Controlled Synthesis of Cus and Cu9s5 and Their Application in the Photocatalytic Mineralization of Tetracycline
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Biofilm Adhesion on the Sulfide Mineral Bornite & Implications for Astrobiology
University of Rhode Island DigitalCommons@URI Open Access Master's Theses 2019 BIOFILM ADHESION ON THE SULFIDE MINERAL BORNITE & IMPLICATIONS FOR ASTROBIOLOGY Margaret M. Wilson University of Rhode Island, [email protected] Follow this and additional works at: https://digitalcommons.uri.edu/theses Recommended Citation Wilson, Margaret M., "BIOFILM ADHESION ON THE SULFIDE MINERAL BORNITE & IMPLICATIONS FOR ASTROBIOLOGY" (2019). Open Access Master's Theses. Paper 1517. https://digitalcommons.uri.edu/theses/1517 This Thesis is brought to you for free and open access by DigitalCommons@URI. It has been accepted for inclusion in Open Access Master's Theses by an authorized administrator of DigitalCommons@URI. For more information, please contact [email protected]. BIOFILM ADHESION ON THE SULFIDE MINERAL BORNITE & IMPLICATIONS FOR ASTROBIOLOGY BY MARGARET M. WILSON A THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE IN BIOLOGICAL & ENVIRONMENTAL SCIENCE UNIVERSITY OF RHODE ISLAND 2019 MASTER OF SCIENCE IN BIOLOGICAL & ENVIRONMENTAL SCIENCE THESIS OF MARGARET M. WILSON APPROVED: Thesis Committee: Major Professor Dawn Cardace José Amador Roxanne Beinart Nasser H. Zawia DEAN OF THE GRADUATE SCHOOL UNIVERSITY OF RHODE ISLAND 2019 ABSTRACT We present research observing and documenting the model organism, Pseudomonas fluorescens (P. fluorescens), building biofilm on a natural mineral substrate composed largely of bornite (Cu5FeS4), a copper-iron sulfide mineral, with closely intergrown regions of covellite (CuS) and chalcopyrite (CuFeS2). In examining biofilm establishment on sulfide minerals, we investigate a potential habitable niche for microorganisms in extraterrestrial sites. Geochemical microenvironments on Earth and in the lab can also serve as analogs for important extraterrestrial sites, such as sheltered, subsurface microenvironments on Mars. -

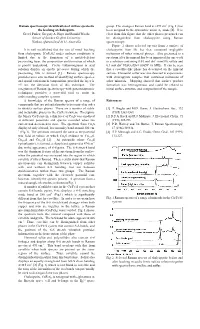
PHASE CHANGES in Cu 2 S AS a FUNCTION of TEMPERATURE 1. PREVIOUS WORK
National Bureau of Standards Special Publication 364, Solid State Chemistry, Proceedings of 5th Materials Research Symposium, issued July 1972. PHASE CHANGES IN cu2s AS A FUNCTION OF TEMPERATURE William R. Cook, Jr. Gould Inc., Gould Laboratories Cleveland, Ohio 44108 and Case Western Reserve University Cleveland, Ohio 44106 The high-'copper phase boundary of Cu2s deviates from stoichiometry above 300 °C, first becoming copper deficient, then above - 1075 °C becoming copper rich. The maximum copper content occurs at the monotectic temperature of 1104 °C. The strong effect of oxygen on the hexagonal-cubic transition in Cu2S was confirmed; the transition was also found to be sensi tive to the type of pretreatment of the material. The high temperature tetragonal "Cu1 96s" phase is stable between Cu1.95S and Cu2s, at temperatures of - 90° to - 140 °C. The tr~nsi tion to the tetragonal phase is extremely sluggish. The true composition of djurleite has been shown to be approximately Cu1.93S. The phases near the chalcocite-digenite region of the diagram may be grouped into those with hexagonal close packing of sulfur atoms and those with cubic close packing of sulfurs. This is important in understanding rates of transformation among the various phases that occur in this area of the diagram. Key words: Chalcocite; cu2s; digenite; djurleite; nonstoichiometry; phase relations. 1. PREVIOUS WORK Fairly thorough explorations of the Cu-s phase diagram between cu2s and cu1• ,shave been made by a number of previous workers [1-8] 1 The resulting diagram may be seen in figure 1 taken largely from Roseboom [7] and Riu [8]. -

IN-VITRO EVALUATION of the ANTICANCER ACTIVITY of Cu(II)AMINA(CYSTEINE)DITHIOCARBAMATE
Sys Rev Pharm 2020;11(9):43-51 A multifaceted review journal in the field of pharmacy IN-VITRO EVALUATION OF THE ANTICANCER ACTIVITY OF Cu(II)AMINA(CYSTEINE)DITHIOCARBAMATE Desy Kartina1, Abdul Wahid Wahab 2, Ahyar Ahmad3, Rizal Irfandi 4, Prihantono5, And Indah Raya6* 1Department of Chemistry, Faculty of Mathematics, and Natural Science, Hasanuddin University, Makassar 90245, Indonesia 2Department of Chemistry, Faculty of Mathematics, and Natural Science, Hasanuddin University, Makassar 90245, Indonesia 3Department of Chemistry, Faculty of Mathematics, and Natural Science, Hasanuddin University, Makassar 90245, Indonesia 4Department of Biology Education, Faculty of Teacher Training and Education, Puangrimaggalatung University Sengkang, 90915, Indonesia. 5Departement of Surgery, Faculty of Medicinal, Hasanuddin University, Makassar Indonesia, 90245 6*Department of Chemistry, Faculty of Mathematics, and Natural Science, Hasanuddin University, Makassar 90245, Indonesia ABSTRACT The Complex of Cu(II)cysteinedithiocarbamate has been synthesized, it Keywords: Anticancer, Cu(ll)Amina(cysteine)dithiocarbamate. was prepared by the “in situ method” and characterized by using Correspondance: Ultraviolet-Visible (UV-Vis), Infra-Red (IR) spectroscopy, X-Ray Indah Raya Fluorescence (XRF) instruments. While melting point and conductivity Department of Chemistry, also measured. The presence of UV-Vis maximum spectrums of Faculty of Mathematics, and Natural Science, Cu(II)cysteinedithiocarbamate at 296 nm and 436 nm indicated that Hasanuddin University, electronic transition π → π * dan n → π * of CS2 and N=C=S myotis. The Makassar 90245, Indonesia presence of the wavelength in the region of 399-540 cm-1 of IR spectra Email id : [email protected], [email protected] is indicated that has been coordination occurred between Cu(II) with Sulphur (S), Nitrogen (N), and Oxygen(O) atoms respectively from cysteinedithiocarbamate ligands. -
Raman Spectroscopic Identification of Surface Species in the Leaching Of
Raman spectroscopic identification of surface species in group. The strongest Raman band at »293 cm-1 (Fig. 1) has the leaching of chalcopyrite been assigned to the symmetric anion A1 mode [4]. It is Gretel Parker, Gregory A. Hope and Ronald Woods clear from this figure that the other phases presented can School of Science Griffith University be distinguished from chalcopyrite using Raman Nathan, Queensland 4111, Australia spectroscopy. Figure 2 shows selected spectra from a surface of It is well established that the rate of metal leaching chalcopyrite from Mt Isa that contained negligible from chalcopyrite [CuFeS2] under ambient conditions is inclusions of other mineral phases. Also presented is a limited due to the formation of a metal-deficient spectrum after the mineral has been immersed for one week passivating layer, the composition and formation of which in a solution containing 0.03 mol dm-3 iron(III) sulfate and -3 is poorly understood. Cyclic voltammograms in acid 0.1 mol dm H2SO4 (Eh = 0.865V vs SHE). It can be seen solution display an anodic pre-wave during which the that a covellite-like phase has developed on the mineral passivating film is formed [1]. Raman spectroscopy surface. Elemental sulfur was also detected in experiments provides an in situ method of identifying surface species, with chalcopyrite samples that contained inclusions of and spatial variations in composition, provided the layer is other minerals. Mapping showed that surface product >5 nm, the detection limit of this technique. The formation was heterogeneous and could be related to integration of Raman spectroscopy with potentiodynamic initial surface structure and composition of the sample. -

Recent Advances in Microwave-Assisted Copper-Catalyzed Cross-Coupling Reactions
catalysts Review Recent Advances in Microwave-Assisted Copper-Catalyzed Cross-Coupling Reactions Younis Baqi Department of Chemistry, College of Science, Sultan Qaboos University, P.O. Box 36, Muscat 123, Oman; [email protected]; Tel.: +968-2414-1473 Abstract: Cross-coupling reactions furnishing carbon–carbon (C–C) and carbon–heteroatom (C–X) bond is one of the most challenging tasks in organic syntheses. The early developed reaction protocols by Ullmann, Ullman–Goldberg, Cadiot–Chodkiewicz, Castro–Stephens, and Corey–House, utilizing elemental copper or its salts as catalyst have, for decades, attracted and inspired scientists. However, these reactions were suffering from the range of functional groups tolerated as well as severely restricted by the harsh reaction conditions often required high temperatures (150–200 ◦C) for extended reaction time. Enormous efforts have been paid to develop and achieve more sustainable reaction conditions by applying the microwave irradiation. The use of controlled microwave heating dramatically reduces the time required and therefore resulting in increase in the yield as well as the efficiency of the reaction. This review is mainly focuses on the recent advances and applications of copper catalyzed cross-coupling generation of carbon–carbon and carbon–heteroatom bond under microwave technology. Keywords: cross-coupling reaction; Cu catalyst; microwave irradiation; methodology; synthesis 1. Introduction Carbon–carbon (C–C) and carbon–heteroatom (C–X) bond formations through cross- Citation: Baqi, Y. Recent Advances in coupling reactions represents as one of the most useful strategy in the synthetic organic Microwave-Assisted chemistry, hence many procedures and methodologies have been developed and published Copper-Catalyzed Cross-Coupling in the literature. -

Supergene Mineralisation of the Boyongan Porphyry Copper-Gold Deposit, Surigao Del Norte, Philippines
Supergene Mineralisation of the Boyongan Porphyry Copper-Gold Deposit, Surigao del Norte, Philippines by Allan Maglaya Ignacio B.Sc. Geology, National Institute of Geological Sciences University of the Philippines Thesis submitted in partial fulfilment of the requirements of the Masters of Economic Geology Degree Centre for Ore Deposit Research, University of Tasmania December, 2005 DECLARATION OF ORIGINALITY This thesis contains no material which has been accepted for a degree of diploma by the University of Tasmania or any other institution, except by way of background information and duly acknowledged in the thesis, and contains no previous material previously pub- lished or written by another person except where due acknowledgement is given. Allan Maglaya Ignacio 01 December 2005 _________________________ STATEMENT OF AUTHORITY OF ACCESS This thesis may not to be made available for loan or copying for 1.5 years following the date this statement was signed. Following that time, the thesis may be available for loan and lim- ited copying in accordance with Copyright Act 1968. Allan Maglaya Ignacio 01 December 2005 _________________________ TABLE OF CONTENTS Page (s) LIST OF FIGURES …………………………………………………….. i - iii LIST OF APPENDICES ………………………………………………… iv ACKNOWLEDGMENTS ………………………………………………. v ABSTRACT ……………………………………………………………... vi - vii 1.0 INTRODUCTION ………………………………………………………. 1 - 8 1.1 Introduction …………………………………………………………. 1 1.2 Aims and Objectives ……………………………………………….. 1 1.3 Methods Employed …………………………………………………. 2 1.4 Location and Accessibility …………………………………………. 3 1.5 Climate ……………………………………………………………... 5 1.6 Previous Work ……………………………………………………… 5 2.0 GEOLOGICAL SETTING ………………………………………………. 9 - 37 2.1 Introduction ………………………………………………………. 9 2.2 Regional Tectonics …………….…………………………………. 9 2.3 Regional and Local Stratigraphy ………………………………... 11 2.3.1 Basement (Cretaceous-Paleogene) ………………………. 11 2.3.2 Bacuag Formation (Oliogocene-Miocene) .…………….. -

AN OCCURRENCE of the ASSEMBLAOE, NATIVE SULFTIR- COVELLITE-"Cu5 6,Fe,S6.S"", AUCANQUILCHA, CHILE Aran H. Cr-Etr&L
THE AMERICAN MINERALOGIST, VOL. 55, MAY_JUNE, 1970 AN OCCURRENCE OF THE ASSEMBLAOE, NATIVE SULFTIR- COVELLITE-"Cu5 6,Fe,S6.s"",AUCANQUILCHA, CHILE AraN H. Cr-etr<, Deportmentof GeologicalSciences, Queen's U niaersity, Kingston, Ontario. AssrnA.cr A mineral with composition near to CusFeSo has been found associated with nzrtive sulfur and covellite in the volcanic sulfur deposit at Aucanquilcha. Microprobe, optical and X-ray powder diffraction data match closell'a previously reported occurrence at Nu- kundamu, Fiji. Comparison with equilibrium synthesis by Kullerud and others indir:ates formation in the temperature range 434-482"C. INrnonucrroN Electron probe microanalysis(L6vy, 1967; Sillitoe and Clark, lt69) of naturally-occurring,supergene idaite has cast considerabledoub1. on the equation (Frenzel, 1959) of this not uncommon sulfi.de with the phaseof generalformula Cur r,Fe,Se.s,1 (Yund, 1963), which has h,een synthesizedby Merwin and Lombard (1937), Roseboomand Kullerud (1958),and Yund and Kullerud (1966).Idaite has been shown to have the compositionCurFeSa, or Cu3FeS4-,1orrd there is as yet no evidence of solid solution in nature between this and more copper-rich comF,osi- tions.The well-establishedX-ray powder data"(a:3.772 A; c:11.1U A; Yund, 1963) for hexagonal Cus.s,Fe"Se.r, are only with difficulty recon- ciled with thoseof natural idaite (Frenzel,1959, 1963), and L6vy (I\167) has suggestedthat Frenzel'soriginal powder data may be adequately fitted to a stannite-type,tetragonal cell. A differencein crystal struclure between thesephases is supported by the dissimilar reflectivity disper- sion profi.lesof natural idaite (L6vy, 1967; Sillitoe and Clark, 1969) and the synthetic "Cu5FeS6"of Merwin and l-ombard (1937; inLlvy, 1967). -

Copper ( II ) Chloride
Copyright © Tarek Kakhia. All rights reserved. http://tarek.kakhia.org Copper Contents 1 Introduction 2 History o 2.1 Copper Age o 2.2 Bronze Age o 2.3 Antiquity and Middle Ages o 2.4 Modern period 3 Characteristics o 3.1 Color o 3.2 Group 11 of the periodic table o 3.3 Occurrence o 3.4 Mechanical properties o 3.5 Electrical properties o 3.6 Corrosion . 3.6.1 Pure water and air/oxygen . 3.6.2 In contact with other metals . 3.6.3 Sulfide media . 3.6.4 Ammonia media . 3.6.5 Chloride media o 3.7 Germicidal effect o 3.8 Isotopes 4 Production 5 Applications o 5.1 Piping o 5.2 Electrical applications o 5.3 Architecture / Industry o 5.4 Household products o 5.5 Coinage o 5.6 Biomedical applications o 5.7 Chemical applications o 5.8 Other 6 Alloys 7 Compounds 8 Biological role 9 Recycling 1 Copyright © Tarek Kakhia. All rights reserved. http://tarek.kakhia.org 1 Introduction : Copper is a chemical element with the symbol Cu ( Latin : cuprum ) and atomic number 29 . It is a ductile metal with very high thermal and electrical conductivity. Pure copper is rather soft and malleable and a freshly - exposed surface has a pinkish or peachy color. It is used as a thermal conductor, an electrical conductor, a building material, and a constituent of various metal alloys. Copper metal and alloys have been used for thousands of years. In the Roman era, copper was principally mined on Cyprus, hence the origin of the name of the metal as Cyprium, "metal of Cyprus", later shortened to Cuprum. -

Copper Monosulfide (Cus) Powder SAFTY DATA SHEET
Copper Monosulfide (CuS) Powder US Research Nanomaterials, Inc. www.us-nano.com SAFTY DATA SHEET Revised Date 2/21/2016 1. PRODUCT AND COMPANY IDENTIFICATION 1.1 Product identifiers Product name: Copper Monosulfide (CuS) Powder Product Number : US1126M Copper Monosulfide (CuS) CAS#: 1317-40-4 1.2 Relevant identified uses of the substance or mixture and uses advised against Identified uses : Research 1.3 Details of the supplier of the safety data sheet Company: US Research Nanomaterials, Inc. 3302 Twig Leaf Lane Houston, TX 77084 USA Telephone: +1 832-460-3661 Fax: +1 281-492-8628 1.4 Emergency telephone number Emergency Phone # : (832) 359-7887 2. HAZARDS IDENTIFICATION 2.1 Classification of the substance or mixture This substance or mixture is not hazardous and is not classified under GHS. May cause irritation of skin, eyes, and respiratory tract. For the full text of the H-Statements mentioned in this Section, see Section 16. 2.2 GHS Label elements, including precautionary statements Pictogram Signal word None Hazard statement(s) H319 Causes serious eye irritation. H335 May cause respiratory irritation. Precautionary statement(s) P261 Avoid breathing dust/ fume/ gas/ mist/ vapors/ spray. P264 Wash skin thoroughly after handling. P271 Use only outdoors or in a well-ventilated area. P280 Wear protective gloves/ eye protection/ face protection. P304 + P340 IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing. P305 + P351 + P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. P312 Call a POISON CENTER or doctor/ physician if you feel unwell. -

MENDELSOHN (F.), Editor. the Geology of the Northern Rhodesian Copper- Belt
996 BOOK REVIEWS instance 'pits', ' anisotropy', ' cellular texture', ' native iron', are fol- lowed by a 'text-card' with a more detailed description of the sections concerned in English and German. The price is DM 0.75 per photo-card, DM 0.45 per text-card. These prices are reduced by 10% for University departments and academic staff, by 15% for students. The great value of this unique publication is self-evident and does not need further appraisal. Ore-microscopists will welcome the card index as a high-quality source of reference, and university staff will find it a great help in teaching. Economic geologists and nfineralogists will appreciate the straightforward and clear approach, which provides an excellent introduction for research workers less well acquainted with the subject matter. Ore dressing workers and metallurgists will be able to Use the card index for the solution of their problems without having to go into details of ore mineralogy. The authors should be warmly con- gratulated on this first part of a very fine piece of work. A. F. H. MENDELSOHN (F.), editor. The Geology of the Northern Rhodesian Copper- belt. London (Macdonald & Co. Ltd.), 1961, xvi+523 pp., 185 figs. Price 8r Twenty-three authors have contributed to this comprehensive volume. Part 1 provides a general account of the physiography, stratigraphy, structure, metamorphism, and ore deposits of the Copperbelt together with a history of mineral exploration and an account of the methods employed. Part 2 gives detailed descriptions of the individual deposits. In addition to synthesizing the considerable volume of published work on the subject the authors draw extensively on material from unpublished company reports. -

Silver-Rich Central Idaho
Silver-rich Disseminated Sulfides From a Tungsten-bearing Quartz Lode Big Creek District Central Idaho GEOLOGICAL SURVEY PROFESSIONAL PAPER 594-C Silver-rich Disseminated Sulfides From a Tungsten-bearing Quartz Lode Big Creek District Central Idaho By B. F. LEONARD, CYNTHIA W. MEAD, and NANCY CONKLIN SHORTER CONTRIBUTIONS TO GENERAL GEOLOGY GEOLOGICAL SURVEY PROFESSIONAL PAPER 594-C Study of a low-grade tungsten deposit whose associated suljide minerals, extracted as waste, are rich in silver and contain some gold UNITED STATES GOVERNMENT PRINTING OFFICE, WASHINGTON : 1968 UNITED STATES DEPARTMENT OF THE INTERIOR STEWART L. UDALL, Secretary GEOLOGICAL SURVEY William T. Pecora, Director For sale by the Superintendent of Documents, U.S. Government Printing Office Washington, D.C. 20402 - Price 35 cents (paper cover) CONTENTS Page Page Abstract __________________________________________ _ C1 Mineralogy and paragenetic sequence-Continued Introduction ______________________________________ _ 1 Alteration products ____________________________ _ C15 Location and operation _____________________________ _ 4 Paragenetic sequence ___________________________ _ 16 Geology __________________________________________ _ 4 Geologic thermometry __________________________ _ 18 Ore deposit _______________________________________ _ 5 Classification and origin of hypogene mineralization ____ _ 19 Mineralogy and paragenetic sequence ________________ _ 6 Oxidation and enrichment ___ ------------------------ 20 Typical ore ___________________________________ _ 6 Economic considerations ____________________________ _ 21 Gangue indicated by mill products _______________ _ 7 Acknowledgments ___________________ .:. ______________ _ 23 Tungsten minerals _______ ,______________________ _ 8 References ________________________________________ _ 23 Sulfides and related minerals ____________________ _ 8 ILLUSTRATIONS [Plates follow page C24f - PLATE 1. Drawing and X-ray micrographs of acanthite and copper sulfides on galena. 2. Drawing and X-ray micrographs of electrum in pyrite. 3. -

Covellite) Probed by NQR
Phase transition and anomalous electronic behavior in layered dichalcogenide CuS (covellite) probed by NQR R.R. Gainov 1*, A.V. Dooglav 1, I.N. Pen’kov 2, I.R. Mukhamedshin 1,3, N.N. Mozgova 4, A.V. Evlampiev 1, and I.A. Bryzgalov 5 1 Department of Physics, Magnetic RadioSpectroscopy Laboratory, Kazan State University, Kazan, Kremlevskaya str. 18, 420008, Russian Federation 2 Department of Geology, Kazan State University, Kazan, Kremlevskaya str. 4/5, 420111, Russian Federation 3 Laboratoire de Physique des Solides, UMR 8502, Universite Paris-Sud, 91405 Orsay, France 4 Institute of geology of ore deposits, petrography, mineralogy and geochemistry (Russian Academy of Science), Staromonetny per. 35, Moscow 109017, Russian Federation 5 Department of Geology, Moscow State University, Moscow, Vorob’evy gory, 119991, Russian Federation * Corresponding author: tel.: 7-843-2315175; fax.: 7-843-2387201. Electronic address: [email protected] (R.R. Gainov). Nuclear quadrupole resonance (NQR) on copper nuclei has been applied for studies of the electronic properties of quasi-two-dimensional low-temperature superconductor CuS (covellite) in the temperature region between 1.47 and 290 K. Two NQR signals corresponding to two non- equivalent sites of copper in the structure, Cu(1) and Cu(2), has been found. The temperature dependences of copper quadrupole frequencies, line-widths and spin-lattice relaxation rates, which so far had never been investigated so precisely for this material, altogether demonstrate the structural phase transition near 55 K, which accompanies transformations of electronic spectrum not typical for simple metals. The analysis of NQR results and their comparison with literature data show that the valence of copper ions at both sites is intermediate in character between monovalent and divalent states with the dominant of the former.