A Genomewide Functional Network for the Laboratory Mouse
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Parkin Antibody (C-Term) Purified Rabbit Polyclonal Antibody (Pab) Catalog # AP6402B
10320 Camino Santa Fe, Suite G San Diego, CA 92121 Tel: 858.875.1900 Fax: 858.622.0609 Parkin Antibody (C-term) Purified Rabbit Polyclonal Antibody (Pab) Catalog # AP6402B Specification Parkin Antibody (C-term) - Product Information Application IF, WB, IHC-P, FC,E Primary Accession O60260 Other Accession NP_004553 Reactivity Human, Mouse Host Rabbit Clonality Polyclonal Isotype Rabbit Ig Antigen Region 387-417 Parkin Antibody (C-term) - Additional Information Gene ID 5071 Other Names E3 ubiquitin-protein ligase parkin, 632-, Confocal immunofluorescent analysis of Parkinson juvenile disease protein 2, Parkin Antibody (C-term)(Cat#AP6402b) with Parkinson disease protein 2, PARK2, PRKN NCI-H460 cell followed by Alexa Fluor 488-conjugated goat anti-rabbit lgG Target/Specificity (green).Actin filaments have been labeled This Parkin antibody is generated from with Alexa Fluor 555 phalloidin (red).DAPI rabbits immunized with a KLH conjugated was used to stain the cell nuclear (blue). synthetic peptide between 387-417 amino acids from the C-terminal region of human Parkin. Dilution IF~~1:10~50 WB~~1:1000 IHC-P~~1:50~100 FC~~1:10~50 Format Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. Storage Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C Park2 Antibody (C-term) (Cat. #AP6402b) in small aliquots to prevent freeze-thaw western blot analysis in K562 cell line lysates cycles. (35ug/lane).This demonstrates the Park2 antibody detected the Park2 protein (arrow). -

Molecular and Physiological Basis for Hair Loss in Near Naked Hairless and Oak Ridge Rhino-Like Mouse Models: Tracking the Role of the Hairless Gene
University of Tennessee, Knoxville TRACE: Tennessee Research and Creative Exchange Doctoral Dissertations Graduate School 5-2006 Molecular and Physiological Basis for Hair Loss in Near Naked Hairless and Oak Ridge Rhino-like Mouse Models: Tracking the Role of the Hairless Gene Yutao Liu University of Tennessee - Knoxville Follow this and additional works at: https://trace.tennessee.edu/utk_graddiss Part of the Life Sciences Commons Recommended Citation Liu, Yutao, "Molecular and Physiological Basis for Hair Loss in Near Naked Hairless and Oak Ridge Rhino- like Mouse Models: Tracking the Role of the Hairless Gene. " PhD diss., University of Tennessee, 2006. https://trace.tennessee.edu/utk_graddiss/1824 This Dissertation is brought to you for free and open access by the Graduate School at TRACE: Tennessee Research and Creative Exchange. It has been accepted for inclusion in Doctoral Dissertations by an authorized administrator of TRACE: Tennessee Research and Creative Exchange. For more information, please contact [email protected]. To the Graduate Council: I am submitting herewith a dissertation written by Yutao Liu entitled "Molecular and Physiological Basis for Hair Loss in Near Naked Hairless and Oak Ridge Rhino-like Mouse Models: Tracking the Role of the Hairless Gene." I have examined the final electronic copy of this dissertation for form and content and recommend that it be accepted in partial fulfillment of the requirements for the degree of Doctor of Philosophy, with a major in Life Sciences. Brynn H. Voy, Major Professor We have read this dissertation and recommend its acceptance: Naima Moustaid-Moussa, Yisong Wang, Rogert Hettich Accepted for the Council: Carolyn R. -

RNA Epigenetics: Fine-Tuning Chromatin Plasticity and Transcriptional Regulation, and the Implications in Human Diseases
G C A T T A C G G C A T genes Review RNA Epigenetics: Fine-Tuning Chromatin Plasticity and Transcriptional Regulation, and the Implications in Human Diseases Amber Willbanks, Shaun Wood and Jason X. Cheng * Department of Pathology, Hematopathology Section, University of Chicago, Chicago, IL 60637, USA; [email protected] (A.W.); [email protected] (S.W.) * Correspondence: [email protected] Abstract: Chromatin structure plays an essential role in eukaryotic gene expression and cell identity. Traditionally, DNA and histone modifications have been the focus of chromatin regulation; however, recent molecular and imaging studies have revealed an intimate connection between RNA epigenetics and chromatin structure. Accumulating evidence suggests that RNA serves as the interplay between chromatin and the transcription and splicing machineries within the cell. Additionally, epigenetic modifications of nascent RNAs fine-tune these interactions to regulate gene expression at the co- and post-transcriptional levels in normal cell development and human diseases. This review will provide an overview of recent advances in the emerging field of RNA epigenetics, specifically the role of RNA modifications and RNA modifying proteins in chromatin remodeling, transcription activation and RNA processing, as well as translational implications in human diseases. Keywords: 5’ cap (5’ cap); 7-methylguanosine (m7G); R-loops; N6-methyladenosine (m6A); RNA editing; A-to-I; C-to-U; 2’-O-methylation (Nm); 5-methylcytosine (m5C); NOL1/NOP2/sun domain Citation: Willbanks, A.; Wood, S.; (NSUN); MYC Cheng, J.X. RNA Epigenetics: Fine-Tuning Chromatin Plasticity and Transcriptional Regulation, and the Implications in Human Diseases. Genes 2021, 12, 627. -

Genome-Wide Association Study of Diabetic Kidney Disease Highlights Biology Involved in Glomerular Basement Membrane Collagen
CLINICAL RESEARCH www.jasn.org Genome-Wide Association Study of Diabetic Kidney Disease Highlights Biology Involved in Glomerular Basement Membrane Collagen Rany M. Salem ,1 Jennifer N. Todd,2,3,4 Niina Sandholm ,5,6,7 Joanne B. Cole ,2,3,4 Wei-Min Chen,8 Darrell Andrews,9 Marcus G. Pezzolesi,10 Paul M. McKeigue,11 Linda T. Hiraki,12 Chengxiang Qiu,13 Viji Nair,14 Chen Di Liao,12 Jing Jing Cao,12 Erkka Valo ,5,6,7 Suna Onengut-Gumuscu,8 Adam M. Smiles,15 Stuart J. McGurnaghan,16 Jani K. Haukka,5,6,7 Valma Harjutsalo,5,6,7,17 Eoin P. Brennan,9 Natalie van Zuydam,18,19 Emma Ahlqvist,20 Ross Doyle,9 Tarunveer S. Ahluwalia ,21 Maria Lajer,21 Maria F. Hughes,9 Jihwan Park,13 Jan Skupien,15 Athina Spiliopoulou,11 Andrew Liu,22 Rajasree Menon,14,23 Carine M. Boustany-Kari,24 Hyun M. Kang,23,25 Robert G. Nelson,26 Ronald Klein,27 Barbara E. Klein,27 Kristine E. Lee ,27 Xiaoyu Gao,28 Michael Mauer,29 Silvia Maestroni,30 Maria Luiza Caramori,29 Ian H. de Boer ,31 Rachel G. Miller,32 Jingchuan Guo ,32 Andrew P. Boright,12 David Tregouet,33,34 Beata Gyorgy,33,34 Janet K. Snell-Bergeon,35 David M. Maahs,36 Shelley B. Bull ,37 Angelo J. Canty,38 Colin N.A. Palmer,39 Lars Stechemesser,40 Bernhard Paulweber,40 Raimund Weitgasser,40,41 Jelizaveta Sokolovska,42 Vita Rovıte,43 Valdis Pırags, 42,44 Edita Prakapiene,45 Lina Radzeviciene,46 Rasa Verkauskiene,46 Nicolae Mircea Panduru,6,47 Leif C. -

Growth and Gene Expression Profile Analyses of Endometrial Cancer Cells Expressing Exogenous PTEN
[CANCER RESEARCH 61, 3741–3749, May 1, 2001] Growth and Gene Expression Profile Analyses of Endometrial Cancer Cells Expressing Exogenous PTEN Mieko Matsushima-Nishiu, Motoko Unoki, Kenji Ono, Tatsuhiko Tsunoda, Takeo Minaguchi, Hiroyuki Kuramoto, Masato Nishida, Toyomi Satoh, Toshihiro Tanaka, and Yusuke Nakamura1 Laboratories of Molecular Medicine [M. M-N., M. U., K. O., T. M., T. Ta., Y. N.] and Genome Database [T. Ts.], Human Genome Center, Institute of Medical Science, The University of Tokyo, Tokyo 108-8639, Japan; Department of Obstetrics and Gynecology, School of Medicine, Kitasato University, Sagamihara 228-8555, Japan [H. K.]; Department of Obstetrics and Gynecology, Institute of Clinical Medicine, University of Tsukuba, Tsukuba 305-8576, Japan [M. N.]; and Department of Obstetrics and Gynecology, Ibaraki Seinan Central Hospital, Tsukuba 306-0433, Japan [T. S.] ABSTRACT Akt/protein kinase B, cell survival, and cell proliferation (8). Over- expression of PTEN can decrease cell proliferation and tumorigenicity The PTEN tumor suppressor gene encodes a multifunctional phospha- (9, 10), an observation attributed to the ability of PTEN to induce cell tase that plays an important role in inhibiting the phosphatidylinositol-3- cycle arrest and apoptosis (11, 12). kinase pathway and downstream functions that include activation of Akt/protein kinase B, cell survival, and cell proliferation. Enforced ex- Thus, lack of PTEN expression may affect a complex set of pression of PTEN in various cancer cell lines decreases cell proliferation transcriptional targets. However, no systematic assessment of PTEN- through arrest of the cell cycle, accompanied in some cases by induction regulated targets in cancer cells has been reported to date. -

Role and Regulation of the P53-Homolog P73 in the Transformation of Normal Human Fibroblasts
Role and regulation of the p53-homolog p73 in the transformation of normal human fibroblasts Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen Julius-Maximilians-Universität Würzburg vorgelegt von Lars Hofmann aus Aschaffenburg Würzburg 2007 Eingereicht am Mitglieder der Promotionskommission: Vorsitzender: Prof. Dr. Dr. Martin J. Müller Gutachter: Prof. Dr. Michael P. Schön Gutachter : Prof. Dr. Georg Krohne Tag des Promotionskolloquiums: Doktorurkunde ausgehändigt am Erklärung Hiermit erkläre ich, dass ich die vorliegende Arbeit selbständig angefertigt und keine anderen als die angegebenen Hilfsmittel und Quellen verwendet habe. Diese Arbeit wurde weder in gleicher noch in ähnlicher Form in einem anderen Prüfungsverfahren vorgelegt. Ich habe früher, außer den mit dem Zulassungsgesuch urkundlichen Graden, keine weiteren akademischen Grade erworben und zu erwerben gesucht. Würzburg, Lars Hofmann Content SUMMARY ................................................................................................................ IV ZUSAMMENFASSUNG ............................................................................................. V 1. INTRODUCTION ................................................................................................. 1 1.1. Molecular basics of cancer .......................................................................................... 1 1.2. Early research on tumorigenesis ................................................................................. 3 1.3. Developing -

Reading Through the Building Blocks of the Genome: Exonic Variation in PD
UNIVERSITY OF CATANIA INTERNATIONAL PhD PROGRAM IN NEUROSCIENCE XXIX Cycle VALENTINA LA COGNATA READING THROUGH THE BUILDING BLOCKS OF THE GENOME: EXONIC VARIATION IN PARKINSON'S DISEASE PhD Thesis Coordinator: Supervisors: Prof. Salvatore Salomone Prof. Velia D’Agata Dr. Sebastiano Cavallaro January 2017 Copyright © V. La Cognata, 2017 All rights reserved. No part of this book may be reproduced, stored in a retrieval system or transmitted in any form or by any means, without prior permission of the author. The copyright of the published papers remains with the publishers. SUMMARY Abstract ...................................................................................................... 5 Chapter 1 .................................................................................................... 7 PARKINSON’S DISEASE ....................................................................................................... 9 HAS EXONIC VARIATION A ROLE IN PD? ............................................................................12 AIMS OF THE PhD WORK ...................................................................................................13 Chapter 2 .................................................................................................. 15 ABSTRACT ........................................................................................................................17 INTRODUCTION ................................................................................................................18 CNVs: A PREVALENT -

Identification of Molecular Targets in Head and Neck Squamous Cell Carcinomas Based on Genome-Wide Gene Expression Profiling
1489-1497 7/11/07 18:41 Page 1489 ONCOLOGY REPORTS 18: 1489-1497, 2007 Identification of molecular targets in head and neck squamous cell carcinomas based on genome-wide gene expression profiling SATOYA SHIMIZU1,2, NAOHIKO SEKI2, TAKASHI SUGIMOTO2, SHIGETOSHI HORIGUCHI1, HIDEKI TANZAWA3, TOYOYUKI HANAZAWA1 and YOSHITAKA OKAMOTO1 Departments of 1Otorhinolaryngology, 2Functional Genomics and 3Clinical Molecular Biology, Graduate School of Medicine, Chiba University, 1-8-1 Inohana, Chuo-ku, Chiba 260-8670, Japan Received May 21, 2007; Accepted June 28, 2007 Abstract. DNA amplifications activate oncogenes and are patients and metastases develop in 15-25% of patients (1). hallmarks of nearly all advanced cancers including head and Many factors, such as TNM stage, pathological grade and neck squamous cell carcinoma (HNSCC). Some oncogenes tumor site, influence the prognosis of HNSCC but are not show both DNA copy number gain and mRNA overexpression. sufficient to predict outcome. In addition, treatment often Chromosomal comparative genomic hybridization and oligo- results in impairment of functions such as speech and nucleotide microarrays were used to examine 8 HNSCC cell swallowing, cosmetic disfiguration and mental pain. These lines and a plot of gene expression levels relative to their inflictions significantly erode quality of life. To overcome this position on the chromosome was produced. Three highly situation, there is a need to find novel biomarkers that classify up-regulated genes, NT5C3, ANLN and INHBA, were patients into prognostic groups, to aid identification of high- identified on chromosome 7p14. These genes were subjected risk patients who may benefit from different treatments. to quantitative real-time RT-PCR on cDNA and genomic Comparative genomic hybridization (CGH) has facilitated DNA derived from 8 HNSCC cell lines. -

Subgroup-Specific Structural Variation Across 1,000 Medulloblastoma Genomes
ARTICLE doi:10.1038/nature11327 Subgroup-specific structural variation across 1,000 medulloblastoma genomes A list of authors and their affiliations appears at the end of the paper Medulloblastoma, the most common malignant paediatric brain tumour, is currently treated with nonspecific cytotoxic therapies including surgery, whole-brain radiation, and aggressive chemotherapy. As medulloblastoma exhibits marked intertumoural heterogeneity, with at least four distinct molecular variants, previous attempts to identify targets for therapy have been underpowered because of small samples sizes. Here we report somatic copy number aberrations (SCNAs) in 1,087 unique medulloblastomas. SCNAs are common in medulloblastoma, and are predominantly subgroup-enriched. The most common region of focal copy number gain is a tandem duplication of SNCAIP, a gene associated with Parkinson’s disease, which is exquisitely restricted to Group 4a. Recurrent translocations of PVT1, including PVT1-MYC and PVT1-NDRG1, that arise through chromothripsis are restricted to Group 3. Numerous targetable SCNAs, including recurrent events targeting TGF-b signalling in Group 3, and NF-kB signalling in Group 4, suggest future avenues for rational, targeted therapy. Brain tumours are the most common cause of childhood oncological Twenty-eight regions of recurrent high-level amplification (copy death, and medulloblastoma is the most common malignant paediatric number $ 5) were identified (Fig. 1d and Supplementary Table 7). brain tumour. Current medulloblastoma therapy including surgical The most prevalent amplifications affected members of the MYC resection, whole-brain and spinal cord radiation, and aggressive family with MYCN predominantly amplified in SHH and Group 4, chemotherapy supplemented by bone marrow transplant yields five- MYC in Group 3, and MYCL1 in SHH medulloblastomas. -
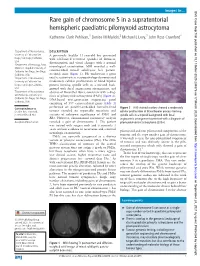
Rare Gain of Chromosome 5 in a Supratentorial Hemispheric
Images in… BMJ Case Rep: first published as 10.1136/bcr-2020-234878 on 17 March 2020. Downloaded from Rare gain of chromosome 5 in a supratentorial hemispheric paediatric pilomyxoid astrocytoma Katherine Clark Pehlivan,1 Denise M Malicki,2 Michael L Levy,3 John Ross Crawford4 1Department of Neurosciences, DESCRIPTION University of California San A previously healthy 11- year- old boy presented Diego, San Diego, California, with self- limited recurrent episodes of dizziness, USA disorientation and visual changes with a normal 2Department of Pathology, Rady neurological examination. MRI revealed a well- Children’s Hospital University of California San Diego, San Diego, circumscribed mixed solid/cystic left parieto- California, USA occipital mass (figure 1). He underwent a gross 3Department of Neurosurgery, total resection where neuropathology demonstrated University of California San moderately cellular proliferation of bland bipolar Diego, San Diego, California, process- forming spindle cells in a myxoid back- USA ground with focal angiocentric arrangement, and 4 Department of Neurosciences absence of Rosenthal fibres, consistent with a diag- and Pediatrics, University of nosis of pilomyxoid astrocytoma (PMA) (figure 2). California San Diego, San Diego, DNA- based next- generation sequencing panel California, USA consisting of 397 cancer- related genes (table 1) performed on paraffin- embedded formalin- fixed Correspondence to Figure 2 H&E- stained sections showed a moderately Dr John Ross Crawford; tumour revealed no reportable mutations and cellular proliferation of bland bipolar process- forming jrcrawford@ ucsd. edu variants of unknown significance of PMS1 and spindle cells in a myxoid background with focal RB1. However, chromosomal microarray analysis angiocentric arrangement consistent with a diagnosis of Accepted 4 March 2020 revealed a gain of chromosome 5. -

Short- and Long-Term Impact of Hyperoxia on the Blood and Retinal Cells’ Transcriptome in a Mouse Model of Oxygen- Induced Retinopathy
www.nature.com/pr BASIC SCIENCE ARTICLE OPEN Short- and long-term impact of hyperoxia on the blood and retinal cells’ transcriptome in a mouse model of oxygen- induced retinopathy Magdalena Zasada1, Anna Madetko-Talowska2, Cecilie Revhaug3,4, Anne Gro W. Rognlien3,4, Lars O. Baumbusch3,Teofila Książek2, Katarzyna Szewczyk2, Agnieszka Grabowska2, Miroslaw Bik-Multanowski2, Jacek Józef Pietrzyk1, Przemko Kwinta1 and Ola Didrik Saugstad3,4 BACKGROUND: We aimed to identify global blood and retinal gene expression patterns in murine oxygen-induced retinopathy (OIR), a common model of retinopathy of prematurity, which may allow better understanding of the pathogenesis of this severe ocular prematurity complication and identification of potential blood biomarkers. METHODS: A total of 120 C57BL/6J mice were randomly divided into an OIR group, in which 7-day-old pups were maintained in 75% oxygen for 5 days, or a control group. RNA was extracted from the whole-blood mononuclear cells and retinal cells on days 12, 17, and 28. Gene expression in the RNA samples was evaluated with mouse gene expression microarrays. RESULTS: There were 38, 1370 and 111 genes, the expression of which differed between the OIR and control retinas on days 12, 17, and 28, respectively. Gene expression in the blood mononuclear cells was significantly altered only on day 17. Deptor and Nol4 genes showed reduced expression both in the blood and retinal cells on day 17. 1234567890();,: CONCLUSION: There are sustained marked changes in the global pattern of gene expression in the OIR mice retinas. An altered expression of Deptor and Nol4 genes in the blood mononuclear cells requires further investigation as they may indicate retinal neovascularization. -

Identification of Significant Genes with Poor Prognosis in Ovarian Cancer Via Bioinformatical Analysis
Feng et al. Journal of Ovarian Research (2019) 12:35 https://doi.org/10.1186/s13048-019-0508-2 RESEARCH Open Access Identification of significant genes with poor prognosis in ovarian cancer via bioinformatical analysis Hao Feng1†, Zhong-Yi Gu2†, Qin Li2, Qiong-Hua Liu3, Xiao-Yu Yang4* and Jun-Jie Zhang2* Abstract Ovarian cancer (OC) is the highest frequent malignant gynecologic tumor with very complicated pathogenesis. The purpose of the present academic work was to identify significant genes with poor outcome and their underlying mechanisms. Gene expression profiles of GSE36668, GSE14407 and GSE18520 were available from GEO database. There are 69 OC tissues and 26 normal tissues in the three profile datasets. Differentially expressed genes (DEGs) between OC tissues and normal ovarian (OV) tissues were picked out by GEO2R tool and Venn diagram software. Next, we made use of the Database for Annotation, Visualization and Integrated Discovery (DAVID) to analyze Kyoto Encyclopedia of Gene and Genome (KEGG) pathway and gene ontology (GO). Then protein-protein interaction (PPI) of these DEGs was visualized by Cytoscape with Search Tool for the Retrieval of Interacting Genes (STRING). There were total of 216 consistently expressed genes in the three datasets, including 110 up-regulated genes enriched in cell division, sister chromatid cohesion, mitotic nuclear division, regulation of cell cycle, protein localization to kinetochore, cell proliferation and Cell cycle, progesterone-mediated oocyte maturation and p53 signaling pathway, while 106 down-regulated genes enriched in palate development, blood coagulation, positive regulation of transcription from RNA polymerase II promoter, axonogenesis, receptor internalization, negative regulation of transcription from RNA polymerase II promoter and no significant signaling pathways.