Methyl Bromide
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Bromomethane CAS #: 74-83-9 Revised By: RRD Toxicology Unit Revision Date: August 14, 2015
CHEMICAL UPDATE WORKSHEET Chemical Name: Bromomethane CAS #: 74-83-9 Revised By: RRD Toxicology Unit Revision Date: August 14, 2015 (A) Chemical-Physical Properties Part 201 Value Updated Value Reference Source Comments Molecular Weight (g/mol) 94.94 94.94 EPI EXP Physical State at ambient temp Liquid Gas MDEQ Melting Point (˚C) 179 -93.70 EPI EXP Boiling Point (˚C) 3.5 3.50 EPI EXP Solubility (ug/L) 1.45E+7 1.52E+07 EPI EXP Vapor Pressure (mmHg at 25˚C) 1672 1.62E+03 EPI EXP HLC (atm-m³/mol at 25˚C) 1.42E-2 7.34E-03 EPI EXP Log Kow (log P; octanol-water) 1.18 1.19 EPI EXP Koc (organic carbon; L/Kg) 14.5 13.22 EPI EST Ionizing Koc (L/kg) NR NA NA Diffusivity in Air (Di; cm2/s) 0.08 1.00E-01 W9 EST Diffusivity in Water (Dw; cm2/s) 8.0E-6 1.3468E-05 W9 EST CHEMICAL UPDATE WORKSHEET Bromomethane (74-83-9) Part 201 Value Updated Value Reference Source Comments Soil Water Partition Coefficient NR NR NA NA (Kd; inorganics) Flash Point (˚C) NA 194 PC EXP Lower Explosivity Level (LEL; 0.1 0.1 CRC EXP unit less) Critical Temperature (K) 467.00 EPA2004 EXP Enthalpy of Vaporization 5.71E+03 EPA2004 EXP (cal/mol) Density (g/mL, g/cm3) 1.6755 CRC EXP EMSOFT Flux Residential 2 m 2.69E-05 2.80E-05 EMSOFT EST (mg/day/cm2) EMSOFT Flux Residential 5 m 6.53E-05 6.86E-05 EMSOFT EST (mg/day/cm2) EMSOFT Flux Nonresidential 2 m 3.83E-05 4.47E-05 EMSOFT EST (mg/day/cm2) EMSOFT Flux Nonresidential 5 m 9.24E-05 1.09E-04 EMSOFT EST (mg/day/cm2) 2 CHEMICAL UPDATE WORKSHEET Bromomethane (74-83-9) (B) Toxicity Values/Benchmarks Source/Reference/ Comments/Notes Part 201 Value Updated Value Date /Issues Reference Dose 1.4E-3 2.0E-2 OPP, 2013 (RfD) (mg/kg/day) Rat subchronic Tier 1 Source: Complete gavage study EPA-OPP: (Danse et al., Basis: OPP is the more current than IRIS, PPRTV and ATSDR. -

SAFETY DATA SHEET Bromomethane (R40 B1) SECTION 1
SAFETY DATA SHEET Bromomethane (R40 B1) Issue Date: 16.01.2013 Version: 1.0 SDS No.: 000010021848 Last revised date: 02.02.2017 1/17 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1 Product identifier Product name: Bromomethane (R40 B1) Additional identification Chemical name: Bromomethane Chemical formula: CH3Br INDEX No. 602-002-00-2 CAS-No. 74-83-9 EC No. 200-813-2 REACH Registration No. Not available. 1.2 Relevant identified uses of the substance or mixture and uses advised against Identified uses: Industrial and professional. Perform risk assessment prior to use. Using gas alone or in mixtures for the calibration of analysis equipment. Using gas as feedstock in chemical processes. Formulation of mixtures with gas in pressure receptacles. Uses advised against Consumer use. 1.3 Details of the supplier of the safety data sheet Supplier Linde Gas GmbH Telephone: +43 50 4273 Carl-von-Linde-Platz 1 A-4651 Stadl-Paura E-mail: [email protected] 1.4 Emergency telephone number: Emergency number Linde: + 43 50 4273 (during business hours), Poisoning Information Center: +43 1 406 43 43 SDS_AT - 000010021848 SAFETY DATA SHEET Bromomethane (R40 B1) Issue Date: 16.01.2013 Version: 1.0 SDS No.: 000010021848 Last revised date: 02.02.2017 2/17 SECTION 2: Hazards identification 2.1 Classification of the substance or mixture Classification according to Directive 67/548/EEC or 1999/45/EC as amended. T; R23/25 Xi; R36/37/38 Xn; R48/20 Muta. 3; R68 N; R50 N; R59 The full text for all R-phrases is displayed in section 16. -
The Following Studies Received Ethical Approval by Institutional And/Or National Review Committees If Appropriate
The following studies received ethical approval by institutional and/or national review committees if appropriate. Manchester AVA Spring Meeting April 2017 Cardiovascular findings during pre-anesthetic assessment in dogs undergoing two different pre-anesthetic assessment protocols and usefulness of the diagnostic tests performed S Diez-Bernal1, G Ortiz-Diez2, SP Monteagudo-Franco3, P Ruhi-Velasco4, C Garcia- Echarri4, A Perez-Higueras4, V Salazar-Nussio5. 1Anesthesia Service, University of Bern, Bern, Switzerland; 2Cardiology Service, Alfonso X El Sabio University, Madrid, Spain; 3Diagnostic Imaging Service, Alfonso X El Sabio University, Madrid, Spain; 4Alfonso X El Sabio University, Madrid, Spain; 5Anesthesia Service, Alfonso X El Sabio University, Madrid, Spain. The objectives of this study were to describe cardiovascular abnormalities (CVA) found during pre-anesthetic assessment in dogs in a veterinary teaching hospital and to evaluate the usefulness of the different diagnostic tests performed. One hundred and eight client-owned dogs underwent a basic pre-anesthetic assessment protocol including history, physical examination, hematology and biochemistry performed and evaluated by an anesthesia specialist; and a comprehensive pre-anesthetic assessment protocol that included, in addition to the tests performed in the basic assessment protocol, thoracic radiographs, ECG, and echocardiography evaluated by a licensed veterinarian. CVA were identified in twenty-five of ninety-seven dogs (25.8%) by cardiac auscultation; in 15 dogs (13.9%) by ECG; in 6 dogs (5.6%) by thoracic radiographs; and in 39 dogs (36.1%) by echocardiography (95% of which were diagnosed of chronic valvular heart disease, CVHD). Since CVHD was the cardiovascular condition that showed the highest prevalence, sensitivity, specificity and likelihood ratios of the other performed clinical tests were calculated for the diagnosis of this condition (taking echocardiography as gold standard). -

History Lectured on Midwifery at St Bartholomew’S Hospital and and Was in Attendance at the Births of All of Her Children
J R Coll Physicians Edinb 2012; 42:274–9 Paper http://dx.doi.org/10.4997/JRCPE.2012.317 © 2012 Royal College of Physicians of Edinburgh Sir Charles Locock and potassium bromide MJ Eadie Honorary Research Consultant and Emeritus Professor, Faculty of Health Sciences, University of Queensland, Royal Brisbane and Women’s Hospital, Australia ABSTRACT On 12 May 1857, Edward Sieveking read a paper on epilepsy to the Correspondence to M Eadie Royal Medical and Chirurgical Society in London. During the discussion that Faculty of Health Sciences, followed Sir Charles Locock, obstetrician to Queen Victoria, was reported to have University of Queensland, Royal Brisbane and Women’s commented that during the past 14 months he had used potassium bromide to Hospital, Herston, successfully stop epileptic seizures in all but one of 14 or 15 women with ‘hysterical’ Brisbane 4029, Australia or catamenial epilepsy. This report of Locock’s comment has generally given him credit for introducing the first reasonably effective antiepileptic drug into medical Tel 61 2 (0)7 38311704 e-mail [email protected] practice. However examination of the original reports raises questions as to how soundly based the accounts of Locock’s comments were. Subsequently, others using the drug to treat epilepsy failed to obtain the degree of benefit that the reports of Locock’s comments would have led them to expect. The drug might not have come into more widespread use as a result, had not Samuel Wilks provided good, independent evidence for the drug’s antiepileptic efficacy in 1861. KEYWORDS Epilepsy treatment, Charles Locock, potassium bromide, Edward Sieveking, Samuel Wilks DECLaratIONS OF INTERESTS No conflicts of interest declared. -

Toxicological Review of Chloral Hydrate (CAS No. 302-17-0) (PDF)
EPA/635/R-00/006 TOXICOLOGICAL REVIEW OF CHLORAL HYDRATE (CAS No. 302-17-0) In Support of Summary Information on the Integrated Risk Information System (IRIS) August 2000 U.S. Environmental Protection Agency Washington, DC DISCLAIMER This document has been reviewed in accordance with U.S. Environmental Protection Agency policy and approved for publication. Mention of trade names or commercial products does not constitute endorsement or recommendation for use. Note: This document may undergo revisions in the future. The most up-to-date version will be made available electronically via the IRIS Home Page at http://www.epa.gov/iris. ii CONTENTS—TOXICOLOGICAL REVIEW for CHLORAL HYDRATE (CAS No. 302-17-0) FOREWORD .................................................................v AUTHORS, CONTRIBUTORS, AND REVIEWERS ................................ vi 1. INTRODUCTION ..........................................................1 2. CHEMICAL AND PHYSICAL INFORMATION RELEVANT TO ASSESSMENTS ..... 2 3. TOXICOKINETICS RELEVANT TO ASSESSMENTS ............................3 4. HAZARD IDENTIFICATION ................................................6 4.1. STUDIES IN HUMANS - EPIDEMIOLOGY AND CASE REPORTS .................................................6 4.2. PRECHRONIC AND CHRONIC STUDIES AND CANCER BIOASSAYS IN ANIMALS ................................8 4.2.1. Oral ..........................................................8 4.2.2. Inhalation .....................................................12 4.3. REPRODUCTIVE/DEVELOPMENTAL STUDIES ..........................13 -
RR Program's RCL Spreadsheet Update
RR Program’s RCL Spreadsheet Update March 2017 RR Program RCL Spreadsheet Update DNR-RR-052e The Wisconsin DNR Remediation and Redevelopment Program (RR) has updated the numerical soil standards in the August 2015 DNR-RR- 052b RR spreadsheet of residual contaminant levels (RCLs). The RCLs were determined using the U.S. EPA RSL web- calculator by accepting EPA exposure defaults, with the exception of using Chicago, IL, for the climatic zone. This documentThe U.S. provides EPA updateda summary its Regionalof changes Screening to the direct-contact Level (RSL) RCLs website (DC-RCLs) in June that2015. are To now reflect in the that March 2017 spreadsheet.update, the The Wisconsin last page ofDNR this updated document the has numerical the EPA exposuresoil standards, parameter or residual values usedcontaminant in the RCL levels calculations. (RCLs), in the Remediation and Redevelopment program’s spreadsheet of RCLs. This document The providesU.S. EPA a RSL summary web-calculator of the updates has been incorporated recently updated in the Julyso that 2015 the spreadsheet.most up-to-date There toxicity were values no changes for chemi - cals madewere certainlyto the groundwater used in the RCLs,RCL calculations. but there are However, many changes it is important in the industrial to note that and the non-industrial web-calculator direct is only a subpartcontact of the (DC) full RCLsEPA RSL worksheets. webpage, Tables and that 1 andthe other 2 of thissubparts document that will summarize have important the DC-RCL explanatory changes text, generic tablesfrom and the references previous have spreadsheet yet to be (Januaryupdated. -

A High Volume Sampling System for Isotope Determination of Volatile Halocarbons and Hydrocarbons
Atmos. Meas. Tech., 4, 2073–2086, 2011 www.atmos-meas-tech.net/4/2073/2011/ Atmospheric doi:10.5194/amt-4-2073-2011 Measurement © Author(s) 2011. CC Attribution 3.0 License. Techniques A high volume sampling system for isotope determination of volatile halocarbons and hydrocarbons E. Bahlmann, I. Weinberg, R. Seifert, C. Tubbesing, and W. Michaelis Institute for Biogeochemistry and Marine Chemistry, Hamburg, Germany Received: 15 March 2011 – Published in Atmos. Meas. Tech. Discuss.: 8 April 2011 Revised: 31 August 2011 – Accepted: 7 September 2011 – Published: 4 October 2011 Abstract. The isotopic composition of volatile organic com- 1 Introduction pounds (VOCs) can provide valuable information on their sources and fate not deducible from mixing ratios alone. Compound specific isotope ratio mass spectrometry In particular the reported carbon stable isotope ratios of (CSIRMS) of non methane volatile organic compounds chloromethane and bromomethane from different sources (NMVOCs) emerged as a powerful tool to distinguish dif- 13 cover a δ C-range of almost 100 ‰ making isotope ra- ferent sources and to provide information on sinks (Rudolph tios a very promising tool for studying the biogeochemistry et al., 1997; Rudolph and Czuba, 2000; Tsunogai et al., of these compounds. So far, the determination of the iso- 1999; Bill et al., 2002; Thompson et al., 2002; Goldstein and topic composition of C1 and C2 halocarbons others than Shaw, 2003 and references therein; Archbold et al., 2005; chloromethane is hampered by their low mixing ratios. Redeker -

Canine Status Epilepticus Care
Vet Times The website for the veterinary profession https://www.vettimes.co.uk CANINE STATUS EPILEPTICUS CARE Author : Stefano Cortellini, Luisa de Risio Categories : Vets Date : August 2, 2010 Stefano Cortellini and Luisa de Risio discuss emergency management techniques for a condition that can claim the lives of 25 per cent of afflicted dogs – as the quicker the start of treatment, the better the chances of control STATUS epilepticus (SE) is a neurological emergency with a mortality of up to 25 per cent in dogs (Bateman, 1999). SE can be defined as continuous epileptic seizure (ES) activity lasting longer than five minutes, or as two or more ES with incomplete recovery of consciousness interictally. SE has also been defined as continuous seizure activity lasting for 30 minutes or longer. However, emergency treatment to stop the ES should be administered well before the defined 30-minute time. The most common type of SE is generalised tonic-clonic status. When this is prolonged, the tonic- clonic clinical manifestations can become subtle, with only small muscle twitching and altered mentation. This status is called electromechanical dissociation, as continued abnormal electrical activity in the brain persists while the motor manifestations are minimal to absent. In these cases, emergency anti-epileptic treatment is necessary as for tonic-clonic status. SE can be divided into two stages. The first stage is characterised by generalised tonicclonic seizures and an increase in autonomic activity that causes tachycardia, hypertension, 1 / 7 hyperglycaemia, hyperthermia and increased cerebral blood flow. The second stage of SE starts after about 30 minutes and is characterised by hypotension, hypoglycaemia, hyperthermia, hypoxia, decreased cerebral blood flow, cerebral oedema and increased intracranial pressure. -
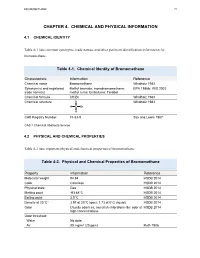
Toxicological Profile for Bromomethane
BROMOMETHANE 72 CHAPTER 4. CHEMICAL AND PHYSICAL INFORMATION 4.1 CHEMICAL IDENTITY Table 4-1 lists common synonyms, trade names, and other pertinent identification information for bromomethane. Table 4-1. Chemical Identity of Bromomethane Characteristic Information Reference Chemical name Bromomethane Windholz 1983 Synonym(s) and registered Methyl bromide; monobromomethane; EPA 1986b; IRIS 2002 trade name(s) methyl fume; Embafume; Terabol Chemical formula CH3Br Windholz 1983 Chemical structure H Windholz 1983 H C Br H CAS Registry Number 74-83-9 Sax and Lewis 1987 CAS = Chemical Abstracts Service 4.2 PHYSICAL AND CHEMICAL PROPERTIES Table 4-2 lists important physical and chemical properties of bromomethane. Table 4-2. Physical and Chemical Properties of Bromomethane Property Information Reference Molecular weight 94.94 HSDB 2014 Color Colorless HSDB 2014 Physical state Gas HSDB 2014 Melting point -93.68°C HSDB 2014 Boiling point 3.5°C HSDB 2014 Density at 20°Ca 3.97 at 20°C (gas); 1.73 at 0°C (liquid) HSDB 2014 Odor Usually odorless; sweetish chloroform-like odor at HSDB 2014 high concentrations Odor threshold: Water No data Air 80 mg/m3 (20 ppm) Ruth 1986 BROMOMETHANE 73 4. CHEMICAL AND PHYSICAL INFORMATION Table 4-2. Physical and Chemical Properties of Bromomethane Solubility: Water at 20°C 15.2 g/L at 25°C HSDB 2014 18.5 g/L at 20°C 13.4 g/L at 25°C Organic solvents Readily soluble in lower alcohols, ethers, esters, HSDB 2014 ketones, halogenated hydrocarbons, aromatic hydrocarbons, and carbon disulfide; freely soluble in benzene, carbon tetrachloride, and carbon disulfide; miscible in ethanol and chloroform Partition coefficients: Log Kow 1.19 HSDB 2014 Log Koc 0.95–1.3 Yates et al. -

Chloral Hydrate
NTP TECHNICAL REPORT ON THE TOXICOLOGY AND CARCINOGENESIS STUDY OF CHLORAL HYDRATE (AD LIBITUM AND DIETARY CONTROLLED) (CAS NO. 302-17-0) IN MALE B6C3F1 MICE (GAVAGE STUDY) NATIONAL TOXICOLOGY PROGRAM P.O. Box 12233 Research Triangle Park, NC 27709 December 2002 NTP TR 503 NIH Publication No. 03-4437 U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Public Health Service National Institutes of Health FOREWORD The National Toxicology Program (NTP) is made up of four charter agencies of the U.S. Department of Health and Human Services (DHHS): the National Cancer Institute (NCI), National Institutes of Health; the National Institute of Environmental Health Sciences (NIEHS), National Institutes of Health; the National Center for Toxicological Research (NCTR), Food and Drug Administration; and the National Institute for Occupational Safety and Health (NIOSH), Centers for Disease Control and Prevention. In July 1981, the Carcinogenesis Bioassay Testing Program, NCI, was transferred to the NIEHS. The NTP coordinates the relevant programs, staff, and resources from these Public Health Service agencies relating to basic and applied research and to biological assay development and validation. The NTP develops, evaluates, and disseminates scientific information about potentially toxic and hazardous chemicals. This knowledge is used for protecting the health of the American people and for the primary prevention of disease. The studies described in this Technical Report were performed under the direction of the NCTR and were conducted in compliance with NTP laboratory health and safety requirements and must meet or exceed all applicable federal, state, and local health and safety regulations. Animal care and use were in accordance with the Public Health Service Policy on Humane Care and Use of Animals. -

Removal of Carbamazepine Onto Modified Zeolitic Tuff In
water Article Removal of Carbamazepine onto Modified Zeolitic Tuff in Different Water Matrices: Batch and Continuous Flow Experiments Othman A. Al-Mashaqbeh 1,* , Diya A. Alsafadi 2, Layal Z. Alsalhi 1, Shannon L. Bartelt-Hunt 3 and Daniel D. Snow 4 1 Emerging Pollutants Research Unit, Royal Scientific Society, Amman 11941, Jordan; [email protected] 2 Biocatalysis and Biosynthesis Research Unit, Royal Scientific Society, Amman 11941, Jordan; [email protected] 3 College of Engineering, University of Nebraska–Lincoln, Omaha, NE 68583, USA; [email protected] 4 Water Sciences Laboratory, University of Nebraska, Lincoln, NE 68583, USA; [email protected] * Correspondence: [email protected] Abstract: Carbamazepine (CBZ) is the most frequently detected pharmaceutical residues in aquatic environments effluent by wastewater treatment plants. Batch and column experiments were con- ducted to evaluate the removal of CBZ from ultra-pure water and wastewater treatment plant (WWTP) effluent using raw zeolitic tuff (RZT) and surfactant modified zeolite (SMZ). Point zero net charge (pHpzc), X-ray diffraction (XRD), X-ray fluorescence (XRF), and Fourier Transform Infrared (FTIR) were investigated for adsorbents to evaluate the physiochemical changes resulted from the modification process using Hexadecyltrimethylammonium bromide (HDTMA-Br). XRD and FTIR Citation: Al-Mashaqbeh, O.A.; showed that the surfactant modification of RZT has created an amorphous surface with new alkyl Alsafadi, D.A.; Alsalhi, L.Z.; groups on the surface. The pHpzc was determined to be approximately 7.9 for RZT and SMZ. The Bartelt-Hunt, S.L.; Snow, D.D. results indicated that the CBZ uptake by SMZ is higher than RZT in all sorption tests (>8 fold). -

Federal Register/Vol. 84, No. 230/Friday, November 29, 2019
Federal Register / Vol. 84, No. 230 / Friday, November 29, 2019 / Proposed Rules 65739 are operated by a government LIBRARY OF CONGRESS 49966 (Sept. 24, 2019). The Office overseeing a population below 50,000. solicited public comments on a broad Of the impacts we estimate accruing U.S. Copyright Office range of subjects concerning the to grantees or eligible entities, all are administration of the new blanket voluntary and related mostly to an 37 CFR Part 210 compulsory license for digital uses of increase in the number of applications [Docket No. 2019–5] musical works that was created by the prepared and submitted annually for MMA, including regulations regarding competitive grant competitions. Music Modernization Act Implementing notices of license, notices of nonblanket Therefore, we do not believe that the Regulations for the Blanket License for activity, usage reports and adjustments, proposed priorities would significantly Digital Uses and Mechanical Licensing information to be included in the impact small entities beyond the Collective: Extension of Comment mechanical licensing collective’s potential for increasing the likelihood of Period database, database usability, their applying for, and receiving, interoperability, and usage restrictions, competitive grants from the Department. AGENCY: U.S. Copyright Office, Library and the handling of confidential of Congress. information. Paperwork Reduction Act ACTION: Notification of inquiry; To ensure that members of the public The proposed priorities do not extension of comment period. have sufficient time to respond, and to contain any information collection ensure that the Office has the benefit of SUMMARY: The U.S. Copyright Office is requirements. a complete record, the Office is extending the deadline for the extending the deadline for the Intergovernmental Review: This submission of written reply comments program is subject to Executive Order submission of written reply comments in response to its September 24, 2019 to no later than 5:00 p.m.