Urine Mescaline Screening with a Biochip Array Immunoassay and Quantification by Gas Chromatography–Mass Spectrometry
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Drug and Medication Classification Schedule
KENTUCKY HORSE RACING COMMISSION UNIFORM DRUG, MEDICATION, AND SUBSTANCE CLASSIFICATION SCHEDULE KHRC 8-020-1 (11/2018) Class A drugs, medications, and substances are those (1) that have the highest potential to influence performance in the equine athlete, regardless of their approval by the United States Food and Drug Administration, or (2) that lack approval by the United States Food and Drug Administration but have pharmacologic effects similar to certain Class B drugs, medications, or substances that are approved by the United States Food and Drug Administration. Acecarbromal Bolasterone Cimaterol Divalproex Fluanisone Acetophenazine Boldione Citalopram Dixyrazine Fludiazepam Adinazolam Brimondine Cllibucaine Donepezil Flunitrazepam Alcuronium Bromazepam Clobazam Dopamine Fluopromazine Alfentanil Bromfenac Clocapramine Doxacurium Fluoresone Almotriptan Bromisovalum Clomethiazole Doxapram Fluoxetine Alphaprodine Bromocriptine Clomipramine Doxazosin Flupenthixol Alpidem Bromperidol Clonazepam Doxefazepam Flupirtine Alprazolam Brotizolam Clorazepate Doxepin Flurazepam Alprenolol Bufexamac Clormecaine Droperidol Fluspirilene Althesin Bupivacaine Clostebol Duloxetine Flutoprazepam Aminorex Buprenorphine Clothiapine Eletriptan Fluvoxamine Amisulpride Buspirone Clotiazepam Enalapril Formebolone Amitriptyline Bupropion Cloxazolam Enciprazine Fosinopril Amobarbital Butabartital Clozapine Endorphins Furzabol Amoxapine Butacaine Cobratoxin Enkephalins Galantamine Amperozide Butalbital Cocaine Ephedrine Gallamine Amphetamine Butanilicaine Codeine -

Orthomolecular Approach to the Treatment of Schizophrenia
.c= DOCUMENT RESUME ED 123 858 EC 090 226 . _AUTHOR Lall, Geeta Rani . TITLE Grthoolecular Approach to- the Treatment of Schizophrenia, Childhood'Psychoses; and Allied Disorders Such as: Hyperactivity, Autism, Hypoglycemia, and Sub Clinical Pellagra. 1 INSTITUTION Andrews Univ., Berrien Sprints, Mich. Depts of Education. PUB DATE -May 76 15p. EDRS.. PRICE MU-n.83 5C-$1.67 Plus Post'age.. - DESCRIPTORS *Emotionally Disturbed; Exceptional Child Research; *Medical Treatment; *Nutrition; *Psychiatry; ' Psychosis; Schizophrenia IDENTIFIERS Orthomolecular Psychiatry ABSTRACT . Pressnted is research' to support the orthomo lecular rather than the psychodynamic approach to treating schizdpkrenia, psychoses, and allied disorders in c41dren. The orthomolecular approach, also known as orthomolecular psychiatry, is reported to t involve the administration ofmegavitamins (following a study to determine biochemical needs), appropriate diet, exercise, psychbtherapy, and other drugs. Advantages of the orthomolecular approach are noted to include better .treatment results; lest expensive and time-consuming treatment; less guilt feelings and alienation of families; removal of much of the stigma and fear ofthe medical pfoblem because of good prognosis; and more objective, accurate, and easily obtained diagnosis. ISB) -***********A******************************************************* . * DocumentS'acquired'by ERIC include many informal unpublished. 'Jic * .materials not available from other sources. EPIC makes ver effort * * to obtain ,,the best copy. available. Neverth4less, items . marginal * * reproducibility are often encountered and this affects the quality * * of the microfiche and hardcopy reproductions ERIC makes available * * via the EPIC Dowlent Reproduction Service (EbRS). EDRS is not * * responsible for the - quality of the original document. Teproductions'.* * supplied by EDRS are the best tiat can be made fiom the original. * ********44*****************************4********4********************** ,-, VI . -

Abram Hoffer, MD, Phd Dr
July 2002 Issue | Abram Hoffer, MD, PhD Dr. Jeffrey Bland - http://jeffreybland.com July 2002 Issue | Abram Hoffer, MD, PhD http://jeffreybland.com/knowledgebase/july-2002-issue-abram-hoffer-md-phd/ DOWNLOAD AUDIO | Welcome to Functional Medicine Update for July 2002. This month I will follow up on a theme from our Ninth International Symposium on Functional Medicine, which we held in May at the Diplomat Hotel and Spa in Fort Lauderdale, Florida. I refer to orthomolecular psychiatry, a topic Dr. Abram Hoffer brought to our attention at the symposium. Together with Dr. Linus Pauling, Dr. Hoffer was an originator of that field. I begin with a quote from an article titled "Orthomolecular Psychiatry," which was published in Science magazine in 1968.1 Dr. Pauling, the principal author of that article, won two Nobel Prizes, one for chemistry and one for peace. "The methods principally used now for treating patients with mental disease are psychotherapy (psychoanalysis and related efforts to provide insight and to decrease environmental stress), chemotherapy (mainly with the use of powerful synthetic drugs, such as chlorpromazine, or powerful natural products from plants, such as reserpine), and convulsive or shock therapy (electroconvulsive therapy, insulin coma therapy, pentylenetetraze! shock therapy). I have reached the conclusion, through arguments summarized in the following paragraphs, that another general method or treatment, which may be called orthomolecular therapy, may be found to be of great value, and may turn out to be the best -
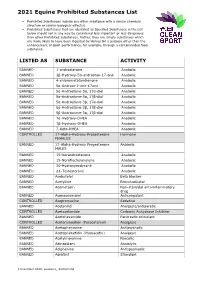
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

Effectsofmescaline,LSD-25, and Adrenochromeondepthelectograms, M.N
1 LSD187 Arch.Neurol.Psychiatr., Chicago 7__5_579 , (1956). EffectsofMescaline,LSD-25, and AdrenochromeonDepthElectograms, M.n BERT E. $¢HWARZ, M.D. psychosis and three who had chronic schizophrenia CARL W. SEM-JACOISEN, M.D. were studied by means of the techniques of depth ond electrography developed and defined in this labora- MAGNUS C. Pt'riRSiH, M.D., Rochester, Minn. tory.* One of the epileptic and two of the schizo- +++ phrenic patients also received adrenochrome. Each l__$_l1_l3_lllull$l$_Mlllllll$__u$$llu_lll__[1$l_l__ll_l1_l_l___lillllpatientuIllu_l_l_hadLllMuexten_lliil__l_lsive _controll1lll_[1ll___li_lstudies,ul1_[_lastingi__l_lias_llong as two weeks per patient, previous to the time of In a recent report concerning the re- administration of the drug. Further control obser- versibility by chlorpromazine of psychoses vations were repeated every day of the study. Elec- induced by means of mescaline and d-lysergic trocardiograms were recorded simultaneously with depth electrograms in two patients. acid diethylamide (LSD-25), careful atten- The dose of mescaline varied between 400 rag. tion was given to the clinical and electro- and 1 gm., whereas that of LSD-25 ranged from encephalographic changes? Although the 50_ to 1503'. The dose of adrenochrome# varied clinical changes of this reversal effect were between 50 and 75 mg. The mescaline and LSD- timmediate and striking, the conventional 25 were administered orally ; the adrenochrome was electroenceph.alographic changes were mini- given intravenously as a freshly prepared solution mal, and it was difficult to assess them. It in 10 cc. of sterile water. At the presumed height of the reactions to mescaline and LSD-25, each was noted in another recent study that patient received an intravenous injection of 50 rag. -

The Toxicologist
Preface This second issue of the Toxicologist is devoted exclusively to the abstracts of the platform and poster sessions of the 21st Annual Conference of the Society of Toxicology, held at the Sheraton-Boston Hotel, Boston, MA, February, 22-26, 1982. The abstracts are reproduced, without editing, as accepted by the Program Committee of the Society of Toxicology, and appear in the same numerical sequence as the papers appear in the Conference's official program. An alphabetical author index, listing the corresponding paper number, appears on pp. 194. Copies of this issue are available at $5 each (U.S. funds) from Society of Toxicology 475 Wolf Ledges Parkway Akron, OH 44311-1087 ® 1982, Society of Toxicology 1 BUTYLATEDHYDROXYTOLUENE DOES NOT INHIBIT CIPRO ment of a 6-MC protein or amino acid adduct in FIBRATE AND DI-(2-ETHYLHEXYL)PHTHALATEINDUCED 6-MC photoallergenicity. (Supported in part by PEROXISOMEPROLIFERATION IN RAT LIVER. N.D. the Research Institute for Fragrance Materials). Lalwani, H. Hayashi, M.K. Reddy, C. Moehle, and ) J.K. Reddy, Dept. Pathol., Northwestern Univ. Med. Sch., Chicago, IL. 3 ALTERATIONOF CALCIUM-DEPENDENTGABARELEASE AND Evidence now suggests that hepatic peroxisome THEENZYME ACTIVITIES OF GLUTAMICACID DECARBOXY proliferators as a class are carcinogenic. It is LASE(GAD) AND GABA-y-KETOGLUTARATE TRANSAMINASE hypothesized that persistant proliferation of (GABA-T)IN RESPONSETO THEACUTE AND CHRONIC AD peroxisomes and increase in peroxisomal S-oxida MINISTRATIONOF PHENCYCLIDINE(PCP). J. C. Norris, tion initiates the neoplastic transformation of T. Nabeshima, S. P. Sivam and I. K. Ho, Dept. liver cells by increasing the intracellular pro Pharmacol. & Toxicol., Univ. -
Task Force Report on Megavitamin and Orthomolecular Therapy In
MEGAVITAMIN AND ORTHOMOLECULAR THERAPY IN PSYCHIATRY A Report of the APA Task Force on Vitamin Therapy in Psychiatry Morris A. Lipton, Ph.D., M.D., Chairman Thomas A. Ban, M.D. Francis J. Kane, M.D. Jerome Levine, M.D. Loren R. Mosher, M.D. Richard Wittenborn, Ph.D. (Consultant) Approved for Publication by the APA Council on Research and Development Sidney Malitz, M.D., Chairman Russell R. Monroe, M.D., Vice-Chairman Bernard C. Glueck, M.D. Louis Jolyon West, M.D. John J. Schwab, M.D. Walter Shervington, D.C. (Consultant) Fritz Henn, M.D., Falk Fellow Stuart Yudofsky, M.D., Falk Fellow American Psychiatric Association 1700 Eighteenth Street N.W. Washington, D.C. 20009 Task Force Reports This is the seventh report in a monograph series authorized by the Board of Trustees of the American Psychiatric Association to give wider dissemination to the findings of the Association's many commissions, committees and task forces that are called upon from time to time to evaluate the state of the art in a problem area of current concern to the profession, to related disciplines, and often to the public. Manifestly, the findings, opinions, and conclusions of Task Force Reports do not necessarily represent the views of the officers, trustees, or all members of the Association. Each report, however, does represent the thoughtful judgment and con sensus of the task force of experts who formulated it and it is considered a useful and substantive contribution to the ongoing analysis and evaluation of problems, programs, issues, and practices in a given area of concern. -
Chemical Structure-Related Drug-Like Criteria of Global Approved Drugs
Molecules 2016, 21, 75; doi:10.3390/molecules21010075 S1 of S110 Supplementary Materials: Chemical Structure-Related Drug-Like Criteria of Global Approved Drugs Fei Mao 1, Wei Ni 1, Xiang Xu 1, Hui Wang 1, Jing Wang 1, Min Ji 1 and Jian Li * Table S1. Common names, indications, CAS Registry Numbers and molecular formulas of 6891 approved drugs. Common Name Indication CAS Number Oral Molecular Formula Abacavir Antiviral 136470-78-5 Y C14H18N6O Abafungin Antifungal 129639-79-8 C21H22N4OS Abamectin Component B1a Anthelminithic 65195-55-3 C48H72O14 Abamectin Component B1b Anthelminithic 65195-56-4 C47H70O14 Abanoquil Adrenergic 90402-40-7 C22H25N3O4 Abaperidone Antipsychotic 183849-43-6 C25H25FN2O5 Abecarnil Anxiolytic 111841-85-1 Y C24H24N2O4 Abiraterone Antineoplastic 154229-19-3 Y C24H31NO Abitesartan Antihypertensive 137882-98-5 C26H31N5O3 Ablukast Bronchodilator 96566-25-5 C28H34O8 Abunidazole Antifungal 91017-58-2 C15H19N3O4 Acadesine Cardiotonic 2627-69-2 Y C9H14N4O5 Acamprosate Alcohol Deterrant 77337-76-9 Y C5H11NO4S Acaprazine Nootropic 55485-20-6 Y C15H21Cl2N3O Acarbose Antidiabetic 56180-94-0 Y C25H43NO18 Acebrochol Steroid 514-50-1 C29H48Br2O2 Acebutolol Antihypertensive 37517-30-9 Y C18H28N2O4 Acecainide Antiarrhythmic 32795-44-1 Y C15H23N3O2 Acecarbromal Sedative 77-66-7 Y C9H15BrN2O3 Aceclidine Cholinergic 827-61-2 C9H15NO2 Aceclofenac Antiinflammatory 89796-99-6 Y C16H13Cl2NO4 Acedapsone Antibiotic 77-46-3 C16H16N2O4S Acediasulfone Sodium Antibiotic 80-03-5 C14H14N2O4S Acedoben Nootropic 556-08-1 C9H9NO3 Acefluranol Steroid -
Successful Treatment of Schizophrenia Requires Optimal Daily Doses of Vitamin B3
Guest Alternative Medicine Review Volume 13, Number 4 2008 Editorial Successful Treatment of Schizophrenia Requires Optimal Daily Doses of Vitamin B3 For over 50 years Dr. Abram Hoffer has been educating clinicians about the need to correctly (optimally) dose schizophrenics with vitamin B3 (niacin; niacinamide). For the past 10 years I have, likewise, educated numerous naturopathic and medical doctors about the very same thing. For some reason, both types of clinicians routinely treat schizophrenic patients with plenty of vitamins, minerals, and other natural health products, but they never provide enough vitamin B3. In these authors opinions, schizophrenic patients cannot get well if not provided with optimal doses of vitamin B3. This prevents the real acceptance of nutritional treatment since clinicians will not observe favorable results when inadequate treatment is provided; their schizophrenic patients will continue to suffer needlessly. To understand the importance of vitamin B3 treatment, some background information is needed. Schizophrenia is characterized by a combination of perceptual changes (e.g., hallucinations) and thought disorders (e.g., delusions).1 These aberrant mental states, which can lead to psychotic behavior, cause a tremendous amount of emotional and psychological suffering. The cause of schizophrenia, the subject of much debate, is considered a biochemical imbalance, although certain genetic factors most certainly play a role. The majority of scientists and psychiatrists subscribe to the dopamine excess theory of schizophrenia; i.e., that too much dopamine is largely responsible for the symptoms of psychosis. However, since 1952, Hoffer, the founding father of orthomolecular medicine, has researched, published, and expanded on the adrenochrome theory of schizophrenia.1,2 He and his colleagues, Drs. -
Clinical Procedures in Treating Terminally Ill Cancer Patients with Vitamin C
Clinical Procedures in Treating Terminally Ill Cancer Patients with Vitamin C Abram Hoffer, M.D., Ph.D.1 Let me tell you what I am not. I am not an cut down the production of adrenalin, and if we oncologist, I'm not a pathologist, I'm not a GP, I could also prevent the oxidation of adrenalin to am a psychiatrist. Therefore you may want to adrenochrome, then we might have a therapy for know what a psychiatrist is doing messing about our patients. And that immediately led us to look with cancer. I think that's a legitimate question so at two chemicals. One is called nicotinic acid or I'd like to tell you briefly how I got into this very vitamin B3. Vitamin B3 is known to be a methyl interesting field. acceptor, which, by depleting the body of its In 1951, I was made director of psychiatric methyl groups could cut down the conversion of research for the Department of Health for the noradrenaline to adrenalin and that would be province of Saskatchewan. I didn't really know helpful, we thought. Secondly, we wanted to use what to do. I had one major advantage, I think, vitamin C as an antioxidant. Looking back now it over my colleagues. I didn't know any seems that we were 30 or 40 years ahead of psychiatry. You may laugh but that's very antioxidant theories. We wanted to decrease the important because I didn't have anyone who oxidation of adrenaline to adrenochrome. could tell me what we could not do. -

(12) Patent Application Publication (10) Pub. No.: US 2016/0361298 A1 Novick Et Al
US 20160361298A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2016/0361298 A1 Novick et al. (43) Pub. Date: Dec. 15, 2016 (54) METHODS AND COMPOSITIONS FOR A63/496 (2006.01) TREATING CANCER A 6LX 39/395 (2006.01) (71) Applicant: Globavir BioSciences, Inc., Los Altos, A63/496 (2006.01) CA (US) A63L/452 (2006.01) A6II 45/06 (2006.01) (72) Inventors: Paul Novick, San Francisco, CA (US); C07K 6/28 (2006.01) Jonathan Choy, Lafayette, CA (US); A6II 3/478 (2006.01) Sumit Mahajan, Foster City, CA (US); Ritika Prasad, Mountain View, CA A6II 3/167 (2006.01) (US); Shalabh Gupta, Mountain View, (52) U.S. Cl. CA (US) CPC ....... A61K 31/4174 (2013.01); A61K 31/41 78 (2013.01); A61K 31/4184 (2013.01); A61 K (21) Appl. No.: 15/179,764 3 1/496 (2013.01); A61K 31/167 (2013.01); (22) Filed: Jun. 10, 2016 A61K 31/4196 (2013.01); A61K 31/4152 (2013.01); A61K 45/06 (2013.01); C07K Related U.S. Application Data 16/2818 (2013.01); A61K 39/3955 (2013.01); (60) Provisional application No. 62/174,430, filed on Jun. A6 IK 2039/505 (2013.01) 11, 2015, provisional application No. 62/174,424, filed on Jun. 11, 2015, provisional application No. (57) ABSTRACT 62/188.413, filed on Jul. 2, 2015, provisional appli cation No. 62/195,750, filed on Jul. 22, 2015. Described are compositions and methods for treating cancer. Some methods comprise the administration of an effective Publication Classification amount of at least one inhibitor of tryptophan 2,3-dioxy (51) Int. -

III 1 J"I National Criminal Justice Reference Service '1 ! ,------II1
If you have issues viewing or accessing this file contact us at NCJRS.gov. ----- ---- - ----,- .. ----~-___r----------- -------------------------------------- n j 1 ; ! ~ ' .. III 1 j"i National Criminal Justice Reference Service '1 ! ,---------------------------------------------------- II1 \1 II nCJrs I I LOS ANGELES SUPERIOR COURT I} 11 .,1 r ; 1 This microfiche was produced from documents received for '[ t . !J inclusion in the NCJRS data base. Since NCJRS cannot exercise . 1 control over the physical condition of the documents submitted, , .~ the individual frame quality will vary. The resolution chart on i! this frame may be llsed to evaluate the document quality. Dru.g TerD1i:nQlogy Gen.era.l Glossa.ry 2 2 5 1.0 ~~ 11111 ,8 11111 . 3 B~ 11111 ,2 ENGLISH -SPANISH / SPANISH- ENGLISH R" .2 .;- ~1fI36 EI:.. lAO '- " III 1.1 LoL:.i.o. 111111.8 111111.25 111111.4 111111.6 MICROCOPY RESOLUTION TEST CHART NATIONAL BUREAU OF STANDARDS-1963-A Microfilming procedures used to create this fiche comply with RICHARD SCHAUER, Presiding Judge the standards set forth in 41CFR 101-11.504. FRANK S. ZOLIN, Executive Officer Points of view or opinions stated in this document are EDITORS those of the author(s) and do not represent the official Alexandre Rainof, Ph.D. position or policies of the U. S. Department of Justice. Alberto Natividad Date Filmed National Institute of Justice AUTHORS Alexandre Rainof, Ph.D United States Department of Justice 3/01/81 Washington, D. C. 20531 Alberto Natividad Joseph V. A. Partallsky Gilbert Geilim MJ.\RCH, 1980 ------ - --~ ---'.--------~~~---------- .' LOS ANGELES SUPERIOR COURT LOS ANGELES, CALIFORNIA 90012 FRANK S, ZOLlN, EXECUTIVE OFFICER TELEPHONE A DRUG TERMINOLOGY-GENERAL GLOSSARY (213) 974-5401 INTRODUCTION English-Spanish/Spanish-English with The growing number of drug and alcohol related cases Eight Special Sections entering the criminal justice system necessitated the development and of a comprehensive glossary of narcotic, drug and alcohol related A Selected and Annotated Bibliography terms.