UNIVERSITY of CALIFORNIA RIVERSIDE a Screening-Level
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Role and Fate of Added Phosphates in Salted Cod Products
The role and fate of added phosphates in salted cod products Kristín Anna Þórarinsdóttir Sigurjón Arason Guðjón Þorkelsson Nýsköpun og neytendur Skýrsla Matís 27-10 Júlí 2010 ISSN 1670-7192 Titill / Title The role and fate of added phosphates in salted cod products / Hlutverk og afdrif viðbætts fosfats í saltfiski Höfundar / Authors Kristín Anna Þórarinsdóttir, Sigurjón Arason, Guðjón Þorkelsson Skýrsla / Report no. 27‐10 Útgáfudagur / Date: Júlí 2010 Verknr. / project no. 1008‐1934, 1008‐1935 Styrktaraðilar / funding: AGS, AVS Ágrip á íslensku: Markmið verkefnisins var að meta afdrif viðbætts fosfats í saltfiski. Ljóst er að magn þess lækkar við verkun og útvötnun. Sama gildir um fosföt sem eru náttúrulega til staðar í fiskvöðva. Þess vegna er heildarmagn fosfats í útvötnuðum afurðum yfirleitt lægra en í ferskum fiski. Hins vegar hefur verið sýnt fram á að viðbætt fosföt (dí‐ og trífosföt) finnast bæði í verkuðum og útvötnum fiski. Það er þó háð magni viðbætts fosfats í afurðinni og hvaða söltunarferlum er beitt, þ.e. hvort fosfati var bætt í fiskinn með sprautun eða pæklun. Lítið eða ekkert greinist í útvötnuðum afurðum ef pæklun er beitt. Munur á milli ferla getur stafað af söltunaraðferð (sprautun/pæklun), gerð og upphaflegu magni viðbætts fosfats og verkunartíma. Frekari rannsókna er þörf til að meta áhrif af mismunandi söltunarferlum á afdrif fosfats í söltuðum þorskvöðva. Lykilorð á íslensku: Saltfiskur, viðbætt fosfat, niðurbrot, verkunarferlar, sprautun, pæklun Summary in English: The aim of this study was to investigate the fate of added phosphates in salted cod products. The content of both added phosphates and naturally occurring phosphates, decreases during salting and rehydration. -

Indene CH Activation, Indole Π Vs. Nitrogen Lone-Pair Coordinati
HHS Public Access Author manuscript Author ManuscriptAuthor Manuscript Author Organometallics Manuscript Author . Author Manuscript Author manuscript; available in PMC 2016 April 15. Published in final edited form as: Organometallics. 2007 ; 26(2): 281–287. doi:10.1021/om0606643. Reactions of Indene and Indoles with Platinum Methyl Cations: Indene C-H Activation, Indole π vs. Nitrogen Lone-Pair Coordination Travis J. Williams, Jay A. Labinger, and John E. Bercaw Arnold and Mabel Beckman Laboratories of Chemical Synthesis, California Institute of Technology, Pasadena, CA 91125 (U. S. A.) Abstract Reactions of indene and various substituted indoles with [(diimine)PtII(Me)(TFE)]+ cations have been studied (diimine = ArN = C(Me) − C(Me) = NAr; TFE = 2,2,2-trifluoroethanol). Indene displaces the TFE ligand from platinum to form a stable π coordination complex that, upon heating, undergoes C-H activation with first order kinetics, ΔH‡ = 29 kcal/mol, ΔS‡ = 10 eu, and a kinetic isotope effect of 1.1 at 60 °C. Indoles also initially form coordination complexes through the C2=C3 olefin, but these undergo rearrangement to the corresponding N-bound complexes. The relative rates of initial coordination and rearrangement are affected by excess acid or methyl substitution on indole. Introduction Selective C-H bond activation is a potentially valuable approach to synthetic problems in areas ranging from fuels and bulk chemicals to fine chemicals and pharmaceutical synthesis. 1 Studies of C-H activation in our laboratory have focused on models of the Shilov system,2 particularly [(diimine)PtII(Me)(solv)]+ (2, diimine = ArN = C(Me) − C(Me) = NAr; solv = 2,2,2-trifluoroethanol (TFE), H2O).3 These cations are capable of activating a variety of II carbon-hydrogen bonds.4 Cations 2 can be generated by protonolysis of (diimine)Pt Me2 species 1 in TFE with aqueous HBF43, 4ab or BX3 (X = C6F5,4cd F5), the latter producing H+ by boron coordination to TFE.4c In deuterated solvent 2 is formed as a mixture of two isotopologs. -

October 1, 2020 Webinar
Phosphorus Specialties: the cornerstone of synthesis for pharmaceutical applications Eamonn Conrad, Ph.D., Global BD Manager Dino Amoroso, Ph.D., NA Account Manager William Stibbs, Ph.D. Senior BD Manager Overview • Solvay Phosphorus Specialties, Strem Chemicals Inc. Partnership • Chemistry for the Manufacture of Phosphine Ligands • Applications in Pharmaceutical Catalysis • Applications in Life Sciences • Summary and Questions *Solvay partners with Strem Chemicals for sample distribution page 2 Phosphorus Specialties Strem Chemicals, Inc. Solvay partners with Strem Chemicals for sample distribution! Established in 1964 More than 55 years of experience in manufacturing and handling high quality inorganics and organometallics 5,000+ specialty chemicals available Laboratory Chemicals for R&D cGMP Products Manufactured in Kilo-lab Suites High Pressure Materials Custom Synthesis Projects Customers include: Corporate Headquarters Academic, industrial and government R&D laboratories Corporate Headquarters European Headquarters Commercial scale businesses in the pharmaceutical, Newburyport, MA USA Strasbourg, France microelectronics, chemical & petrochemical industries Phosphorus Specialties Samples available from Strem Who We Are Phosphorus Specialties Mining Solutions Polymer Additives Putting our science to work for customers to develop differentiated products and technologies Dedicated on-site technical service and applications expertise to support our customers’ needs Deep customer relationships and ongoing collaborations to solve demanding -

Chemical Resistance List
Chemical Resistance List Resistance Substance Permeation Time/Level to Degradation Fluoro- natural chloro- nitrile/ nitrile carbon butyl latex prene chloroprene rubber NR CR CR NBR FKM IIR NR NR CR CR NBR NBR NBR NBR NBR FKM IIR IIR NBR NBR 395 450, 451 720, 722 717 727 730, 732 740, 741 743 754 764 890 897 898 chemical physical 403 706 723, 725 733, 836 742, 757 state 708 726 736 - 739 759 - 0 - 0 0 + 1-methoxy-2-propanol paste 4 2 2 3 4 4 B 1 3 4 6 6 - 0 - 0 0 + 1-methoxy-2-propyl acetate liquid 3 1 1 3 3 A B 2 3 6 6 - 0 0 - 0 + 1-methyl-2-pyrrolidone liquid 5 2 3 3 3 2 A B 1 3 3 6 6 - 0 + + + - 1,1,2-trichlorotrifluoroethane liquid 1 0 5 4 6 6 1 1 2 1 6 1 2 - - - - - - 1.2-epoxy ethane (ethylene oxide) liquid B A A A A 0 0 0 B 1 2 - - - - - - 1.2-epoxy propane (propylene oxide) liquid B A A A 1 A 0 0 0 B 1 2 + + + + + + 1.2-propanediol liquid 6 6 6 6 6 6 6 6 6 6 6 6 6 - + - + + 0 2-ethyl hexyl acrylate liquid 2 1 1 5 6 1 1 2 6 2 3 - 0 0 0 + + 2-mercaptoethanol liquid 3 2 4 4 4 4 1 1 3 6 6 6 - - - 0 0 - 2-methoxy-2-methyl propane liquid 1 B B 2 4 A 1 4 1 3 2 2 - - - - - 0 3-hexanone liquid 1 B 1 1 1 0 0 0 0 0 3 3 - - - - - 0 4-heptanone liquid 1 A 1 1 1 A 0 0 0 B 3 3 - - - - - + acetaldehyde liquid 1 1 1 1 B 0 0 0 A 0 6 6 0 0 0 - - + acetic acid anhydride liquid 6 3 3 3 3 2 A B 1 B 2 6 6 + + + + + + acetic acid, 10 % liquid 6 6 6 6 6 6 6 6 6 6 6 6 6 0 + + + + + acetic acid, 50 % liquid 5 4 6 6 6 2 4 6 6 6 6 - - - - 0 + acetic acid, conc. -

Studies on Metal Complex Formation of Environmentally Friendly Aminopolycarboxylate Chelating Agents
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Helsingin yliopiston digitaalinen arkisto Studies on metal complex formation of environmentally friendly aminopolycarboxylate chelating agents Helena Hyvönen Laboratory of Inorganic Chemistry Department of Chemistry Faculty of Science University of Helsinki Finland Academic dissertation To be presented with the permission of the Faculty of Science of the University of Helsinki for public criticism in Auditorium A110 of the Department of Chemistry, A.I. Virtasen aukio 1, on June 25th 2008 at 12 o’clock noon Helsinki 2008 Supervisor Professor Heikki Saarinen Department of Chemistry University of Helsinki Finland Reviewers Professor Konstantin Popov Physical and Colloid Chemistry Department Moscow State University of Food Production Russia Professor Mika Sillanpää Department of Environmental Sciences University of Kuopio Finland Opponent Professor Lauri Lajunen Department of Chemistry University of Oulu Finland © Helena Hyvönen ISBN 978-952-92-4005-0 (paperback) ISBN 978-952-10-4741-1 (PDF) http://ethesis.helsinki.fi Yliopistopaino Helsinki 2008 2 Abstract For decades, ethylenediaminetetraacetic acid (EDTA) and other aminopolycarboxylates with similar complexation properties and applicability have been widely used as chelating agents in various branches of industry. Recently, the low biodegradability of these ligands and their accumulation in the environment has become cause for concern, because of the persistence of these ligands and their metal complexes in nature. Ethylenediaminedisuccinic acid (EDDS), iminodisuccinic acid (ISA), N-bis[2-(1,2- dicarboxyethoxy)ethyl]aspartic acid (BCA6), N-bis[2-(1,2-dicarboxyethoxy)ethyl]- glycine (BCA5), N-bis[2-(1,2-dicarboxyethoxy)ethyl]methylglycine (MBCA5) and N- tris[(1,2-dicarboxy-ethoxy)ethyl]amine (TCA6) are more environmentally benign and potential candidates to replace EDTA, and also diethylenetriaminepentaacetic acid (DTPA), in several applications. -

1/2 Toxic Compound Data Sheet Name: Indene CAS Number: 00095
1/2 Toxic Compound Data Sheet Name: Indene CAS Number: 00095-13-6 Justification: This compound is listed in Ohio Administrative Code 3745 - 114 - 01 because it fulfills one or more of the following criteria: substances that are known to be, or may reasonably be anticipated to be, carcinogenic, mutagenic, teratogenic, or neurotoxic, causes reproductive dysfunction, is acutely or chronically toxic, or causes the threat of adverse environmental effects through ambient concentrations, bioaccumulation, or atmospheric deposition. lndene is acutely toxic with effects on the upper respiratory system (mucous membranes), pulmonary irritation, liver and kidney effects. Molecular Weight (g/mol): 116.15 Synonyms: Indonaphthene U.S. EPA Carcinogenic Classification (IRIS): Not listed on IRIS. PBT: Not listed as persistent, bioaccumulative and toxic. NTP: Not listed by the National Toxicology Program. HAP: Not listed as a hazardous air pollutant (HAP) by U.S. EPA. 112r: Not listed under Section 112(r) of the Clean Air Act. ACGIH: TLV: 10 ppm or 47,505 µg/m3. Critical effects include upper respiratory irritation or damage, pulmonary irritation, and liver and kidney effects. HSDB: Listed in the Hazardous Substances Data Bank. Inhalation of indene vapors is expected to cause irritation of mucous membranes. International IARC: Not listed by International Agency for Research on Cancer (IARC). ATSDR (MRL): Not listed by ATSDR. DataSheet Indene.wpd 2/2 Reference Material 1. American Conference of Governmental Industrial Hygienists (ACGIH) 2006. TLVs and BEIs: -

Using SMILES Strings for the Description of Chemical Connectivity in the Crystallography Open Database
Quirós et al. J Cheminform (2018) 10:23 https://doi.org/10.1186/s13321-018-0279-6 RESEARCH ARTICLE Open Access Using SMILES strings for the description of chemical connectivity in the Crystallography Open Database Miguel Quirós1* , Saulius Gražulis2,3 , Saulė Girdzijauskaitė3, Andrius Merkys2 and Antanas Vaitkus2 Abstract Computer descriptions of chemical molecular connectivity are necessary for searching chemical databases and for predicting chemical properties from molecular structure. In this article, the ongoing work to describe the chemical connectivity of entries contained in the Crystallography Open Database (COD) in SMILES format is reported. This col- lection of SMILES is publicly available for chemical (substructure) search or for any other purpose on an open-access basis, as is the COD itself. The conventions that have been followed for the representation of compounds that do not ft into the valence bond theory are outlined for the most frequently found cases. The procedure for getting the SMILES out of the CIF fles starts with checking whether the atoms in the asymmetric unit are a chemically accept- able image of the compound. When they are not (molecule in a symmetry element, disorder, polymeric species,etc.), the previously published cif_molecule program is used to get such image in many cases. The program package Open Babel is then applied to get SMILES strings from the CIF fles (either those directly taken from the COD or those produced by cif_molecule when applicable). The results are then checked and/or fxed by a human editor, in a computer-aided task that at present still consumes a great deal of human time. -

Phosphine-Catalyzed Additions of Nucleophiles and Electrophiles to Α
PHOSPHINE-CATALYZED ADDITIONS OF NUCLEOPHILES AND ELECTROPHILES TO α,β–UNSATURATED CARBONYL COMPOUNDS Reported by Michael Scott Bultman November 4, 2004 INTRODUCTION Organophosphorous compounds are becoming increasingly important in organic synthesis. Phosphines serve as precursors to phosphonium ylides in the Wittig reaction,1 and as nucleophilic triggers in the Mitsunobu2 and Staudinger3 reactions. In these processes, the phosphine is stoichiometrically consumed and converted into a phosphine oxide. Phosphines are also commonly used as ligands for transition metal-catalyzed reactions, to modulate reactivity and stereocontrol.4 On the other hand, the use of phosphines as nucleophilic catalysts for organic reactions has only gained attention in the last ten years. First reported by Rauhut and Currier in 1963,5 phosphine catalysis has since been reinvestigated after the phosphine ligands in some transition-metal-catalyzed reactions were found to be better catalysts than the metal/phosphine complexes alone!6 Phosphines are well suited for catalyzing the addition of both nucleophiles and electrophiles to electron deficient alkenes, alkynes, and allenes. Activation of these α,β-unsaturated carbonyl systems with the phosphine enables the formation of new bonds at the α-, β-, and γ-positions. This report will highlight these different modes of addition to α,β-unsaturated carbonyl systems under phosphine catalysis that allow for the formation of a wide array of products from a single class of substrates. GENERAL REACTIVITY OF PHOSPHINES Key characteristics required for successful nucleophilic catalysis lie in the balance of leaving group ability, nucleophilicity, and ease of ylid formation. Increasing leaving group ability can often be + correlated with decreasing basicity. -
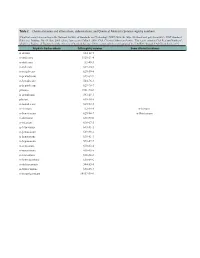
Table 2. Chemical Names and Alternatives, Abbreviations, and Chemical Abstracts Service Registry Numbers
Table 2. Chemical names and alternatives, abbreviations, and Chemical Abstracts Service registry numbers. [Final list compiled according to the National Institute of Standards and Technology (NIST) Web site (http://webbook.nist.gov/chemistry/); NIST Standard Reference Database No. 69, June 2005 release, last accessed May 9, 2008. CAS, Chemical Abstracts Service. This report contains CAS Registry Numbers®, which is a Registered Trademark of the American Chemical Society. CAS recommends the verification of the CASRNs through CAS Client ServicesSM] Aliphatic hydrocarbons CAS registry number Some alternative names n-decane 124-18-5 n-undecane 1120-21-4 n-dodecane 112-40-3 n-tridecane 629-50-5 n-tetradecane 629-59-4 n-pentadecane 629-62-9 n-hexadecane 544-76-3 n-heptadecane 629-78-7 pristane 1921-70-6 n-octadecane 593-45-3 phytane 638-36-8 n-nonadecane 629-92-5 n-eicosane 112-95-8 n-Icosane n-heneicosane 629-94-7 n-Henicosane n-docosane 629-97-0 n-tricosane 638-67-5 n-tetracosane 643-31-1 n-pentacosane 629-99-2 n-hexacosane 630-01-3 n-heptacosane 593-49-7 n-octacosane 630-02-4 n-nonacosane 630-03-5 n-triacontane 638-68-6 n-hentriacontane 630-04-6 n-dotriacontane 544-85-4 n-tritriacontane 630-05-7 n-tetratriacontane 14167-59-0 Table 2. Chemical names and alternatives, abbreviations, and Chemical Abstracts Service registry numbers.—Continued [Final list compiled according to the National Institute of Standards and Technology (NIST) Web site (http://webbook.nist.gov/chemistry/); NIST Standard Reference Database No. -

S1508 2.5 KG Sodium Tripolyphosphate
Scientific Documentation S1508, Sodium Tripolyphosphate, FCC Not appropriate for regulatory submission. Please visit www.spectrumchemical.com or contact Tech Services for the most up‐to‐date information contained in this information package. Spectrum Chemical Mfg Corp 769 Jersey Avenue New Brunswick, NJ 08901 Phone 732.214.1300 Ver4.01 27.April.2016 Dear Customer, Thank you for your interest in Spectrum’s quality products and services. Spectrum has been proudly serving our scientific community for over 45 years. It is our mission to manufacture and distribute fine chemicals and laboratory products with Quality and delivery you can count on every time. To accomplish our mission, Spectrum utilizes our sourcing leverage and supplier qualification expertise in offering one of the industry’s most comprehensive line of fine chemical products under one brand, in packaging configurations designed to meet your research and production requirements. Our product grades include: USP, NF, BP, EP, JP, FCC, ACS, KSA, Reagent grade, as well as DEA controlled substances. We operate facilities in the United States on the East Coast, West Coast, as well as in Shanghai, China in order to provide the best logistical support for our customers. At Spectrum, Quality is priority number one. Suppliers with the best qualifications are preferred and we employ full-functioning in-house analytical laboratories at each of our facilities. Our facilities and systems are USFDA registered and ISO certified. We frequently host customer audits and cherish opportunities for improvements. Quality is engrained into our culture. Quality is priority number one. In the following pages, we have designed and prepared documented scientific information to aid you in your initial qualification or your continual use of our products. -

Guidelines for Pyrophoric Materials
Guidelines for Pyrophoric Materials Definition and Hazards Pyrophoric materials are substances that ignite instantly upon exposure to air, moisture in the air, oxygen or water. Other common hazards include corrosivity, teratogenicity, and organic peroxide formation, along with damage to the liver, kidneys, and central nervous system. Examples include metal hydrides, finely divided metal powders, nonmetal hydride and alkyl compounds, white phosphorus, alloys of reactive materials and organometallic compounds, including alkylithiums. Additional pyrophoric materials are listed in Appendix A. Failure to follow proper handling techniques could result in serious injury or death. Controlling the Hazards . If possible, use safer chemical alternatives. A “dry run” of the experiment should be performed using low-hazard materials, such as water or solvent, as appropriate. Limit the amount purchased and the amount stored. Do not accumulate unneeded pyrophoric materials. BEFORE working with pyrophoric materials, read the MSDS sheets. The MSDS must be reviewed before using an unfamiliar chemical and periodically as a reminder. A Standard Operating Procedure (SOP) should be prepared and reviewed for each process involving pyrophoric materials. In lab training should be completed and documented. If possible, use the “buddy system”. Working alone with pyrophorics is strongly discouraged. All glassware used for pyrophorics should be oven-dried and free of moisture. Review the location of the safety shower, eyewash, telephone, and fire extinguisher. Keep an appropriate fire extinguisher or extinguishing material close at hand. Additional controls when handling liquid pyrophoric materials . Secure pyrophoric reagent bottle to stand. Secure the syringe so if the plunger blows out of the body of the syringe the contents will not splash anyone. -

TRI.N.BUTYL PHOSPHATE
This report contains the collective views of an in- ternational group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the Interna- tional Labour Organisation, or the World Health Organization. Environmental Health Cnteria ll2 TRI.n.BUTYL PHOSPHATE Published under the joint sponsorshipof the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization First draft prepared by Dr A. Nakamura, National Institute for Hygienic Sciences,Japan World Health Organization Geneva,1991 The International Progranne on Chemical Safety (IPCS) is a joint venture of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization. The main objec- tive of the IPCS is to carry out and disseminate evaluations of the effects of chemicals on human health and the quality of the environ- ment. Supporting activities include the development of epidemiological, experimental laboratory, and risk-assessment methods that could produce internationally comparable results, and the development of manpower in the field of toxicology. Other activities carried out by the IPCS include the development of know-how for coping with chemical accidents, coordination of laboratory testing and epidemiological studies, and promotion of research on the mechanisms of the biological action of chemicals. WHO Library Cataloguing in Publication Data Tri-n-butyl phosphate. (Environmental health criteria ; l12) l. Phosphoric acid esters - adverse effects 2. Phosphoric acid esters - toxicity I. Series rsBN92 4 r57rr2 8 (NLM Classification: QV 627) rssN0250-863X @World Health Organization l99l Publications of the \ilorld Health Organization enjoy copyright pro- tection in accordance with the provisions of Protocol 2 of the Univer- sal Copyright Convention.