Citicoline (CDP-Choline)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Role of Citicoline in the Management of Traumatic Brain Injury
pharmaceuticals Review Role of Citicoline in the Management of Traumatic Brain Injury Julio J. Secades Medical Department, Ferrer, 08029 Barcelona, Spain; [email protected] Abstract: Head injury is among the most devastating types of injury, specifically called Traumatic Brain Injury (TBI). There is a need to diminish the morbidity related with TBI and to improve the outcome of patients suffering TBI. Among the improvements in the treatment of TBI, neuroprotection is one of the upcoming improvements. Citicoline has been used in the management of brain ischemia related disorders, such as TBI. Citicoline has biochemical, pharmacological, and pharmacokinetic characteristics that make it a potentially useful neuroprotective drug for the management of TBI. A short review of these characteristics is included in this paper. Moreover, a narrative review of almost all the published or communicated studies performed with this drug in the management of patients with head injury is included. Based on the results obtained in these clinical studies, it is possible to conclude that citicoline is able to accelerate the recovery of consciousness and to improve the outcome of this kind of patient, with an excellent safety profile. Thus, citicoline could have a potential role in the management of TBI. Keywords: CDP-choline; citicoline; pharmacological neuroprotection; brain ischemia; traumatic brain injury; head injury Citation: Secades, J.J. Role of 1. Introduction Citicoline in the Management of Traumatic brain injury (TBI) is among the most devastating types of injury and can Traumatic Brain Injury. result in a different profile of neurological and cognitive deficits, and even death in the most Pharmaceuticals 2021, 14, 410. -

Choline for a Healthy Pregnancy
To support healthy for a Healthy weight gain and keep up with the nutritional needs of both mom and Pregnancy the developing baby, CHOLINE additional nutrients are necessary. Nine out of 10 Americans don’t meet the daily recommended choline intake of 550 mg1,2 and it can be challenging to reach this goal even when choosing choline-containing foods like beef, eggs, wheat germ and Brussels sprouts. Choline is particularly important during pregnancy for both mom and baby because it supports healthy brain growth and offers protection against neural tube defects. Women are encouraged to take a prenatal supplement before and during pregnancy to ensure they’re meeting vitamin and mineral recommendations. In fact, the American Medical Association recommends that choline be included in all prenatal vitamins to help ensure women get enough choline to maintain a normal pregnancy.3 Look for a prenatal supplement that contains folic acid, iron, DHA (omega-3s), vitamin D and choline. Consider smart swaps to get the most choline in your diet for a healthy pregnancy, as well as optimal health after baby arrives. PREGNANCY EATING PATTERN* CHOLINE-FOCUSED PREGNANCY EATING PATTERN* 1 1 hard-cooked egg 1 2 cups toasted whole grain oat cereal / 1 large peach 1 cup nonfat milk 1 1 slice whole grain bread /3 cup blueberries 1 1 tablespoon jelly /3 cup sliced banana BREAKFAST 1 cup nonfat milk 1 /2 whole grain bagel 1 whole wheat tortilla 2 tablespoons peanut butter 2 tablespoons peanut butter 1 small apple 1 SNACK 1 /2 large banana /2 cup nonfat vanilla Greek yogurt 2 slices whole grain bread 3 oz. -

Relief of Hypersensitivity After Nerve Injury from Systemic Donepezil
Saranya devi Relief of Hypersensitivity after Nerve Injury from ALN Systemic Donepezil Involves Spinal Cholinergic and γ-Aminobutyric Acid Mechanisms Spinal ACh and GABA Mechanisms for Donepezil Analgesia Masafumi Kimura, M.D.,* Ken-ichiro Hayashida, D.V.M., Ph.D.,† James C. Eisenach, M.D.,‡ KIMURA ET AL. Shigeru Saito M.D.,§ Hideaki Obata, M.D.‖ XX ABSTRACT What We Already Know about This Topic • Donepezil increases central nervous system acetylcholine lev- Downloaded from http://pubs.asahq.org/anesthesiology/article-pdf/118/1/173/259568/0000542-201301000-00030.pdf by guest on 28 September 2021 10.1097/ALN.0b013e318277a81c Background: Evoking spinal release of acetylcholine (ACh) els and is used for treatment of dementia produces antinociception in normal animals and reduces • Increases in spinal acetylcholine levels have been implicated in the relief of neuropathic pain hypersensitivity after nerve injury, and some studies suggest January that ACh-mediated analgesia relies on γ-aminobutyric acid (GABA)-ergic signaling in the spinal cord. In this study, the authors tested the spinal mechanisms underlying the anti- What This Article Tells Us That Is New 118 hypersensitivity effects of donepezil, a central nervous sys- • Donepezil increased spinal acetylcholine levels and γ- tem–penetrating cholinesterase inhibitor, in a rat model of aminobutyric acid levels to reduce nociceptive responses after nerve injury and represents a potential therapeutic pathway to neuropathic pain. reduce pain after nerve injury Methods: Male Sprague-Dawley rats were anesthetized, and L5 spinal nerve ligation was performed unilaterally. With- A antagonist; and CGP 35348 (30 μg), a γ-aminobutyric drawal threshold to a paw pressure test was measured before acid receptor type B antagonist. -

Eletriptan Smpc
For the use only of a neurologist/ specialist or a Hospital or a Laboratory Generic Name Citicoline Trade Name Ceham 1. NAME OF THE MEDICINAL PRODUCT: Citicoline 2. QUALITATIVE AND QUANTITATIVE COMPOSITION: Each film coated tablet contains: Citicoline sodium equivalent to Citicoline……………………………500mg 3. PHARMACEUTICAL FORM: Film coated tablet 4. CLINICAL PARTICULARS 4.1 Therapeutic Indications: For the treatment of disturbance of consciousness as resulting from head injuries, brain operation and acute stage of cerebral infarction in adult only. For the treatment of patients with serious cerebral injuries of vascular traumatic nature with or without loss of consciousness & for treatment of degenerative damages & chronic cerebral vascular injuries in senile dementia. 4.2 Posology and Method of Administration The usually recommended dose of Citicoline is 500mg to 1000mg daily and can increase up to 2000mg. Elderly: No dosage adjustment is required in this patient population and the usually recommended adult dose can be administered. 4.3 Contraindications Hypersensitivity to citicoline or any other component of the formulation 4.4 Special Warnings and Special Precautions for Use: Citicoline may cause hypotension and in case necessary the hypotensive effect can be treated with corticosteroids or sympathomimetics. 4.5 Interaction with Other Medicinal Products and Other Forms of Interaction: Citicoline must not be used with medicines containing meclophenoxates (or centrophenoxine). Citicoline increases the effects of L-dopa. 4.6 Fertility, Pregnancy and Lactation There are no adequate and well controlled studies of citicoline during pregnancy and lactation. Citicoline should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. -

More Treatments on Deck for Alcohol Use Disorder
News & Analysis Medical News & Perspectives More Treatments on Deck for Alcohol Use Disorder Jeff Lyon hirteen years have passed since the Raye Z. Litten, PhD, acting director of the ment for AUD were actually prescribed an US Food and Drug Administration NIAAA’s Division of Medications Develop- AUD medication. T (FDA) last approved a new medica- ment and principal investigator of the trial, It is a complicated problem, says John tion to help the nation’s millions of people says the agency is “excited.” Rotrosen, MD, a psychiatrist and addiction withalcoholusedisorder(AUD)stopormod- Nevertheless, the science of alcohol ad- specialist at New York University School of erate their drinking. diction has seen its share of medicines that Medicine. “To a large extent primary care Only 3 such formulations exist, and 1, failed to live up to their early promise dur- physicians don’t screen for alcoholism,” he disulfiram, dates to the Prohibition era. ing full-scale testing. So Litten said the said. “When they do, they may note it, but Known commercially as Antabuse and agency isn’t putting all its eggs in one bas- don’t really do anything about it.” introduced in 1923, it makes people memo- ket. “You need as many weapons as pos- As far as the failure to prescribe exist- rably ill if they ingest alcohol, but it doesn’t sible to treat a complex disease like alcohol ing medications for AUD, Rotrosen blames stop the cravings. The other 2, naltrexone use disorder,” he said. a lack of knowledge. “A lot of doctors in and acamprosate, approved in 1994 and But therein lies a second difficulty. -

Tor Amendment Tagoor Laboratories Private Limited
ToR Amendment Tagoor Laboratories Private Limited ANNEXURE – I Details of Products as per Granted ToR and Amendment Required Production Capacity S. Ton /Month Therapeutic Product Name No As per Amendment category granted ToR Required 1 Abacavir sulfate 2.0 2.0 Anti HIV 2 Amitriptyline 5.0 10.0 Antidepressant 3 Atrovastatin Calcium 5.0 5.0 Hypercholesterolemia 4 Bupropion 5.0 5.0 Anti depressant 5 carisoprodol 2.0 2.0 Muscle Relaxant 6 Clopidogrelbisulfate 5.0 5.0 Antithrombotic 7 Cyclobenzaprine HCl 5.0 5.0 Muscle relaxant 8 Cyproheptadine HCl 5.0 10.0 Anti allergic 9 Desloratadine 5.0 5.0 Antihistamine 10 Domperidone 20.0 30.0 Anti emetic 11 Domperidone maleate 2.0 2.0 Anti emetic 12 Donepezil HCl 1.0 1.0 Alzheimer’s disease 13 Ebastine 5.0 5.0 Anti allergic 14 Esomeprazole Sodium 3.0 3.0 Anti ulcerative Esomeprazole Magnesium 3.0 3.0 Anti ulcerative 15 trihydrate 16 Fexofenadine Hydrochloride 6.0 15.0 Anti histamine 17 Haloperidol 2.0 2.0 Antipsychotic 18 Itopride Hydrochloride 2.0 2.0 Antispasmodics 19 Itraconazole 10.0 10.0 Antifungal 20 Ketrolac Tromethane 2.0 2.0 Anti Inflammatory 21 Lansoprazole 10.0 10.0 Ant ulcerative 22 Loperamide Hydrochloride 10.0 10.0 Anti diarrhea agent 23 Losartan Potassium 6.0 15.0 Anti Hypertensive 24 Nebivolol HCl 2.0 2.0 Anti Hypertensive 25 Nortriptyline HCl 2.0 2.0 Anti depressant 26 Omeprazole 40.0 40.0 Anti ulcerative 27 Omeprazole Sodium 2.0 2.0 Ant ulcerative Omeprazole Magnesium 2.0 2.0 Ant ulcerative 28 Dihydrate 29 Oxatomide 1.0 1.0 Antihistamine Pantoprazole Sodium Sesqui 10.0 20.0 Ant ulcerative 30 Hydrate 31 Pimozide 2.0 2.0 Antipsychotic 32 Pregabalin 2.0 2.0 Epileptic 33 Quetiapine Hemifumarate 2.0 2.0 Antipsychotic Prepared By Rightsource Industrial Solutions Pvt. -

Effect of Combined Therapy with Thrombolysis and Citicoline in a Rat
Journal of the Neurological Sciences 247 (2006) 121–129 www.elsevier.com/locate/jns Effect of combined therapy with thrombolysis and citicoline in a rat model of embolic stroke ⁎ María Alonso de Leciñana a,e, , María Gutiérrez a,d, Jose María Roda a,c, Fernando Carceller a,c, Exuperio Díez-Tejedor a,b a Cerebrovascular Research Unit, La Paz University Hospital, Universidad Autónoma de Madrid, Madrid, Spain b Department of Neurology, La Paz University Hospital, Madrid, Spain c Department of Neurosurgery, La Paz University Hospital, Madrid, Spain d Cerebrovascular Experimental Lab, La Paz University Hospital, Madrid, Spain e Department of Neurology, Ramón y Cajal University Hospital, Madrid, Spain Received 28 April 2005; received in revised form 3 January 2006; accepted 3 March 2006 Available online 22 June 2006 Abstract An approach combining reperfusion mediated by thrombolytics with pharmacological neuroprotection aimed at inhibiting the physiopathological disorders responsible for ischemia-reperfusion damage, could provide an optimal treatment of ischemic stroke. We investigate, in a rat embolic stroke model, the combination of rtPA with citicoline as compared to either alone as monotherapy, and whether the neuroprotector should be provided before or after thrombolysis to achieve a greater reduction of ischemic brain damage. One hundred and nine rats have been studied: four were sham-operated and the rest embolized in the right internal carotid artery with an autologous clot and divided among 5 groups: 1) control; 2) iv rtPA 5 mg/kg 30 min post-embolization 3) citicoline 250 mg/kg ip ×3 doses, 10 min, 24 h and 48 h post-embolization; 4) citicoline combined with rtPA following the same pattern; 5) rtPA combined with citicoline, with a first dose 10 min after thrombolysis. -

Brain Choline Acetyltransferase Activity in Chronic, Human Users of Cocaine
Molecular Psychiatry (1999) 4, 26–32 1999 Stockton Press All rights reserved 1359–4184/99 $12.00 ORIGINAL RESEARCH ARTICLE Brain choline acetyltransferase activity in chronic, human users of cocaine, methamphetamine, and heroin SJ Kish1, KS Kalasinsky2, Y Furukawa1, M Guttman1, L Ang3,LLi4, V Adams5, G Reiber6, RA Anthony6, W Anderson7, J Smialek4 and L DiStefano1 1Human Neurochemical Pathology Laboratory, Centre for Addiction and Mental Health, Toronto, Canada; 2Division of Forensic Toxicology, Armed Forces Institute of Pathology, Washington, DC, USA; 3Department of Pathology (Neuropathology), Sunnybrook Hospital, Toronto, Canada; 4Department of Pathology, University of Maryland, Baltimore, MD; 5Office of the Hillsborough County Medical Examiner, Tampa, FL; 6Northern California Forensic Pathology, Sacramento, CA; 7Office of the Medical Examiner of District 9, Orlando, FL, USA Cognitive impairment has been reported in some chronic users of psychostimulants, raising the possibility that long-term drug exposure might damage brain neuronal systems, including the cholinergic system, which are responsible for normal cognition. We measured the activity of choline acetyltransferase (ChAT), the marker enzyme for cholinergic neurones, in autopsied brain of chronic users of cocaine, methamphetamine, and, for comparison, heroin. As com- pared with the controls, mean ChAT levels were normal in all cortical and subcortical brain areas examined. However, the two of 12 methamphetamine users, who had the highest brain/blood drug levels at autopsy, had a severe (up to 94%) depletion of ChAT activity in cerebral cortex, striatum, and thalamus. Based on the subjects examined in the present study, our neurochemical data suggest that brain cholinergic neurone damage is unlikely to be a typical feature of chronic use of cocaine, methamphetamine, or heroin, but that exposure to very high doses of methamphetamine could impair, at least acutely, cognitive function requir- ing a normal nucleus basalis cholinergic neuronal system. -

Anticraving Therapy for Alcohol Use Disorder: a Clinical Review
Received: 10 April 2018 | Revised: 11 June 2018 | Accepted: 11 June 2018 DOI: 10.1002/npr2.12028 INVITED REVIEW Anticraving therapy for alcohol use disorder: A clinical review Winston W. Shen1,2 1Department of Psychiatry, Wan Fang Medical Center, Taipei Medical University, Abstract Taipei, Taiwan Aim: In this review, the author focused on anticraving therapy for alcohol use disor- 2Department of Psychiatry, College of der (AUD) defined by DMS‐5. A comprehensive review was carried out on the avail- Medicine, Taipei Medical University, Taipei, Taiwan able published papers on anticraving drugs for treating AUD patients. Methods: The author described all drugs with anticraving benefits for treating Correspondence Winston W. Shen, Department of AUD patients approved by the Food and Drug Administration of the United States Psychiatry, Wan Fang Medical Center, No. (US FDA) and European Medicines Agency of the European Union. Then, the com- 111, Section 3, Hsing Long Road, Taipei 116, Taiwan. monly prescribed anticraving drugs and those under development were also Email: [email protected] described. Results: The US FDA‐approved anticraving drugs included acamprosate and naltrex- one, and those approved by European Medicines Agency were gamma‐hydroxybuty- rate and nalmefene. The author also highlighted topiramate, gabapentin, ondansetron, LY196044, ifenprodil, varenicline, ABT‐436, mifepristone, citicoline, and baclofen. The putative mechanisms of action of and the use in clinical practice of those anticraving drugs were also described. Conclusion: Although slowly developing, the field of anticraving drugs is getting into shape as a promising entity of a pharmaceutical class of drugs. Then, the author addressed on the underused issues of those recommended, and suggested anticraving drugs by the practice guideline of the American Psychiatric Association. -
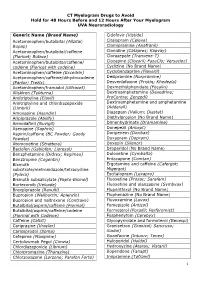
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology Generic Name (Brand Name) Cidofovir (Vistide) Acetaminophen/butalbital (Allzital; Citalopram (Celexa) Bupap) Clomipramine (Anafranil) Acetaminophen/butalbital/caffeine Clonidine (Catapres; Kapvay) (Fioricet; Butace) Clorazepate (Tranxene-T) Acetaminophen/butalbital/caffeine/ Clozapine (Clozaril; FazaClo; Versacloz) codeine (Fioricet with codeine) Cyclizine (No Brand Name) Acetaminophen/caffeine (Excedrin) Cyclobenzaprine (Flexeril) Acetaminophen/caffeine/dihydrocodeine Desipramine (Norpramine) (Panlor; Trezix) Desvenlafaxine (Pristiq; Khedezla) Acetaminophen/tramadol (Ultracet) Dexmethylphenidate (Focalin) Aliskiren (Tekturna) Dextroamphetamine (Dexedrine; Amitriptyline (Elavil) ProCentra; Zenzedi) Amitriptyline and chlordiazepoxide Dextroamphetamine and amphetamine (Limbril) (Adderall) Amoxapine (Asendin) Diazepam (Valium; Diastat) Aripiprazole (Abilify) Diethylpropion (No Brand Name) Armodafinil (Nuvigil) Dimenhydrinate (Dramamine) Asenapine (Saphris) Donepezil (Aricept) Aspirin/caffeine (BC Powder; Goody Doripenem (Doribax) Powder) Doxapram (Dopram) Atomoxetine (Strattera) Doxepin (Silenor) Baclofen (Gablofen; Lioresal) Droperidol (No Brand Name) Benzphetamine (Didrex; Regimex) Duloxetine (Cymbalta) Benztropine (Cogentin) Entacapone (Comtan) Bismuth Ergotamine and caffeine (Cafergot; subcitrate/metronidazole/tetracycline Migergot) (Pylera) Escitalopram (Lexapro) Bismuth subsalicylate (Pepto-Bismol) Fluoxetine (Prozac; Sarafem) -

Choline an Essential Nutrient for Public Health
Continuing Education Elevating Awareness and Intake of Choline An Essential Nutrient for Public Health Marie Caudill, PhD, RD Steven Zeisel, MD, PhD Kerry-Ann da Costa, PhD Betsy Hornick, MS, RD Emerging science has revealed that choline plays important Choline is the newest roles in health throughout the life cycle with potentially essential nutrient. serious health consequences associated with inadequate choline intakes. Recent national consumption data indicate that the vast majority of Americans are falling short of recommended intakes. Increased education, including In 2009, the Choline Science Summit was held in Washington, District of Columbia, bringing together recommendations to consume more choline-rich foods, prominent scientists and researchers along with nutrition is needed to improve choline intakes for optimal health. leaders from organizations including the American Nutr Today. 2011;46(5):235–241 College of Obstetricians and Gynecologists, American Dietetic Association, American Society for Nutrition, International Life Sciences Institute, National WIC Association, and US Department of Agriculture (USDA). holine was discovered as a vitamin in 1862, The symposium addressed the latest science on choline but it was more than a century later that it and gathered insights for helping raise awareness Cwas recognized as an essential nutrient. It was and intake of this essential nutrient, especially among officially recognized as an essential nutrient by the vulnerable populations. It included discussions of Institute of Medicine (IOM) in 1998, and an adequate choline’s critical role in human health and development intake (AI) was established based on estimated throughout the life cycle, the role of genetics in dietary intakes and studies reporting liver damage determining choline requirements, and a closer look with lower choline intakes.1 Choline is essential for at the gaps in choline requirements compared with liver and brain function, lipid metabolism, and for actual intakes. -

The Relationship Between Choline Bioavailability from Diet, Intestinal Microbiota Composition, and Its Modulation of Human Diseases
nutrients Review The Relationship between Choline Bioavailability from Diet, Intestinal Microbiota Composition, and Its Modulation of Human Diseases Natalia Arias 1,2,*, Silvia Arboleya 3 , Joseph Allison 2, Aleksandra Kaliszewska 2, Sara G. Higarza 1,4 , Miguel Gueimonde 3 and Jorge L. Arias 1,4 1 Instituto de Neurociencias del Principado de Asturias (INEUROPA), 33003 Oviedo, Asturias, Spain; [email protected] (S.G.H.); [email protected] (J.L.A.) 2 Department of Basic and Clinical Neuroscience, Institute of Psychiatry, Psychology and Neuroscience, King’s College London, Denmark Hill, London SE5 8AF, UK; [email protected] (J.A.); [email protected] (A.K.) 3 Department of Microbiology and Biochemistry of Dairy Products, Instituto de Productos Lácteos de Asturias (IPLA-CSIC), 33003 Oviedo, Asturias, Spain; [email protected] (S.A.); [email protected] (M.G.) 4 Laboratory of Neuroscience, Department of Psychology, University of Oviedo, Plaza Feijóo, s/n, 33003 Oviedo, Asturias, Spain * Correspondence: [email protected] Received: 14 July 2020; Accepted: 30 July 2020; Published: 5 August 2020 Abstract: Choline is a water-soluble nutrient essential for human life. Gut microbial metabolism of choline results in the production of trimethylamine (TMA), which, upon absorption by the host is converted into trimethylamine-N-oxide (TMAO) in the liver. A high accumulation of both components is related to cardiovascular disease, inflammatory bowel disease, non-alcoholic fatty liver disease, and chronic kidney disease. However, the relationship between the microbiota production of these components and its impact on these diseases still remains unknown. In this review, we will address which microbes contribute to TMA production in the human gut, the extent to which host factors (e.g., the genotype) and diet affect TMA production, and the colonization of these microbes and the reversal of dysbiosis as a therapy for these diseases.