH meter. -

Uranium Waste Focus Sheet
URANIUM WASTE Read below for information about safe packaging, labeling and disposing of uranium and thorium compounds. DESCRIPTION LABEL Uranium waste consists of solids and liquids Ensure container is contaminated with uranium and thorium compounds. properly labelled with Examples include uranyl acetate, uranyl nitrate, information about the uranyl formate and thorium nitrate. uranium or thorium compound, concentrations and a “Caution Radioactive Materials” sticker. DISPOSE Uranyl acetate and similar compounds are generally licensed, however any liquid or solid waste must be disposed of as radiological waste. Due to toxicity element of most Uranium and STORE Thorium compounds, liquid waste may be designated as Mixed Waste. Labs planning or concerned about generating uranium waste must consult with Solids Radiation Safety for disposal pricing, guidelines and For contaminated solids, designate an appropriate alternative options. sized container in a secure area (e.g., back of a fume hood in a locked lab). Label the container with a Uranium and thorium solid waste, powders or “Caution Radioactive Material” sticker. crystals will be collected by Radiation Safety for disposal. Liquids Store contaminated liquids in an appropriate strong To arrange a pick-up of uranium waste, complete a plastic container in secondary containment. Keep the Radioactive Waste Collection Request. waste container close to your work area to minimize chance of spilling. When not working, place waste in a posted and secure storage area. Liquid waste containers must always be properly labelled, and securely closed when not in use. Please contact EH&S Radiation Safety at 206.543.0463 or [email protected] for more information. Page 1 | September 2019 www.ehs.washington.edu | 206.543.7262 | [email protected] . -

Uranium Toxicity and Chelation Therapy
Pure Appl. Chem. 2014; 86(7): 1105–1110 Conference paper Glen D. Lawrence*, Kamalkumar S. Patel and Aviva Nusbaum Uranium toxicity and chelation therapy Abstract: Uranium toxicity has been a concern for more than 100 years. The toxicology of many forms of uranium, ranging from dust of several oxides to soluble uranyl ion, was thoroughly studied during the Man- hattan Project in the United States in the 1940s. The development of depleted uranium kinetic penetrators as armor-piercing incendiary weaponry produced a novel form of uranium environmental contamination, which led to greater susceptibility to the adverse health effects of the toxic heavy metal after its use in various military conflicts. The aerosol from burning uranium penetrator fragments is rapidly dissolved in biological fluids and readily absorbed from the lungs, leading to a wide range of toxic effects. We have studied some chelating agents for uranyl ion, including citrate ion and desferal (desferrioxamine B), which may be effective for minimizing the toxic effects of this insidious heavy metal. Some characteristics of the desferrioxamine complex are presented, along with information about the use of citrate as an effective chelating agent for therapy of uranium toxicity. Keywords: bioactivity; environmental chemistry; IUPAC Congress-44; metal complexes; solubility; toxi- cology; uranium. DOI 10.1515/pac-2014-0109 Introduction Uranium is the heaviest naturally occurring element in the earth’s crust; it is more abundant than gold or silver. Although discovered in 1789, it found few commercial uses for a century and a half beyond its use as a coloring agent for pottery and glass. Radioactivity was first discovered in uranium in 1896, soon after the discovery of X-rays. -

Material Safety Data Sheet
SAFETY DATA SHEET URANYL NITRATE SOLUTION SECTION 1: CHEMICAL PRODUCT & COMPANY IDENTIFICATION New Brunswick Laboratory U.S. Department of Energy 9800 South Cass Avenue Argonne, IL 60439 1-630-252-CRMS (2767) Emergency Phone Numbers: 1-630-252-6130 or 1-630-252-5731 Chemical Name: Uranyl Nitrate Solution, UO2(NO3)2 Other Identifiers: Certified Reference Material (CRM) standard or Safeguards Measurement Evaluation (SME) sample Use and Restrictions: This material is prepared for use as a standard or in inter-laboratory comparison programs at analytical laboratories, which routinely handle uranium and/or plutonium. NBL expects that recipients of their material are in compliance with 29 CFR 1910.1200(h) which requires employers to provide employees with effective information and training on hazardous chemicals in their work area. SECTION 2: HAZARDS IDENTIFICATION OSHA HAZARDS: Skin Corrosion/Irritation Category 1B. Serious Eye Damage/Irritation Category 1 TARGET ORGANS: Skin, Eyes, Kidneys. GHS Label elements, including precautionary statements Signal Word: DANGER Pictogram: Page 1 of 12 Hazard Statement(s) H314 Causes severe skin burns and eye damage H373 May cause damage to organs through prolonged or repeated exposure. H411 Toxic to aquatic life with long lasting effects. Precautionary statement(s) P260 Do not breathe dust/ fume/ gas/ mist/ vapors/ spray. P262 Do not get in eyes, on skin, or on clothing. P264 Wash skin thoroughly after handling. P273 Avoid release to the environment. P280 Wear protective gloves/protective clothing/eye protection/face protection P310 Immediately call a POISON CENTER or doctor/ physician if swallowed or inhaled. Other Hazard(s): Radioactive NFPA RATINGS (SCALE 0-4): Health=3 Fire=0 Reactivity=0 Special Hazard= OX SECTION 3: COMPOSITION/INFORMATION ON INGREDIENTS Chemical Name: Uranyl Nitrate Solution Common Names/Synonyms: CRM U045; CRM 135; CRM 145; CRM 145-B; CRM U930-D; Safeguards Measurement Evaluation (SME) Low Enrichment, Normal Enrichment, or High Enrichment Solutions; Uranyl Nitrate in Nitric Acid Solution. -

Uranyl Nitrate, 1%
Uranyl Nitrate, 1% 1. Identification Product Name: Uranyl Nitrate, 1% Item #: DI0103014, DI0103029, SKC1048-250, SKC1048-500 Web SDS: S231 Synonyms: N/A Recommended Use: Special Stains Restrictions on Use: N/A Manufacturer: In Case of Emergency: BBC Biochemical Chemtrec US 1-800-424-9300 409 Eleanor Lane, Chemtrec International 703-527-3887 Mount Vernon, WA 98273 1-800-635-4477 2. Hazards Identification OSHA Hazard Classification(s): Acute Toxicity - Oral - Category 4 Acute Toxicity - Inhalation - Category 4 Signal Word: Warning Hazard Statement(s): Harmful if swallowed. Harmful if inhaled. Pictogram(s): Precautionary Statement(s): Prevention: Wash body thoroughly after handling. Do not eat, drink or smoke when using this product. Avoid breathing dust, vapors. Use only outdoors or in a well-ventilated area. Response: If swallowed: Call a doctor if you feel unwell. Rinse mouth. If inhaled: Remove person to fresh air and keep comfortable for breathing. Call a doctor if you feel unwell. Storage: N/A Disposal: Dispose of contents/container in accordance with local regulations. Descriptions of Hazards not otherwise classified: N/A Percent of mixture with unknown acute toxicity: N/A 3. Composition and Information on Ingredients Chemical Name Common Name CAS # Concentration % Uranyl Nitrate 13520-83-7 1 Water 7732-18-5 99 4. First Aid Measures Eye Contact: If in eyes: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. If eye irritation persists: Get medical advice/attention. Skin Contact: If on skin (or hair): Take off immediately all contaminated clothing and wash before reuse. -

Physcio-Chemical Studies on the Systems Uranyl Nitrate Organic Solvent
Durham E-Theses Physcio-chemical studies on the systems uranyl nitrate organic solvent - water Mathieson A. R., How to cite: Mathieson A. R., (1951) Physcio-chemical studies on the systems uranyl nitrate organic solvent - water, Durham theses, Durham University. Available at Durham E-Theses Online: http://etheses.dur.ac.uk/9766/ Use policy The full-text may be used and/or reproduced, and given to third parties in any format or medium, without prior permission or charge, for personal research or study, educational, or not-for-prot purposes provided that: • a full bibliographic reference is made to the original source • a link is made to the metadata record in Durham E-Theses • the full-text is not changed in any way The full-text must not be sold in any format or medium without the formal permission of the copyright holders. Please consult the full Durham E-Theses policy for further details. Academic Support Oce, Durham University, University Oce, Old Elvet, Durham DH1 3HP e-mail: [email protected] Tel: +44 0191 334 6107 http://etheses.dur.ac.uk PSYSIC0-CH3MICAL STUDIES OH THE SYSTEMS DEAUTL 1TITRATE - ORGANIC SOLVENT - WATER A.R. Mathieson, B.Sc.(Dunelm), A.R.I.C. Thesis presented for the degree of M. Sc. in Pure Science of the University of Durham, August 1951 The experimental work described in this thesis was performed at the Atomic Energy Research Establishment, Harwell, during the period November 1947 - December 1949* The results have already been published elsewhere: Mathieson "Stability of Complexes of Uranyl Nitrate with Ketones and Ethers", J.