La2u2se9: an Ordered Lanthanide/Actinide Chalcogenide with a Novel Structure Type
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Additional Teacher Background Chapter 4 Lesson 3, P. 295
Additional Teacher Background Chapter 4 Lesson 3, p. 295 What determines the shape of the standard periodic table? One common question about the periodic table is why it has its distinctive shape. There are actu- ally many different ways to represent the periodic table including circular, spiral, and 3-D. But in most cases, it is shown as a basically horizontal chart with the elements making up a certain num- ber of rows and columns. In this view, the table is not a symmetrical rectangular chart but seems to have steps or pieces missing. The key to understanding the shape of the periodic table is to recognize that the characteristics of the atoms themselves and their relationships to one another determine the shape and patterns of the table. ©2011 American Chemical Society Middle School Chemistry Unit 303 A helpful starting point for explaining the shape of the periodic table is to look closely at the structure of the atoms themselves. You can see some important characteristics of atoms by look- ing at the chart of energy level diagrams. Remember that an energy level is a region around an atom’s nucleus that can hold a certain number of electrons. The chart shows the number of energy levels for each element as concentric shaded rings. It also shows the number of protons (atomic number) for each element under the element’s name. The electrons, which equal the number of protons, are shown as dots within the energy levels. The relationship between atomic number, energy levels, and the way electrons fill these levels determines the shape of the standard periodic table. -

Actinide Ground-State Properties-Theoretical Predictions
Actinide Ground-State Properties Theoretical predictions John M. Wills and Olle Eriksson electron-electron correlations—the electronic energy of the ground state of or nearly fifty years, the actinides interactions among the 5f electrons and solids, molecules, and atoms as a func- defied the efforts of solid-state between them and other electrons—are tional of electron density. The DFT Ftheorists to understand their expected to affect the bonding. prescription has had such a profound properties. These metals are among Low-symmetry crystal structures, impact on basic research in both the most complex of the long-lived relativistic effects, and electron- chemistry and solid-state physics that elements, and in the solid state, they electron correlations are very difficult Walter Kohn, its main inventor, was display some of the most unusual to treat in traditional electronic- one of the recipients of the 1998 behaviors of any series in the periodic structure calculations of metals and, Nobel Prize in Chemistry. table. Very low melting temperatures, until the last decade, were outside the In general, it is not possible to apply large anisotropic thermal-expansion realm of computational ability. And DFT without some approximation. coefficients, very low symmetry crystal yet, it is essential to treat these effects But many man-years of intense research structures, many solid-to-solid phase properly in order to understand the have yielded reliable approximate transitions—the list is daunting. Where physics of the actinides. Electron- expressions for the total energy in does one begin to put together an electron correlations are important in which all terms, except for a single- understanding of these elements? determining the degree to which 5f particle kinetic-energy term, can be In the last 10 years, together with electrons are localized at lattice sites. -
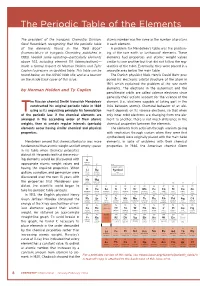
The Periodic Table of the Elements
The Periodic Table of the Elements The president of the Inorganic Chemistry Division, atomic number was the same as the number of protons Gerd Rosenblatt, recognizing that the periodic table in each element. of the elements found in the “Red Book” A problem for Mendeleev’s table was the position- (Nomenclature of Inorganic Chemistry, published in ing of the rare earth or lanthanoid* elements. These 1985) needed some updating—particularly elements elements had properties and atomic weight values above 103, including element 110 (darmstadtium)— similar to one another but that did not follow the reg- made a formal request to Norman Holden and Tyler ularities of the table. Eventually, they were placed in a Coplen to prepare an updated table. This table can be separate area below the main table. found below, on the IUPAC Web site, and as a tear-off The Danish physicist Niels Henrik David Bohr pro- on the inside back cover of this issue. posed his electronic orbital structure of the atom in 1921, which explained the problem of the rare earth by Norman Holden and Ty Coplen elements. The electrons in the outermost and the penultimate orbits are called valence electrons since generally their actions account for the valence of the he Russian chemist Dmitri Ivanovich Mendeleev element (i.e., electrons capable of taking part in the constructed his original periodic table in 1869 links between atoms). Chemical behavior of an ele- Tusing as its organizing principle his formulation ment depends on its valence electrons, so that when of the periodic law: if the chemical elements are only inner orbit electrons are changing from one ele- arranged in the ascending order of their atomic ment to another, there is not much difference in the weights, then at certain regular intervals (periods) chemical properties between the elements. -

ELECTRONEGATIVITY D Qkq F=
ELECTRONEGATIVITY The electronegativity of an atom is the attracting power that the nucleus has for it’s own outer electrons and those of it’s neighbours i.e. how badly it wants electrons. An atom’s electronegativity is determined by Coulomb’s Law, which states, “ the size of the force is proportional to the size of the charges and inversely proportional to the square of the distance between them”. In symbols it is represented as: kq q F = 1 2 d 2 where: F = force (N) k = constant (dependent on the medium through which the force is acting) e.g. air q1 = charge on an electron (C) q2 = core charge (C) = no. of protons an outer electron sees = no. of protons – no. of inner shell electrons = main Group Number d = distance of electron from the nucleus (m) Examples of how to calculate the core charge: Sodium – Atomic number 11 Electron configuration 2.8.1 Core charge = 11 (no. of p+s) – 10 (no. of inner e-s) +1 (Group i) Chlorine – Atomic number 17 Electron configuration 2.8.7 Core charge = 17 (no. of p+s) – 10 (no. of inner e-s) +7 (Group vii) J:\Sciclunm\Resources\Year 11 Chemistry\Semester1\Notes\Atoms & The Periodic Table\Electronegativity.doc Neon holds onto its own electrons with a core charge of +8, but it can’t hold any more electrons in that shell. If it was to form bonds, the electron must go into the next shell where the core charge is zero. Group viii elements do not form any compounds under normal conditions and are therefore given no electronegativity values. -

Periodic Table of the Elements Notes
Periodic Table of the Elements Notes Arrangement of the known elements based on atomic number and chemical and physical properties. Divided into three basic categories: Metals (left side of the table) Nonmetals (right side of the table) Metalloids (touching the zig zag line) Basic Organization by: Atomic structure Atomic number Chemical and Physical Properties Uses of the Periodic Table Useful in predicting: chemical behavior of the elements trends properties of the elements Atomic Structure Review: Atoms are made of protons, electrons, and neutrons. Elements are atoms of only one type. Elements are identified by the atomic number (# of protons in nucleus). Energy Levels Review: Electrons are arranged in a region around the nucleus called an electron cloud. Energy levels are located within the cloud. At least 1 energy level and as many as 7 energy levels exist in atoms Energy Levels & Valence Electrons Energy levels hold a specific amount of electrons: 1st level = up to 2 2nd level = up to 8 3rd level = up to 8 (first 18 elements only) The electrons in the outermost level are called valence electrons. Determine reactivity - how elements will react with others to form compounds Outermost level does not usually fill completely with electrons Using the Table to Identify Valence Electrons Elements are grouped into vertical columns because they have similar properties. These are called groups or families. Groups are numbered 1-18. Group numbers can help you determine the number of valence electrons: Group 1 has 1 valence electron. Group 2 has 2 valence electrons. Groups 3–12 are transition metals and have 1 or 2 valence electrons. -

Periodic Table 1 Periodic Table
Periodic table 1 Periodic table This article is about the table used in chemistry. For other uses, see Periodic table (disambiguation). The periodic table is a tabular arrangement of the chemical elements, organized on the basis of their atomic numbers (numbers of protons in the nucleus), electron configurations , and recurring chemical properties. Elements are presented in order of increasing atomic number, which is typically listed with the chemical symbol in each box. The standard form of the table consists of a grid of elements laid out in 18 columns and 7 Standard 18-column form of the periodic table. For the color legend, see section Layout, rows, with a double row of elements under the larger table. below that. The table can also be deconstructed into four rectangular blocks: the s-block to the left, the p-block to the right, the d-block in the middle, and the f-block below that. The rows of the table are called periods; the columns are called groups, with some of these having names such as halogens or noble gases. Since, by definition, a periodic table incorporates recurring trends, any such table can be used to derive relationships between the properties of the elements and predict the properties of new, yet to be discovered or synthesized, elements. As a result, a periodic table—whether in the standard form or some other variant—provides a useful framework for analyzing chemical behavior, and such tables are widely used in chemistry and other sciences. Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. -

• What I Hope to Convey to You Is That Rare Earths Are Uniquely Required for High Performance Magnets
• What I hope to convey to you is that rare earths are uniquely required for high performance magnets. • First a quick introduction to Arnold – the company I’ve worked for since 1992. • Arnold’s history in magnetics and magnetic materials extends back to 1895 and has included almost every commercially supplied permanent and soft magnetic product. • Today Arnold is focused on: SmCo, Alnico and bonded permanent magnets; precision thin metals – both magnetic and non-magnetic; magnetic assemblies for motors, magnetic levitation, sensing and separation technologies; and most recently we have responded to customer requests to develop and supply ultra-high performance permanent magnet motors for select applications. • Let’s start by answering this question: What makes rare earth elements so special? • The rare earth elements consist of the 15 lanthanide elements (lanthanum to lutetium) plus yttrium and scandium. • Yttrium and scandium are directly above lanthanum in the periodic table and have chemical properties that are very similar to the lanthanides – that is why they are usually included with them. • Note cesium and barium precede lanthanum in row 6 of the periodic table and that hafnium, element number 72, continues row 6 right after lutetium, the lanthanide with the highest atomic number, 71. • Note too, row 4 of the table which contains the transition elements including iron, cobalt and nickel. We’ll be making some comparison between the two groups of elements in later slides. • Rare earth elements (REEs) have considerable chemical similarities thus making them difficult to separate from each other. • That is one reason they were late in being discovered, isolated and incorporated in alloys and compounds. -

Ionic Interactions in Lanthanide Halides
Ionic Interactions in Lanthanide Halides Z. Akdeniz, Z. Çiçek, and M. P. Tosia Department of Physics, University of Istanbul, Istanbul, Turkey and Abdus Salam International Centre for Theoretical Physics, 1-34014 Trieste, Italy a INFM and Classe di Scienze, Scuola Normale Superiore, 1-56126 Pisa, Italy Reprint requests to Prof. M. P. T.; Fax: +39-050-563513; E-mail: [email protected] Z. Naturforsch. 55 a, 861-866 (2000); received September 27, 2000 We determine a model of the ionic interactions in RX3 compounds (where R is a metal in the rare-earth series from La to Lu and X = CI, Br or I) by an analysis of data on the static and dynamic structure of their molecular monomers. The potential energy function that we adopt is patterned after earlier work on Aluminium trichloride [Z. Akdeniz and M. P. Tosi, Z. Naturforsch. 54a, 180 (1999)], but includes as an essential element the electric polarizability of the trivalent metal ion to account for a pyramidal shape of RX3 molecules. From data referring mostly to trihalides of elements at the ends and in the middle of the rare-earth series (/. e. LaX3, GdX3 and LuX3), we propose systematic variations for the effective valence, ionic radius and electric polarizability of the metal ions across the series. As a first application of our results we predict the structure of the Dy2Cl6 and Dy2Br6 molecular dimers and demonstrate by comparison with electron diffraction data that lanthanide-ion polarizability plays a quantitative role also in this state of tetrahedral-like coordination. Key words: Ionic Clusters; Molten Salts. -

The Elements.Pdf
A Periodic Table of the Elements at Los Alamos National Laboratory Los Alamos National Laboratory's Chemistry Division Presents Periodic Table of the Elements A Resource for Elementary, Middle School, and High School Students Click an element for more information: Group** Period 1 18 IA VIIIA 1A 8A 1 2 13 14 15 16 17 2 1 H IIA IIIA IVA VA VIAVIIA He 1.008 2A 3A 4A 5A 6A 7A 4.003 3 4 5 6 7 8 9 10 2 Li Be B C N O F Ne 6.941 9.012 10.81 12.01 14.01 16.00 19.00 20.18 11 12 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 3 Na Mg IIIB IVB VB VIB VIIB ------- VIII IB IIB Al Si P S Cl Ar 22.99 24.31 3B 4B 5B 6B 7B ------- 1B 2B 26.98 28.09 30.97 32.07 35.45 39.95 ------- 8 ------- 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 4 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 39.10 40.08 44.96 47.88 50.94 52.00 54.94 55.85 58.47 58.69 63.55 65.39 69.72 72.59 74.92 78.96 79.90 83.80 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 5 Rb Sr Y Zr NbMo Tc Ru Rh PdAgCd In Sn Sb Te I Xe 85.47 87.62 88.91 91.22 92.91 95.94 (98) 101.1 102.9 106.4 107.9 112.4 114.8 118.7 121.8 127.6 126.9 131.3 55 56 57 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 6 Cs Ba La* Hf Ta W Re Os Ir Pt AuHg Tl Pb Bi Po At Rn 132.9 137.3 138.9 178.5 180.9 183.9 186.2 190.2 190.2 195.1 197.0 200.5 204.4 207.2 209.0 (210) (210) (222) 87 88 89 104 105 106 107 108 109 110 111 112 114 116 118 7 Fr Ra Ac~RfDb Sg Bh Hs Mt --- --- --- --- --- --- (223) (226) (227) (257) (260) (263) (262) (265) (266) () () () () () () http://pearl1.lanl.gov/periodic/ (1 of 3) [5/17/2001 4:06:20 PM] A Periodic Table of the Elements at Los Alamos National Laboratory 58 59 60 61 62 63 64 65 66 67 68 69 70 71 Lanthanide Series* Ce Pr NdPmSm Eu Gd TbDyHo Er TmYbLu 140.1 140.9 144.2 (147) 150.4 152.0 157.3 158.9 162.5 164.9 167.3 168.9 173.0 175.0 90 91 92 93 94 95 96 97 98 99 100 101 102 103 Actinide Series~ Th Pa U Np Pu AmCmBk Cf Es FmMdNo Lr 232.0 (231) (238) (237) (242) (243) (247) (247) (249) (254) (253) (256) (254) (257) ** Groups are noted by 3 notation conventions. -

24. SOLUTION CHEMISTRY of the LANTHANIDE ELEMENTS Gregory
.24. SOLUTION CHEMISTRY OF THE LANTHANIDE ELEMENTS Gregory R. Choppin Department of Chemistry Florida State University Tallahassee, Florida 32306,U.S.A. ABSTRACT Complexes of the lanthanide elements provide insight into the factors which are important in ionically bonded systems. The enthalpy and entropy terms of complexation are a measure of the role of hydration effects while the free energy measures the metal-ligand interaction. The role of ligand basicity in the nature of the complex as an inner sphere or a solvent- separated outer sphere species is reviewed. An equation which has been used successfully to calculate stability constants which agree with experimental values is described and shown to be useful in establishing models for lanthanide complexing. The stability of complexes as a function of canonical structure of organic ligands and of ligand ring size is discussed. A study of charge polarization in conjugated organic ligands complexed by lanthanide cations is described. .25. SOLUTION CHEMISTRY OF THE LANTHANIDE ELEMENTS I. INTRODUCTION The development of separation methods using ion exchange resins has provided chemists with large quantities of very pure individual lanthanide elements. As a result, in the last 30 years research in lanthanide chemistry has been relatively intensive and has led to a rather satisfactory understanding of the chemical behavior of these elements as well as to the growing utilization of them in a variety of technological applications. A central feature of lanthanide chemistry is the strongly ionic character of the bonding between lanthanide cations and other atoms. As a result of this ionicity, they can be classified as "hard" (strongly acidic) cations. -

Reactions of Lanthanides and Actinides in Molten Alkali Metal/Polychalcogenide Fluxes at Intermediate Temperatures (250-600 °C) Anthony C
Chem. Mater. 1997, 9, 387-398 387 Reactions of Lanthanides and Actinides in Molten Alkali Metal/Polychalcogenide Fluxes at Intermediate Temperatures (250-600 °C) Anthony C. Sutorik and Mercouri G. Kanatzidis* Department of Chemistry and Center for Fundamental Materials Research, Michigan State University, East Lansing, Michigan 48824 Received August 26, 1996X From the reaction of the elemental lanthanides and actinides in molten alkali metal/ polychalcogenide salts, several new ternary compounds have been discovered. Specifically, these phases are K4USe8 (I) and ALnQ4 (II), where A ) K, Ln ) Ce or Tb, and Q ) Se or A ) Rb, Ln ) Ce, Q ) Te; and NaLnS3 (III), where Ln ) La, Ce. The K4USe8 crystallizes in the orthorhombic space group Fdd2 (No. 43) with a ) 17.331(4) Å, b ) 20.584(3) Å, c ) 8.811(3) Å, Z ) 8. The KCeSe4 crystallizes in the tetragonal space group P4/nbm (No. 125) with a ) 6.376(2) Å, c ) 8.327(1) Å, Z ) 2. The KTbSe4 crystallizes in the tetragonal space group P4/nbm (No. 125) with a ) 6.255(2) Å, c ) 8.227(1) Å, Z ) 2. The RbCeTe4 crystallizes in the tetragonal space group P4/nbm (No. 125) with a ) 6.952(3) Å, c ) 9.084(4) Å, Z ) 2. The NaCeS3 crystallizes in the orthorhombic space group Pmmn (No. 59) with a ) 5.683(1) Å, b ) 4.238(2) Å, c ) 9.802(2) Å, Z ) 2. The NaLaS3 crystallizes in the orthorhombic space group Pmmn (No. 59) with a ) 5.752(4) Å, b ) 4.2796(6) Å, c ) 9.841(2) Å, Z ) 2. -
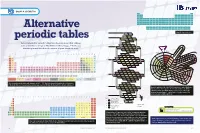
Alternative Periodic Tables Have Been Proposed That Address Mn Fe 25 28 Cr P S Co Some of the Shortcomings of Mendeleev’S Table (See Pp
GROUP 4: CHEMISTRY H He Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Rf Db Sg Bh Hs Mt Ds Rg Cn Nh Fl Mc Lv Ts Og Ce Pr Nd Pm Sm Eu Gd Td Dy Ho Er Tm Yb Lu Alternative Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr n=1 Simplified periodic table H He* 1 2 N O n=2 7 8 C Li Be F• periodic tables 6 3 4 9 B Ne* 5 10 Several alternative periodic tables have been proposed that address Mn Fe 25 28 Cr P S Co some of the shortcomings of Mendeleev’s table (see pp. 2–6). Do you n=3 24 15 16 27 V Si Na Mg Cl• Ni PERIODICPERIODIC 23 14 11 12 17 28 DIVIDE think they work better than the version on your classroom wall? Ti Al Ar* Cu 22 2 2 29 ALKALI Sc Zn 21 30 METALS NOBLE 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 Gd Tb GASES Uue 2 65 Og Ubn Cm 1 H He Eu Tc Ru Dy Fr SUPERACTINIDES Am Bk 63 43 44 66 Rn Pu Sm Mo As Se Rh Ho Np Gd Tb 2 Li Be B C N O F Ne Ra U Eu 62 42 33 34 45 67 Xe Cs n=4 Pa Sm Dy Cf Pm Nb Ge K Ca Br• Pb Er Th Pm 3 Na Mg Al Si P S Cl Ar 61 41 32 19 20 35 46 68 Ts Kr Rb Ba Ac Nd Ho Pr Er Es Nd Zr Ga Kr* Ag Tm At K Sr Tm 4 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 60 40 31 36 47 69 I Ar Ce Fm La Yb Md Pr Y Cd Yb Br Na Ca Lu 59 39 48 70 Cl Ne No 5 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe He Li LANTHANIDES & ACTINIDESLr F Mg Y Ce Lu Be Hf Rf 58 71 O H Sc Zr 6 Cs Ba La Ce Pr Nd Pm Sm Eu Gd Td Dy Ho Er Tm Yb Lu Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn S Ta Db Se