Cd4/Cd8 Panel, Blood
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Role of IL-4 Receptor &Alpha
Role of IL-4 receptor a–positive CD41 T cells in chronic airway hyperresponsiveness Frank Kirstein, PhD, Natalie E. Nieuwenhuizen, PhD,* Jaisubash Jayakumar, PhD, William G. C. Horsnell, PhD, and Frank Brombacher, PhD Cape Town, South Africa, and Berlin, Germany Background: TH2 cells and their cytokines are associated with IL-17–producing T cells and, consequently, increased airway allergic asthma in human subjects and with mouse models of neutrophilia. allergic airway disease. IL-4 signaling through the IL-4 receptor Conclusion: IL-4–responsive T helper cells are dispensable for 1 a (IL-4Ra) chain on CD4 T cells leads to TH2 cell acute OVA-induced airway disease but crucial in maintaining differentiation in vitro, implying that IL-4Ra–responsive CD41 chronic asthmatic pathology. (J Allergy Clin Immunol T cells are critical for the induction of allergic asthma. However, 2016;137:1852-62.) mechanisms regulating acute and chronic allergen-specific T 2 H Key words: responses in vivo remain incompletely understood. TH2 cell, acute allergic airway disease, chronic asthma, Objective: This study defines the requirements for IL-4Ra– cytokine receptors, IL-4, IL-13, gene-deficient mice responsive CD41 T cells and the IL-4Ra ligands IL-4 and IL-13 in the development of allergen-specific TH2 responses during the Allergic asthma is a chronic inflammatory disease of the airways onset and chronic phase of experimental allergic airway disease. characterized by an inappropriate immune response to harmless Methods: Development of acute and chronic ovalbumin environmental antigens. T 2 cells regulate adaptive immune 1 H (OVA)–induced allergic asthma was assessed weekly in CD4 T responses to allergens, and their presence correlates with disease 2 cell–specific IL-4Ra–deficient BALB/c mice (LckcreIL-4Ra /lox) symptoms in human subjects and mice.1 IL-4 plays a crucial role and respective control mice in the presence or absence of IL-4 in the in vitro and in vivo differentiation of TH2 cells, suggesting or IL-13. -
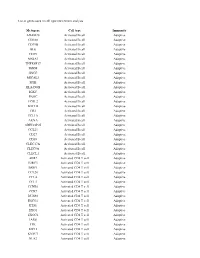
List of Genes Used in Cell Type Enrichment Analysis
List of genes used in cell type enrichment analysis Metagene Cell type Immunity ADAM28 Activated B cell Adaptive CD180 Activated B cell Adaptive CD79B Activated B cell Adaptive BLK Activated B cell Adaptive CD19 Activated B cell Adaptive MS4A1 Activated B cell Adaptive TNFRSF17 Activated B cell Adaptive IGHM Activated B cell Adaptive GNG7 Activated B cell Adaptive MICAL3 Activated B cell Adaptive SPIB Activated B cell Adaptive HLA-DOB Activated B cell Adaptive IGKC Activated B cell Adaptive PNOC Activated B cell Adaptive FCRL2 Activated B cell Adaptive BACH2 Activated B cell Adaptive CR2 Activated B cell Adaptive TCL1A Activated B cell Adaptive AKNA Activated B cell Adaptive ARHGAP25 Activated B cell Adaptive CCL21 Activated B cell Adaptive CD27 Activated B cell Adaptive CD38 Activated B cell Adaptive CLEC17A Activated B cell Adaptive CLEC9A Activated B cell Adaptive CLECL1 Activated B cell Adaptive AIM2 Activated CD4 T cell Adaptive BIRC3 Activated CD4 T cell Adaptive BRIP1 Activated CD4 T cell Adaptive CCL20 Activated CD4 T cell Adaptive CCL4 Activated CD4 T cell Adaptive CCL5 Activated CD4 T cell Adaptive CCNB1 Activated CD4 T cell Adaptive CCR7 Activated CD4 T cell Adaptive DUSP2 Activated CD4 T cell Adaptive ESCO2 Activated CD4 T cell Adaptive ETS1 Activated CD4 T cell Adaptive EXO1 Activated CD4 T cell Adaptive EXOC6 Activated CD4 T cell Adaptive IARS Activated CD4 T cell Adaptive ITK Activated CD4 T cell Adaptive KIF11 Activated CD4 T cell Adaptive KNTC1 Activated CD4 T cell Adaptive NUF2 Activated CD4 T cell Adaptive PRC1 Activated -

T Cells the Usual Subsets
T cells: the usual subsets Chen Dong and Gustavo J. Martinez T cells have important roles in immune responses and function by directly secreting soluble mediators or important for adaptation of immune responses in different microenvironments and might be particularly through cell contact-dependent mechanisms. Many T cell subsets have been characterized. Although relevant for host defence against pathogens that colonize different tissues. Distinct T cell subsets, or effector T cells were originally considered to be terminally differentiated, a growing body of evidence has differentiation states, can be identified based on the cell surface markers expressed and/or the effector challenged this view and suggested that the phenotype of effector T cells is not completely fixed but is molecules produced by a particular T cell population. This Poster summarizes our current understanding of more flexible or plastic. T cells can have ‘mixed’ phenotypes (that is, have characteristics usually the surface markers, transcriptional regulators, effector molecules and functions of the different T cell associated with more than one T cell subset) and can interconvert from one subset phenotype to another, subsets that participate in immune responses. Further knowledge of how these T cell subsets are regulated IMMUNOLOGY although instructive signalling can lead to long-term fixation of cytokine memory. T cell plasticity can be and cooperate with each other will provide us with better tools to treat immune-related diseases. Cytotoxic T cell Exhausted T cell -

T Lymphocytes + and CD8 +CD4 TCR/CD3 Complex in Immortalized Mature -Deficient Γ Signaling Through A
Signaling Through a CD3γ-Deficient TCR/CD3 Complex in Immortalized Mature CD4+ and CD8+ T Lymphocytes This information is current as Alberto Pacheco-Castro, David Alvarez-Zapata, Pilar of September 25, 2021. Serrano-Torres and José R. Regueiro J Immunol 1998; 161:3152-3160; ; http://www.jimmunol.org/content/161/6/3152 Downloaded from References This article cites 47 articles, 19 of which you can access for free at: http://www.jimmunol.org/content/161/6/3152.full#ref-list-1 Why The JI? Submit online. http://www.jimmunol.org/ • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average by guest on September 25, 2021 Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 1998 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. Signaling Through a CD3g-Deficient TCR/CD3 Complex in Immortalized Mature CD41 and CD81 T Lymphocytes1 Alberto Pacheco-Castro,2 David Alvarez-Zapata,2 Pilar Serrano-Torres, and Jose´R. Regueiro3 The biologic role of each CD3 chain and their relative contribution to the signals transduced through the TCR/CD3 complex and to downstream activation events are still controversial: they may be specialized or redundant. -

18F-Farag PET for CD8 Profiling of Tumors and Assessment of Immunomodulation by Chemotherapy
Journal of Nuclear Medicine, published on November 6, 2020 as doi:10.2967/jnumed.120.249078 18F-FAraG PET for CD8 Profiling of Tumors and Assessment of Immunomodulation by Chemotherapy Jelena Levi1*, Samuel Goth1, Lyna Huynh1, Tina Lam1, Tony L Huynh2, Brailee Schulte2, Juliet A Packiasamy1 1CellSight Technologies Incorporated, San Francisco, California; 2Department of Radiology and Biomedical Imaging, University of California, San Francisco, San Francisco, California; *Corresponding Author Jelena Levi, PhD 185 Berry street, San Francisco, 94107, CA Email: [email protected] Running Title: [18F]F-AraG for CD8 profiling of tumors Financial statement: This work was supported by National Institutes of Health Grants NCI SBIR HHSN261201800024C (JL). Conflict of Interest Statement: JL, SG, LH, TL and JP are or were employed by CellSight Technologies. CellSight Technologies Incorporated is commercializing [18F]F-AraG as a PET tracer for evaluation of immune response in immunotherapy. No other potential conflicts of interest relevant to this article exist. ABSTRACT Majority of the clinical trials exploring various combinations of chemo- and immunotherapy rely on serial biopsy to provide information on immune response. The aim of this study was to assess the value of 18F-FAraG as a non-invasive tool that could profile tumors based on the key players in adaptive antitumor response, CD8+ cells, and evaluate immunomodulatory effects of chemotherapy. Methods. To evaluate the ability of 18F-FAraG to report on the presence of CD8+ cells within the TME, we imaged a panel of syngeneic tumor models (MC38, CT26, LLC, A9F1, 4T1 and B16F10), and correlated the signal intensity with the number of lymphocytes found in the tumors. -

Single-Cell RNA Sequencing Demonstrates the Molecular and Cellular Reprogramming of Metastatic Lung Adenocarcinoma
ARTICLE https://doi.org/10.1038/s41467-020-16164-1 OPEN Single-cell RNA sequencing demonstrates the molecular and cellular reprogramming of metastatic lung adenocarcinoma Nayoung Kim 1,2,3,13, Hong Kwan Kim4,13, Kyungjong Lee 5,13, Yourae Hong 1,6, Jong Ho Cho4, Jung Won Choi7, Jung-Il Lee7, Yeon-Lim Suh8,BoMiKu9, Hye Hyeon Eum 1,2,3, Soyean Choi 1, Yoon-La Choi6,10,11, Je-Gun Joung1, Woong-Yang Park 1,2,6, Hyun Ae Jung12, Jong-Mu Sun12, Se-Hoon Lee12, ✉ ✉ Jin Seok Ahn12, Keunchil Park12, Myung-Ju Ahn 12 & Hae-Ock Lee 1,2,3,6 1234567890():,; Advanced metastatic cancer poses utmost clinical challenges and may present molecular and cellular features distinct from an early-stage cancer. Herein, we present single-cell tran- scriptome profiling of metastatic lung adenocarcinoma, the most prevalent histological lung cancer type diagnosed at stage IV in over 40% of all cases. From 208,506 cells populating the normal tissues or early to metastatic stage cancer in 44 patients, we identify a cancer cell subtype deviating from the normal differentiation trajectory and dominating the metastatic stage. In all stages, the stromal and immune cell dynamics reveal ontological and functional changes that create a pro-tumoral and immunosuppressive microenvironment. Normal resident myeloid cell populations are gradually replaced with monocyte-derived macrophages and dendritic cells, along with T-cell exhaustion. This extensive single-cell analysis enhances our understanding of molecular and cellular dynamics in metastatic lung cancer and reveals potential diagnostic and therapeutic targets in cancer-microenvironment interactions. 1 Samsung Genome Institute, Samsung Medical Center, Seoul 06351, Korea. -

B Cell Checkpoints in Autoimmune Rheumatic Diseases
REVIEWS B cell checkpoints in autoimmune rheumatic diseases Samuel J. S. Rubin1,2,3, Michelle S. Bloom1,2,3 and William H. Robinson1,2,3* Abstract | B cells have important functions in the pathogenesis of autoimmune diseases, including autoimmune rheumatic diseases. In addition to producing autoantibodies, B cells contribute to autoimmunity by serving as professional antigen- presenting cells (APCs), producing cytokines, and through additional mechanisms. B cell activation and effector functions are regulated by immune checkpoints, including both activating and inhibitory checkpoint receptors that contribute to the regulation of B cell tolerance, activation, antigen presentation, T cell help, class switching, antibody production and cytokine production. The various activating checkpoint receptors include B cell activating receptors that engage with cognate receptors on T cells or other cells, as well as Toll-like receptors that can provide dual stimulation to B cells via co- engagement with the B cell receptor. Furthermore, various inhibitory checkpoint receptors, including B cell inhibitory receptors, have important functions in regulating B cell development, activation and effector functions. Therapeutically targeting B cell checkpoints represents a promising strategy for the treatment of a variety of autoimmune rheumatic diseases. Antibody- dependent B cells are multifunctional lymphocytes that contribute that serve as precursors to and thereby give rise to acti- cell- mediated cytotoxicity to the pathogenesis of autoimmune diseases -

CD29 Identifies IFN-Γ–Producing Human CD8+ T Cells With
+ CD29 identifies IFN-γ–producing human CD8 T cells with an increased cytotoxic potential Benoît P. Nicoleta,b, Aurélie Guislaina,b, Floris P. J. van Alphenc, Raquel Gomez-Eerlandd, Ton N. M. Schumacherd, Maartje van den Biggelaarc,e, and Monika C. Wolkersa,b,1 aDepartment of Hematopoiesis, Sanquin Research, 1066 CX Amsterdam, The Netherlands; bLandsteiner Laboratory, Oncode Institute, Amsterdam University Medical Center, University of Amsterdam, 1105 AZ Amsterdam, The Netherlands; cDepartment of Research Facilities, Sanquin Research, 1066 CX Amsterdam, The Netherlands; dDivision of Molecular Oncology and Immunology, Oncode Institute, The Netherlands Cancer Institute, 1066 CX Amsterdam, The Netherlands; and eDepartment of Molecular and Cellular Haemostasis, Sanquin Research, 1066 CX Amsterdam, The Netherlands Edited by Anjana Rao, La Jolla Institute for Allergy and Immunology, La Jolla, CA, and approved February 12, 2020 (received for review August 12, 2019) Cytotoxic CD8+ T cells can effectively kill target cells by producing therefore developed a protocol that allowed for efficient iso- cytokines, chemokines, and granzymes. Expression of these effector lation of RNA and protein from fluorescence-activated cell molecules is however highly divergent, and tools that identify and sorting (FACS)-sorted fixed T cells after intracellular cytokine + preselect CD8 T cells with a cytotoxic expression profile are lacking. staining. With this top-down approach, we performed an un- + Human CD8 T cells can be divided into IFN-γ– and IL-2–producing biased RNA-sequencing (RNA-seq) and mass spectrometry cells. Unbiased transcriptomics and proteomics analysis on cytokine- γ– – + + (MS) analyses on IFN- and IL-2 producing primary human producing fixed CD8 T cells revealed that IL-2 cells produce helper + + + CD8 Tcells. -

Identification of 18 Immune Cell Subsets Using 13-Color Panel
Immunophenotyping Identification of 18 immune cell subsets in human blood using a 13-color panel Background Cell type Function Phenotype Flow cytometry has become the method of choice for Eosinophils Parasitic immunity CD45+, SSCmid/hi, CD14 –, CD16 –, CD19– immunophenotyping and identifying specific cellular + mid/hi subsets. Within seconds, it provides a thorough overview of Neutrophils Innate Immunity CD45 , SSC , CD14 –, CD16+, CD19– the major cell types that constitute a sample. Using multiple + mid markers simultaneously increases the number of parameters Classical Phagocytosis of CD45 , SSC , monocytes pathogens and CD14+, CD16– that can be analyzed per run and decreases the amount of antigen presentation starting material required to perform an assay. This can be Intermediate Phagocytosis of CD45+, SSCmid, critical for precious sample material and long-term immune- monocytes pathogens and CD14+, CD16mid monitoring studies. In this application note, we demonstrate antigen presentation 13-color immunophenotyping of human peripheral blood Non-classical Phagocytosis of CD45+, SSCmid, + + mononuclear cells (PBMCs) using the MACSQuant® Analyzer 16, monocytes pathogens and CD14 , CD16 antigen presentation a compact and reliable benchtop flow cytometer equipped + low + with three lasers. The markers selected allow for the Class-switched Adaptive immunity CD45 , SSC , CD19 , memory B cells CD27+, IgD–, CD14– simultaneous identification and analysis of 18 different cell Non-switched Adaptive immunity CD45+, SSClow, CD19+, populations, thus maximizing the amount of information that memory B cells CD27+, IgD+, CD14– can be retrieved from the sample material analyzed. This is Naive B cells Adaptive immunity – CD45+, SSClow, CD19+, critical when input material is limited, as is often the case for non-antigen CD27–, IgD+, CD14– pediatric or disease studies. -

Rational Design of a CD4 Mimic That Inhibits HIV-1 Entry and Exposes Cryptic Neutralization Epitopes
RESEARCH ARTICLE Rational design of a CD4 mimic that inhibits HIV-1 entry and exposes cryptic neutralization epitopes Loïc Martin1, François Stricher1, Dorothée Missé2, Francesca Sironi3, Martine Pugnière4, Philippe Barthe5, Rafael Prado-Gotor5, Isabelle Freulon1, Xavier Magne2, Christian Roumestand5, André Ménez1, Paolo Lusso3, Francisco Veas2, and Claudio Vita1* Published online 16 December 2002; doi:10.1038/nbt768 The conserved surfaces of the human immunodeficiency virus (HIV)-1 envelope involved in receptor binding represent potential targets for the development of entry inhibitors and neutralizing antibodies. Using structural information on a CD4-gp120-17b antibody complex, we have designed a 27-amino acid CD4 mimic, CD4M33, that presents optimal interactions with gp120 and binds to viral particles and diverse HIV-1 envelopes with CD4-like affinity.This mini-CD4 inhibits infection of both immortalized and primary cells by HIV-1, including pri- http://www.nature.com/naturebiotechnology mary patient isolates that are generally resistant to inhibition by soluble CD4. Furthermore, CD4M33 pos- sesses functional properties of CD4, including the ability to unmask conserved neutralization epitopes of gp120 that are cryptic on the unbound glycoprotein. CD4M33 is a prototype of inhibitors of HIV-1 entry and, in complex with envelope proteins, a potential component of vaccine formulations, or a molecular target in phage display technology to develop broad-spectrum neutralizing antibodies. HIV-1 avoids the host immune surveillance by exposing -

Flow Cytometry CPT: 88182, 88184, 88185, 88187, 88188, 88189, 86355, 86356, 86357, 86359, 86360, 86361, 86367
Medicare Local Coverage Determination Policy Flow Cytometry CPT: 88182, 88184, 88185, 88187, 88188, 88189, 86355, 86356, 86357, 86359, 86360, 86361, 86367 CMS Policy for Alaska, Arizona, Idaho, Montana, North Dakota, Medically Supportive Oregon, South Dakota, Utah, Washington, and Wyoming ICD Codes are listed Local policies are determined by the performing test location. This is determined by the state on subsequent page(s) in which your performing laboratory resides and where your testing is commonly performed. of this document. Coverage Indications, Limitations, and/or Medical Necessity Flow cytometry (FCM) is a complex process to examine blood, body fluids, CSF, bone marrow, lymph node, tonsil, spleen and other solid tissues. The use of peripheral blood and fine needle aspirate material avoids more invasive procedures for diagnosis. A flow cytometer evaluates the physical and/or chemical characteristics of single cells as the cells pass individually in a fluid stream through a measuring device. Surface receptors, intracellular molecules, and DNA bind with fluorescent dyes that allow detection and evaluation. When light of one wave length excites electrons of certain chemicals to energy levels above their ground state and upon return to ground state emits light of a longer wavelength, fluorescence is produced. A flow cytometer detects cell characteristics by measuring the fluorescence produced by fluorochromes conjugated either directly with cell components or conjugated to antibodies directed against cell components. Indications • Cytopenias and Hypercellular Hematolymphoid Disorders Hematolymphoid neoplasia can present with cytopenias (anemia, leucopenia and/or thrombocytopenia) or elevated leukocyte counts. If medical review and preliminary laboratory testing fails to reveal a cause, bone marrow aspiration and biopsy are indicated to rule out an infiltrative process or a stem cell disorder. -

Tethering IL2 to Its Receptor Il2rb Enhances Antitumor Activity and Expansion of Natural Killer NK92 Cells Youssef Jounaidi, Joseph F
Published OnlineFirst September 15, 2017; DOI: 10.1158/0008-5472.CAN-17-1007 Cancer Therapeutics, Targets, and Chemical Biology Research Tethering IL2 to Its Receptor IL2Rb Enhances Antitumor Activity and Expansion of Natural Killer NK92 Cells Youssef Jounaidi, Joseph F. Cotten, Keith W. Miller, and Stuart A. Forman Abstract IL2 is an immunostimulatory cytokine for key immune cells of IL2 and its receptor IL2Rb joined via a peptide linker (CIRB). including T cells and natural killer (NK) cells. Systemic IL2 NK92 cells expressing CIRB (NK92CIRB) were highly activated and supplementation could enhance NK-mediated immunity in a expanded indefinitely without exogenous IL2. When compared variety of diseases ranging from neoplasms to viral infection. with an IL2-secreting NK92 cell line, NK92CIRB were more acti- However, its systemic use is restricted by its serious side effects and vated, cytotoxic, and resistant to growth inhibition. Direct contact limited efficacy due to activation of T regulatory cells (Tregs). IL2 with cancer cells enhanced the cytotoxic character of NK92CIRB signaling is mediated through interactions with a multi-subunit cells, which displayed superior in vivo antitumor effects in mice. receptor complex containing IL2Ra, IL2Rb, and IL2Rg. Adult Overall, our results showed how tethering IL2 to its receptor natural killer (NK) cells express only IL2Rb and IL2Rg subunits IL2Rb eliminates the need for IL2Ra and IL2Rb, offering a and are therefore relatively insensitive to IL2. To overcome these new tool to selectively activate and empower immune therapy. limitations, we created a novel chimeric IL2-IL2Rb fusion protein Cancer Res; 77(21); 5938–51. Ó2017 AACR. Introduction in order for allogeneic NK cells to be effective, pretransfer lymphodepletion is required to reduce competition for growth Natural killer (NK) cells are lymphocytes endowed with the factors and cytokines (14, 15).