GRAS Notice 000721, Calcium Acetate
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

How to Take Your Phosphate Binders
How to take your phosphate binders Information for renal patients Oxford Kidney Unit Page 2 What are phosphate binders? To reduce the amount of phosphate you absorb from your food you may have been prescribed a medicine called a phosphate binder. Phosphate binders work by binding (attaching) to some of the phosphate in food. This will reduce the amount of phosphate being absorbed into your blood stream. A list of phosphate binders and how to take them is shown below. Phosphate binder How to take it Calcichew (calcium carbonate) Chew thoroughly 10-15 minutes before or immediately before food Renacet (calcium acetate) Phosex (calcium acetate) Osvaren (calcium acetate and magnesium carbonate) Swallow whole after the first Renagel 2-3 mouthfuls of food (sevelemer hydrochloride) Renvela tablets (sevelemer carbonate) Alucaps (aluminium hydroxide) Renvela powder Dissolve in 60ml of water and (sevelemer carbonate) take after the first 2-3 mouthfuls of food Fosrenol tablets Chew thoroughly towards the (lanthanum carbonate) end/immediately after each meal Fosrenol powder Mix with a small amount of (lanthanum carbonate) food and eat immediately Velphoro Chew thoroughly after the first (sucroferric oxyhydroxide) 2-3 mouthfuls The phosphate binder you have been prescribed is: ……………………………………………………………………………………………………………………………………………………….. Page 3 How many phosphate binders should I take? You should follow the dose that has been prescribed for you. Your renal dietitian can advise how best to match your phosphate binders to your meal pattern, as well as which snacks require a phosphate binder. What happens if I forget to take my phosphate binder? For best results, phosphate binders should be taken as instructed. -

Safety Data Sheet
SAFETY DATA SHEET Revision date 06-May-2020 Revision Number 1 1. Identification Product identifier Product Name CALCIUM IODATE, REAGENT Other means of identification Product Code(s) C1130 UN/ID no UN1479 Synonyms None Recommended use of the chemical and restrictions on use Recommended use No information available Restrictions on use No information available Details of the supplier of the safety data sheet Supplier Address Spectrum Chemical Mfg. Corp. 14422 South San Pedro St. Gardena, CA 90248 (310) 516-8000 Emergency telephone number Emergency Telephone Chemtrec 1-800-424-9300 2. Hazard(s) identification Classification Skin corrosion/irritation Category 2 Serious eye damage/eye irritation Category 2A Specific target organ toxicity (single exposure) Category 3 Oxidizing solids Category 2 Hazards not otherwise classified (HNOC) Not applicable Label elements Danger Hazard statements Causes skin irritation Causes serious eye irritation May cause respiratory irritation May intensify fire; oxidizer Appearance Crystalline powder Physical state Solid Odor Odorless Precautionary Statements - Prevention Wash face, hands and any exposed skin thoroughly after handling Avoid breathing dust/fume/gas/mist/vapors/spray Use only outdoors or in a well-ventilated area Keep away from heat Keep/Store away from clothing/ combustible materials Take any precaution to avoid mixing with combustibles Wear protective gloves/eye protection/face protection Precautionary Statements - Response IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing If eye irritation persists: Get medical advice/attention IF ON SKIN: Wash with plenty of water and soap If skin irritation occurs: Get medical advice/attention Take off contaminated clothing and wash it before reuse IF INHALED: Remove person to fresh air and keep comfortable for breathing Call a POISON CENTER or doctor if you feel unwell In case of fire: Use CO2, dry chemical, or foam to extinguish Precautionary Statements - Storage Store in a well-ventilated place. -

Calcium Acetate Capsules
Calcium Acetate Capsules Type of Posting Revision Bulletin Posting Date 27–Dec–2019 Official Date 01–Jan–2020 Expert Committee Chemical Medicines Monographs 6 Reason for Revision Compliance In accordance with the Rules and Procedures of the 2015–2020 Council of Experts, the Chemical Medicines Monographs 6 Expert Committee has revised the Calcium Acetate Capsules monograph. The purpose for the revision is to add Dissolution Test 4 to accommodate FDA-approved drug products with different dissolution conditions and/or tolerances than the existing dissolution tests. • Dissolution Test 4 was validated using a YMC-Pack ODS-A C18 brand of L1 column. The typical retention time for calcium acetate is about 4.3 min. The Calcium Acetate Capsules Revision Bulletin supersedes the currently official monograph. Should you have any questions, please contact Michael Chang, Senior Scientific Liaison (301-230-3217 or [email protected]). C236679-M11403-CHM62015, rev. 00 20191227 Revision Bulletin Calcium 1 Official January 1, 2020 Calcium Acetate Capsules PERFORMANCE TESTS DEFINITION Change to read: Calcium Acetate Capsules contain NLT 90.0% and NMT · DISSOLUTION á711ñ 110.0% of the labeled amount of calcium acetate Test 1 (C4H6CaO4). Medium: Water; 900 mL IDENTIFICATION Apparatus 2: 50 rpm, with sinkers · A. The retention time of the calcium peak of the Sample Time: 10 min solution corresponds to that of the Standard solution, as Mobile phase, Standard solution, Chromatographic obtained in the Assay. system, and System suitability: Proceed as directed in · B. IDENTIFICATION TESTSÐGENERAL á191ñ, Chemical the Assay. Identification Tests, Acetate Sample solution: Pass a portion of the solution under test Sample solution: 67 mg/mL of calcium acetate from through a suitable filter of 0.45-µm pore size. -

Production and Testing of Calcium Magnesium Acetate in Maine
77 Majesty's Stationery Office, London, England, River. Res. Note FPL-0229. Forest Service, U.S. 1948. Department of Agriculture, Madison, Wis., 1974. 14. M.S. Aggour and A. Ragab. Safety and Soundness 20. W.L. James. Effect of Temperature and Moisture of Submerged Timber Bridge PU.ing. FHWA/MD In Content on Internal Friction and Speed of Sound terim Report AW082-231-046. FHWA, U.S. Depart in Douglas Fir. Forest Product Journal, Vol. ment of Transportation, June 1982. 11, No. 9, 1961, pp. 383-390, 15. B.O. Orogbemi. Equipment for Determining the 21. A, Burmester. Relationship Between Sound Veloc Dynami c Modulus of Submerged Bridge Timber Pil ity and Morphological, Physical, and Mechani ·ing. Master's thesis. University of Maryland, cal Properties of Wood. Holz als Roh und Wer College Park, 1980. stoff, Vol. 23, No. 6, 1965, pp. 227-236 (in 16. T.L. Wilkinson. Strength Evaluation of Round German) • Timber Piles. Res. Note FPL-101. Forest Ser 22. c.c. Gerhards. Stress Wave Speed and MOE of vice, U.S. Department of Agriculture, Madison, Weetgum Ranging from 150 to 15 Percent MC. Wis., 1968. Forest Product Journal, Vol. 25, No. 4, 1975, 17. J, Bodig and B.A. Jayne. Mechanics of Wood and pp. 51-57. Wood Composites. Van Nostrand, New York, 1982. 18. R.M. Armstrong. Structural Properties of Timber Piles, Behavior of Deep Foundations. Report STP-670, ASTM, Philadelphia, 1979, pp. 118-152. 19. B.A, Bendtsen. Bending Strength and Stiffness Publication of this paper sponsored by Committee on of Bridge Piles After 85 Years in the Milwaukee Structures Maintenance. -

Phosphate Binders
Pharmacy Info Sheet Phosphate Binders calcium acetate, calcium carbonate (Tums, Calsan, Apocal, Ocal), calcium liquid, aluminum hydroxide (Basaljel, Amphojel), sevelamer (Renagel), lanthanum (Fosrenol) What it does: Phosphate binders are used to treat high Special considerations for lanthamum and blood phosphorus levels. sevelamer: Calcium acetate, calcium carbonate, calcium Lanthanum should be taken during or liquid, aluminum hydroxide, lanthanum and immediately after a meal. Taking a dose on an sevelamer bind dietary phosphate. When the empty stomach can cause nausea and kidneys fail, phosphorus builds up in the body vomiting. Chew the tablet completely before because the kidneys can no longer remove swallowing. DO NOT swallow tablets whole. much phosphorus. Phosphate binders are used to lower the amount of phosphorus Sevelamer should be taken just before eating. absorbed from food to limit development of Swallow the tablet whole – Renagel should not bone and blood vessel disease. be cut or chewed. The contents of sevelamer tablets expand in water and could cause Aluminum hydroxide and calcium carbonate choking if cut chewed or crushed. may also be prescribed as antacids. Calcium preparations may also be prescribed as Phosphate binders may interfere with the calcium supplements. Use them only as absorption of certain drugs such as iron prescribed. When these medications are supplements, antibiotics, digoxin, ranitidine, prescribed as calcium supplements or antiseizure, and antiarrhythmic medications. antacids, take between meals. If you are prescribed any of these drugs, take them at least 1 hour before or 3 hours after your How it works: phosphate binder. Kidney disease can cause phosphate to accumulate which results in bone and blood What to do if you miss a dose: vessel disease. -

Solubility Product Constant (Ksp) for a Salt of Limited Solubility
CHM130 Solubility Product Experiment Experiment: Solubility Product Constant (Ksp) for a Salt of Limited Solubility Introduction: The equilibrium process in this experiment is a saturated aqueous solution of calcium iodate, Ca(IO3)2. The relevant solubility equation and solubility product expression, are both shown below. - 2+ 2+ 2 Ca(IO3)2(s) <-===== > Ca (aq) + 2IO3 (aq) Ksp = [Ca ] [IO3 ] For a saturated solution of calcium iodate, if you can determine either the molar concentration of calcium ion, or the molar concentration iodate ion, the solubility product constant can be found using the reverse of the process shown above. There was found the silver ion concentration, in a saturated aqueous solution, from a known value for Ksp. In other words, if the calcium ion concentration in today's experiment was found to be 0.1 M, you could immediately say the concentration of iodate ion must be half that value, or 0.05 M, according to the stoichiometry of the solubility equation given above. The solubility product constant could then be found with simple arithmetic. In this experiment, the iodate ion concentration of a saturated calcium iodate solution will be found via a redox titration with sodium thiosulfate, Na2S2O3. The concentration of iodate ion (IO3-) will be determined by titration with a standardized sodium thiosulfate (Na2S2O3) solution in the presence of potassium iodide (KI). Starch will be used as an indicator, and a sharp blue-to-clear transition will mark the equivalence point. The relevant reaction equations are summarized as follows. IO3 (aq) + 5I (aq) + 6H3O (aq) --------> 3I2(aq) + 9H2O(l) This step, which occurs after adding both solid KI, and aqueous acid, to aliquots of saturated iodate solutions, has the net effect of converting iodate ions to aqueous iodine. -
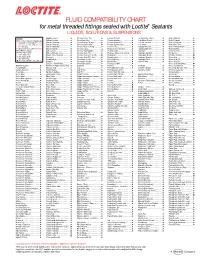
FLUID COMPATIBILITY CHART for Metal Threaded Fittings Sealed with Loctite¨ Sealants LIQUIDS, SOLUTIONS & SUSPENSIONS
FLUID COMPATIBILITY CHART for metal threaded fittings sealed with Loctite® Sealants LIQUIDS, SOLUTIONS & SUSPENSIONS LEGEND: Bagasse Fibers.......................... Chlorobenzene Dry ................... Ferrous Chloride ...................... Ion Exclusion Glycol ................. Nickel Chloride.......................... All Loctite® Anaerobic Sealants are Barium Acetate ........................ Chloroform Dry......................... Ferrous Oxalate......................... Irish Moss Slurry...................... Nickel Cyanide ......................... Compatible Including #242®, 243, Barium Carbonate..................... Chloroformate Methyl............... Ferrous Sulfate10%.................. Iron Ore Taconite ..................... Nickel Fluoborate ..................... 542, 545, 565, 567, 569, 571, 572, Barium Chloride........................ Chlorosulfonic Acid .................. Ferrous Sulfate (Sat)................. Iron Oxide ................................ Nickel Ore Fines ....................... 577, 580, 592 Barium Hydroxide..................... Chrome Acid Cleaning .............. Fertilizer Sol ............................. Isobutyl Alcohol ....................... Nickel Plating Bright ................. † Use Loctite® #270, 271™, 277, 554 Barium Sulfate.......................... Chrome Liquor.......................... Flotation Concentrates.............. Isobutyraldehyde ..................... Nickel Sulfate ........................... Not Recommended Battery Acid .............................. Chrome Plating -

Production of Low-Cost Acetate Deicers from Biomass and Industrial Wastes
Production of Low-Cost Acetate Deicers from Biomass and Industrial Wastes Shang-Tian Yang and Zuwei Jin, The Ohio State University Brian H. ChoUar, Federal Highway Administration Calcium magnesium acetate (CMA), a mixture of calcium 1 rom 10 million to 14 million tons of road salt are acetate and magnesium acetate, is used as an environmen• I used annually in the United States and Canada. tally benign roadway deicer. The present commercial F Salt is an extremely effective snow and ice con• CMA deicer made from glacial acetic acid and dolomitic trol agent and is relatively inexpensive. However, a lime or limestone is more expensive than salt and other study in New York State showed that although 1 ton of deicers. Also, a liquid potassium acetate deicer is used to road salt cost only $25, it caused more than $1,400 in replace urea and glycol in airport runway deicing. Two al• damage (1). Salt is corrosive to concrete and metals ternative low-cost methods to produce these acetate de- used in the nation's infrastructure, is harmful to road• icers from cheap feedstocks, such as biomass and side vegetation, and poses serious threats to environ• industrial wastes, were studied. CMA deicers produced ment and ground-water quality in some regions (2). from cheese whey by fermentation and extraction were FHWA spends about $12.5 billion annually, a sub• tested for their acetate content and deicing property. The stantial portion of which is used to rebuild and resur• CMA solid sample obtained from extraction of the acetic face highways and bridges damaged by salt corrosion. -

Consideration of Mandatory Fortification with Iodine for Australia and New Zealand Food Technology Report
CONSIDERATION OF MANDATORY FORTIFICATION WITH IODINE FOR AUSTRALIA AND NEW ZEALAND FOOD TECHNOLOGY REPORT December 2007 1 Introduction Food Standards Australia New Zealand is considering mandatory fortification of the food supply in Australia and New Zealand with iodine. Generally, the addition of iodine to foods is technologically feasible. However, in some instances the addition of iodine can lead to quality changes in food products such as appearance, taste, odour, texture and shelf life. These changes will depend on the chemical form of iodine used as a fortificant, the chemistry of the food that is being fortified, the food processes involved in manufacture and possible processing interactions that could occur during distribution and storage. Many foods have been fortified with iodine and the potassium salts of iodine compounds have been used as the preferred form. 2 Forms of Iodine Iodine is normally introduced, or supplemented, as the iodide or iodate of potassium, calcium or sodium. The following table lists different chemical forms of iodine along with their important physical properties. Table 1: Physical Properties of Iodine and its Compounds Name Chemical Formula % Iodine Solubility in water (g/L) 0°C 20°C 30°C 40°C 60°C Iodine I2 100 - - 0.3 0.4 0.6 Calcium iodide CaI2 86.5 646 676 690 708 740 Calcium iodate Ca(IO3)2.6H2O 65.0 - 1.0 4.2 6.1 13.6 Potassium iodide KI 76.5 1280 1440 1520 1600 1760 Potassium iodate KIO3 59.5 47.3 81.3 117 128 185 Sodium iodide NaI.2H20 85.0 1590 1790 1900 2050 2570 Sodium iodate NaIO3 64.0 - 25.0 90.0 150 210 Adapted from Mannar and Dunn (1995) 2.1 Potassium Iodide Potassium iodide (KI) is highly soluble in water. -

Calcium Chloride
Iodine Livestock 1 2 Identification of Petitioned Substance 3 4 Chemical Names: 7553-56-2 (Iodine) 5 Iodine 11096-42-7 (Nonylphenoxypolyethoxyethanol– 6 iodine complex) 7 Other Name: 8 Iodophor Other Codes: 9 231-442-4 (EINECS, Iodine) 10 Trade Names: CAS Numbers: 11 FS-102 Sanitizer & Udderwash 12 Udder-San Sanitizer and Udderwash 13 14 Summary of Petitioned Use 15 The National Organic Program (NOP) final rule currently allows the use of iodine in organic livestock 16 production under 7 CFR §205.603(a)(14) as a disinfectant, sanitizer and medical treatment, as well as 7 CFR 17 §205.603(b)(3) for use as a topical treatment (i.e., teat cleanser for milk producing animals). In this report, 18 updated and targeted technical information is compiled to augment the 1994 Technical Advisory Panel 19 (TAP) Report on iodine in support of the National Organic Standard’s Board’s sunset review of iodine teat 20 dips in organic livestock production. 21 Characterization of Petitioned Substance 22 23 Composition of the Substance: 24 A variety of substances containing iodine are used for antisepsis and disinfection. The observed activity of 25 these commercial disinfectants is based on the antimicrobial properties of molecular iodine (I2), which 26 consists of two covalently bonded atoms of elemental iodine (I). For industrial uses, I2 is commonly mixed 27 with surface-active agents (surfactants) to enhance the water solubility of I2 and also to sequester the 28 available I2 for extended release in disinfectant products. Generally referred to as iodophors, these 29 “complexes” consist of up to 20% I2 by weight in loose combination with nonionic surfactants such as 30 nonylphenol polyethylene glycol ether (Lauterbach & Uber, 2011). -

BLUE BOOK 1 Methyl Acetate CIR EXPERT PANEL MEETING
BLUE BOOK 1 Methyl Acetate CIR EXPERT PANEL MEETING AUGUST 30-31, 2010 Memorandum To: CIR Expert Panel Members and Liaisons From: Bart Heldreth Ph.D., Chemist Date: July 30, 2010 Subject: Draft Final Report of Methyl Acetate, Simple Alkyl Acetate Esters, Acetic Acid and its Salts as used in Cosmetics . This review includes Methyl Acetate and the following acetate esters, relevant metabolites and acetate salts: Propyl Acetate, Isopropyl Acetate, t-Butyl Acetate, Isobutyl Acetate, Butoxyethyl Acetate, Nonyl Acetate, Myristyl Acetate, Cetyl Acetate, Stearyl Acetate, Isostearyl Acetate, Acetic Acid, Sodium Acetate, Potassium Acetate, Magnesium Acetate, Calcium Acetate, Zinc Acetate, Propyl Alcohol, and Isopropyl Alcohol. At the June 2010 meeting, the Panel reviewed information submitted in response to an insufficient data announcement for HRIPT data for Cetyl Acetate at the highest concentration of use (lipstick). On reviewing the data in the report, evaluating the newly available unpublished studies and assessing the newly added ingredients, the Panel determined that the data are now sufficient, and issued a Tentative Report, with a safe as used conclusion. Included in this report are Research Institute for Fragrance Materials (RIFM) sponsored toxicity studies on Methyl Acetate and Propyl Acetate, which were provided in “wave 2” at the June Panel Meeting but are now incorporated in full. The Tentative Report was issued for a 60 day comment period (60 days as of the August panel meeting start date). The Panel should now review the Draft Final Report, confirm the conclusion of safe, and issue a Final Report. All of the materials are in the Panel book as well as in the URL for this meeting's web page http://www.cir- safety.org/aug10.shtml. -

Operation, and This Can Most Easily Be Done If the Solution STITUTE
DR. W. MACKIE : CALCIUM IODATE AS AN IODOFORM SUBSTITUTE. 1867 from 101° to 102° F- It will cool down to blood heat while abundance. Chemical equation shows that calcium iodate in passing through the transfusion apparatus. Care should be contact with hydrochloric acid liberates as nearly as may be taken to see that the temperature is maintained during the four-sevenths of its weight of chlorine. On this fact theoreti- operation, and this can most easily be done if the solution cally depends its use as a gastro-intestinal antiseptic. As be prepared in small quantities at a time. It should in all regards antiseptic action something no doubt depends on the cases be filtered through muslin before use. If it be neces- chemical instability of iodates generally and of this iodate in sary to repeat the transfusion another vein must be opened. particular, on the nascent action of the liberated products, When the patient has been revived and the immediate and in this case on the fact that the base forms insoluble danger of death from syncope is staved oil the extreme rest- compounds such as carbonate and phosphate in contact with lessness and headache that so frequently follow may be alkaline discharges, thus probably facilitating the free treated by large doses of opium or morphia. Any further operation of the acid radicle of the salt, on which of course tendency to syncope is best treated by raising the foot of its particular virtue really depends. the bed and the administration of liquid food and stimulants From this view of its action it is to be inferred that in small quantities at frequent intervals.