The Ansa Subthalamica: a Neglected Fiber Tract
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Gene Expression of Prohormone and Proprotein Convertases in the Rat CNS: a Comparative in Situ Hybridization Analysis
The Journal of Neuroscience, March 1993. 73(3): 1258-1279 Gene Expression of Prohormone and Proprotein Convertases in the Rat CNS: A Comparative in situ Hybridization Analysis Martin K.-H. Schafer,i-a Robert Day,* William E. Cullinan,’ Michel Chri?tien,3 Nabil G. Seidah,* and Stanley J. Watson’ ‘Mental Health Research Institute, University of Michigan, Ann Arbor, Michigan 48109-0720 and J. A. DeSeve Laboratory of *Biochemical and 3Molecular Neuroendocrinology, Clinical Research Institute of Montreal, Montreal, Quebec, Canada H2W lR7 Posttranslational processing of proproteins and prohor- The participation of neuropeptides in the modulation of a va- mones is an essential step in the formation of bioactive riety of CNS functions is well established. Many neuropeptides peptides, which is of particular importance in the nervous are synthesized as inactive precursor proteins, which undergo system. Following a long search for the enzymes responsible an enzymatic cascade of posttranslational processing and mod- for protein precursor cleavage, a family of Kexin/subtilisin- ification events during their intracellular transport before the like convertases known as PCl, PC2, and furin have recently final bioactive products are secreted and act at either pre- or been characterized in mammalian species. Their presence postsynaptic receptors. Initial endoproteolytic cleavage occurs in endocrine and neuroendocrine tissues has been dem- C-terminal to pairs of basic amino acids such as lysine-arginine onstrated. This study examines the mRNA distribution of (Docherty and Steiner, 1982) and is followed by the removal these convertases in the rat CNS and compares their ex- of the basic residues by exopeptidases. Further modifications pression with the previously characterized processing en- can occur in the form of N-terminal acetylation or C-terminal zymes carboxypeptidase E (CPE) and peptidylglycine a-am- amidation, which is essential for the bioactivity of many neu- idating monooxygenase (PAM) using in situ hybridization ropeptides. -

MRI Atlas of the Human Deep Brain Jean-Jacques Lemaire
MRI Atlas of the Human Deep Brain Jean-Jacques Lemaire To cite this version: Jean-Jacques Lemaire. MRI Atlas of the Human Deep Brain. 2019. hal-02116633 HAL Id: hal-02116633 https://hal.uca.fr/hal-02116633 Preprint submitted on 1 May 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Distributed under a Creative Commons Attribution - NonCommercial - NoDerivatives| 4.0 International License MRI ATLAS of the HUMAN DEEP BRAIN Jean-Jacques Lemaire, MD, PhD, neurosurgeon, University Hospital of Clermont-Ferrand, Université Clermont Auvergne, CNRS, SIGMA, France This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/ or send a letter to Creative Commons, PO Box 1866, Mountain View, CA 94042, USA. Terminologia Foundational Model Terminologia MRI Deep Brain Atlas NeuroNames (ID) neuroanatomica usages, classical and french terminologies of Anatomy (ID) Anatomica 1998 (ID) 2017 http://fipat.library.dal.ca In -

Memory Loss from a Subcortical White Matter Infarct
J Neurol Neurosurg Psychiatry: first published as 10.1136/jnnp.51.6.866 on 1 June 1988. Downloaded from Journal of Neurology, Neurosurgery, and Psychiatry 1988;51:866-869 Short report Memory loss from a subcortical white matter infarct CAROL A KOOISTRA, KENNETH M HEILMAN From the Department ofNeurology, College ofMedicine, University ofFlorida, and the Research Service, Veterans Administration Medical Center, Gainesville, FL, USA SUMMARY Clinical disorders of memory are believed to occur from the dysfunction of either the mesial temporal lobe, the mesial thalamus, or the basal forebrain. Fibre tract damage at the level of the fornix has only inconsistently produced amnesia. A patient is reported who suffered a cerebro- vascular accident involving the posterior limb of the left internal capsule that resulted in a persistent and severe disorder of verbal memory. The inferior extent of the lesion effectively disconnected the mesial thalamus from the amygdala and the frontal cortex by disrupting the ventral amygdalofugal and thalamic-frontal pathways as they course through the diencephalon. This case demonstrates that an isolated lesion may cause memory loss without involvement of traditional structures associated with memory and may explain memory disturbances in other white matter disease such as multiple sclerosis and lacunar state. Protected by copyright. Memory loss is currently believed to reflect grey day of his illness the patient was transferred to Shands matter damage of either the mesial temporal lobe,' -4 Teaching Hospital at the University of Florida for further the mesial or the basal forebrain.'0 l evaluation. thalamus,5-9 Examination at that time showed the patient to be awake, Cerebrovascular accidents resulting in memory dys- alert, attentive and fully oriented. -

Two Fiber Pathways Connecting Amygdala and Prefrontal Cortex in Humans and Monkeys
bioRxiv preprint doi: https://doi.org/10.1101/561811; this version posted March 20, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Two fiber pathways connecting amygdala and prefrontal cortex in humans and monkeys Davide Folloni1,2*, Jérôme Sallet1,2, Alexandre A. Khrapitchev3, Nicola R. Sibson3, Lennart Verhagen1,2†, Rogier B. Mars2,4† 1Wellcome Integrative Neuroimaging (WIN), Department of Experimental Psychology, University of Oxford, Oxford, United Kingdom 2Wellcome Integrative Neuroimaging (WIN), Centre for Functional MRI of the Brain (FMRIB), Nuffield Department of Clinical Neurosciences, John Radcliffe Hospital, University of Oxford, Oxford, United Kingdom 3Cancer Research UK and Medical Research Council Oxford Institute for Radiation Oncology, Department of Oncology, University of Oxford, Oxford, United Kingdom 4Donders Institute for Brain, Cognition and Behaviour, Radboud University Nijmegen, Nijmegen, The Netherlands †Authors contributed equally to the work *To whom correspondence should be addressed: Address: Davide Folloni, Department of Experimental Psychology, University of Oxford, Tinsley Building, Mansfield Road, Oxford, OX1 3SR, UK E-mail: [email protected] 1 bioRxiv preprint doi: https://doi.org/10.1101/561811; this version posted March 20, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Abstract The interactions between amygdala and prefrontal cortex are pivotal to many neural processes involved in learning, decision-making, emotion, and social regulation. -
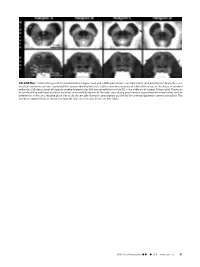
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

Neuroimaging Advances in Deep Brain Stimulation: Review of Indications, Anatomy, and Brain Connectomics
Published August 13, 2020 as 10.3174/ajnr.A6693 REVIEW ARTICLE Neuroimaging Advances in Deep Brain Stimulation: Review of Indications, Anatomy, and Brain Connectomics E.H. Middlebrooks, R.A. Domingo, T. Vivas-Buitrago, L. Okromelidze, T. Tsuboi, J.K. Wong, R.S. Eisinger, L. Almeida, M.R. Burns, A. Horn, R.J. Uitti, R.E. Wharen Jr, V.M. Holanda, and S.S. Grewal ABSTRACT SUMMARY: Deep brain stimulation is an established therapy for multiple brain disorders, with rapidly expanding potential indi- cations. Neuroimaging has advanced the field of deep brain stimulation through improvements in delineation of anatomy, and, more recently, application of brain connectomics. Older lesion-derived, localizationist theories of these conditions have evolved to newer, network-based “circuitopathies,” aided by the ability to directly assess these brain circuits in vivo through the use of advanced neuroimaging techniques, such as diffusion tractography and fMRI. In this review, we use a combination of ultra-high-field MR imaging and diffusion tractography to highlight relevant anatomy for the currently approved indications for deep brain stimulation in the United States: essential tremor, Parkinson disease, drug-resistant epilepsy, dystonia, and obsessive-compulsive disorder. We also review the literature regarding the use of fMRI and diffusion tractography in under- standing the role of deep brain stimulation in these disorders, as well as their potential use in both surgical targeting and de- vice programming. ABBREVIATIONS: AL ¼ ansa lenticularis; ALIC -

Motor Systems Basal Ganglia
Motor systems 409 Basal Ganglia You have just read about the different motor-related cortical areas. Premotor areas are involved in planning, while MI is involved in execution. What you don’t know is that the cortical areas involved in movement control need “help” from other brain circuits in order to smoothly orchestrate motor behaviors. One of these circuits involves a group of structures deep in the brain called the basal ganglia. While their exact motor function is still debated, the basal ganglia clearly regulate movement. Without information from the basal ganglia, the cortex is unable to properly direct motor control, and the deficits seen in Parkinson’s and Huntington’s disease and related movement disorders become apparent. Let’s start with the anatomy of the basal ganglia. The important “players” are identified in the adjacent figure. The caudate and putamen have similar functions, and we will consider them as one in this discussion. Together the caudate and putamen are called the neostriatum or simply striatum. All input to the basal ganglia circuit comes via the striatum. This input comes mainly from motor cortical areas. Notice that the caudate (L. tail) appears twice in many frontal brain sections. This is because the caudate curves around with the lateral ventricle. The head of the caudate is most anterior. It gives rise to a body whose “tail” extends with the ventricle into the temporal lobe (the “ball” at the end of the tail is the amygdala, whose limbic functions you will learn about later). Medial to the putamen is the globus pallidus (GP). -

Diencephalon Diencephalon
Diencephalon Diencephalon • Thalamus dorsal thalamus • Hypothalamus pituitary gland • Epithalamus habenular nucleus and commissure pineal gland • Subthalamus ventral thalamus subthalamic nucleus (STN) field of Forel Diencephalon dorsal surface Diencephalon ventral surface Diencephalon Medial Surface THALAMUS Function of the Thalamus • Sensory relay – ALL sensory information (except smell) • Motor integration – Input from cortex, cerebellum and basal ganglia • Arousal – Part of reticular activating system • Pain modulation – All nociceptive information • Memory & behavior – Lesions are disruptive Classification of Thalamic Nuclei I. Lateral Nuclear Group II. Medial Nuclear Group III. Anterior Nuclear Group IV. Posterior Nuclear Group V. Metathalamic Nuclear Group VI. Intralaminar Nuclear Group VII. Thalamic Reticular Nucleus Classification of Thalamic Nuclei LATERAL NUCLEAR GROUP Ventral Nuclear Group Ventral Posterior Nucleus (VP) ventral posterolateral nucleus (VPL) ventral posteromedial nucleus (VPM) Input to the Thalamus Sensory relay - Ventral posterior group all sensation from body and head, including pain Projections from the Thalamus Sensory relay Ventral posterior group all sensation from body and head, including pain LATERAL NUCLEAR GROUP Ventral Lateral Nucleus Ventral Anterior Nucleus Input to the Thalamus Motor control and integration Projections from the Thalamus Motor control and integration LATERAL NUCLEAR GROUP Prefrontal SMA MI, PM SI Ventral Nuclear Group SNr TTT GPi Cbll ML, STT Lateral Dorsal Nuclear Group Lateral -

Basal Ganglia 519 Basal Ganglia
Basal Ganglia 519 Basal Ganglia You have just read about the different motor-related cortical areas. Premotor areas are involved in planning, while MI is involved in execution; corticospinal fibers from MI target LMNs. What you don’t know is that the cortical areas involved in movement control need some “help” from the basal ganglia in order to smoothly orchestrate motor behaviors. Without the information from the basal ganglia, the cortex is unable to properly direct motor control, and the deficits seen in Parkinson’s and Huntington’s disease and other movement disorders become apparent. The adjacent drawings show level 16 of our series of brain sections. Some of the important “players” can be seen. They are the caudate nucleus and the putamen nucleus. Together they are called the neostriatum or simply striatum. Next to the putamen is the globus pallidus, and it can be seen to have two parts, a lateral (external or outer) segment and a medial (internal or inner) segment. The putamen and globus pallidus, which lie adjacent to each other, are called the lenticular nucleus. Finally, the striatum and the globus pallidus are called the corpus striatum. You can also see a nice thick fiber bundle associated with the globus pallidus called the ansa lenticularis. This fasciculus passes under (ventral) the posterior limb of the internal capsule. While not apparent in this section, the ansa lenticularis then heads dorsally to reach the motor thalamic nuclei (VA/ VL). The ansa arises from cells of the inner segment of the globus pallidus and terminates in the ipsilateral VA/VL. -

The Language Connectome: New Pathways, New Concepts
NROXXX10.1177/1073858413513502The NeuroscientistDick and others 513502research-article2013 Review The Neuroscientist 2014, Vol. 20(5) 453 –467 The Language Connectome: New © The Author(s) 2013 Reprints and permissions: sagepub.com/journalsPermissions.nav Pathways, New Concepts DOI: 10.1177/1073858413513502 nro.sagepub.com Anthony Steven Dick1, Byron Bernal2, and Pascale Tremblay3 Abstract The field of the neurobiology of language is experiencing a paradigm shift in which the predominant Broca–Wernicke– Geschwind language model is being revised in favor of models that acknowledge that language is processed within a distributed cortical and subcortical system. While it is important to identify the brain regions that are part of this system, it is equally important to establish the anatomical connectivity supporting their functional interactions. The most promising framework moving forward is one in which language is processed via two interacting “streams”—a dorsal and ventral stream—anchored by long association fiber pathways, namely the superior longitudinal fasciculus/ arcuate fasciculus, uncinate fasciculus, inferior longitudinal fasciculus, inferior fronto-occipital fasciculus, and two less well-established pathways, the middle longitudinal fasciculus and extreme capsule. In this article, we review the most up-to-date literature on the anatomical connectivity and function of these pathways. We also review and emphasize the importance of the often overlooked cortico-subcortical connectivity for speech via the “motor stream” and associated -

INTERNAL CAPSULE • Projection Fibres- Internal Capsule
INTERNAL CAPSULE • Projection fibres- Internal capsule r ~OnQih..!1c:lll i~J ~asd · I.JS OOi 1n1ssur DEFINITION Rostrum ol co,pi.c:; • Projection fibres c.allosum (white matter) between nucleus • caudate nucleus and /---~.__~---- :r~~l thalamus medially capS,Jle Pos1enu1 1111-0 of 1mema1 • lentiform nucleus capsule Thalamus laterally Actf'CNeflocular part of internal capsule 10.24 Horizontal sec11u11 01 lilt cerebral hemisphere st10win9 the Ooc1pr1~• p<Jlt!' - nl lhti rnla o-n~I r~n-e-11l it • Internal Capsule- A compact bundle of fibres through which the large collections of fibres pass, including- • Thalamocortical fibres • Corticothalamic fibres • Corticopontine fibres • Corticobulbar fibres • Corticospinal fibres • The fibres project from the cerebral cortex to the various nuclei of the extrapyramidal system (e.g., the putamen and caudate nucleus). • It is a continuous sheet of fibres that forms the medial boundary of the lenticular nucleus. • It continues around posteriorly and inferiorly to partially envelop this nucleus. • Inferiorly, many of the fibres of the internal capsule funnel into the cerebral peduncles. • Superiorly, the fibres fan out into the corona radiata. • Here, they travel in the cerebral white matter to reach their cortical origins or destinations. The internal capsule is divided into 5 regions: • The anterior limb is the portion between the lenticular nucleus and the head of the caudate nucleus; • The posterior limb is the portion between the lenticular nucleus and the thalamus; • The genu is the portion at the junction of the above 2 parts and is adjacent to the interventricular foramen; • The retrolenticular part is the portion posterior to the lenticular nucleus; • The sublenticular part is the portion inferior to the lenticular nucleus. -

White Matter Anatomy: What the Radiologist Needs to Know
White Matter Anatomy What the Radiologist Needs to Know Victor Wycoco, MBBS, FRANZCRa, Manohar Shroff, MD, DABR, FRCPCa,*, Sniya Sudhakar, MBBS, DNB, MDb, Wayne Lee, MSca KEYWORDS Diffusion tensor imaging (DTI) White matter tracts Projection fibers Association Fibers Commissural fibers KEY POINTS Diffusion tensor imaging (DTI) has emerged as an excellent tool for in vivo demonstration of white matter microstructure and has revolutionized our understanding of the same. Information on normal connectivity and relations of different white matter networks and their role in different disease conditions is still evolving. Evidence is mounting on causal relations of abnormal white matter microstructure and connectivity in a wide range of pediatric neurocognitive and white matter diseases. Hence there is a pressing need for every neuroradiologist to acquire a strong basic knowledge of white matter anatomy and to make an effort to apply this knowledge in routine reporting. INTRODUCTION (Fig. 1). However, the use of specific DTI sequences provides far more detailed and clini- DTI has allowed in vivo demonstration of axonal cally useful information. architecture and connectivity. This technique has set the stage for numerous studies on normal and abnormal connectivity and their role in devel- DIFFUSION TENSOR IMAGING: THE BASICS opmental and acquired disorders. Referencing established white matter anatomy, DTI atlases, Using appropriate magnetic field gradients, and neuroanatomical descriptions, this article diffusion-weighted sequences can be used to summarizes the major white matter anatomy and detect the motion of the water molecules to and related structures relevant to the clinical neurora- from cells. This free movement of the water mole- diologist in daily practice.