Chapter 1 – Introduction
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-
![A Study of the Impact of Mazie SBE I on the [Alpha]-Polyglucan Produced in Synechocystis Sp](https://docslib.b-cdn.net/cover/1900/a-study-of-the-impact-of-mazie-sbe-i-on-the-alpha-polyglucan-produced-in-synechocystis-sp-121900.webp)
A Study of the Impact of Mazie SBE I on the [Alpha]-Polyglucan Produced in Synechocystis Sp
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 2007 A study of the impact of mazie SBE I on the [alpha]- polyglucan produced in Synechocystis sp. strain PCC 6803 Shayani Deborah Nesaranjani Pieris Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Plant Biology Commons Recommended Citation Pieris, Shayani Deborah Nesaranjani, "A study of the impact of mazie SBE I on the [alpha]-polyglucan produced in Synechocystis sp. strain PCC 6803" (2007). Retrospective Theses and Dissertations. 15850. https://lib.dr.iastate.edu/rtd/15850 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. A study of the impact of mazie SBE I on the -polyglucan produced in Synechocystis sp. strain PCC 6803 by Shayani Deborah Nesaranjani Pieris A dissertation submitted to the graduate faculty in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY Major: Plant Physiology Program of Study Committee: Martin H. Spalding, Major Professor Madan K. Bhattacharyya Jay -lin Jane David J. Oliver Paul M. Scott Iowa State University Ames, Iowa 200 7 Copyright © Shayani Deborah Nesara njani Pieris , 200 7. All right s reserved. UMI Number: 3294976 UMI Microform 3294976 Copyright 2008 by ProQuest Information and Learning Company. All rights reserved. This microform edition is protected against unauthorized copying under Title 17, United States Code. -

Review Article Pullulanase: Role in Starch Hydrolysis and Potential Industrial Applications
Hindawi Publishing Corporation Enzyme Research Volume 2012, Article ID 921362, 14 pages doi:10.1155/2012/921362 Review Article Pullulanase: Role in Starch Hydrolysis and Potential Industrial Applications Siew Ling Hii,1 Joo Shun Tan,2 Tau Chuan Ling,3 and Arbakariya Bin Ariff4 1 Department of Chemical Engineering, Faculty of Engineering and Science, Universiti Tunku Abdul Rahman, 53300 Kuala Lumpur, Malaysia 2 Institute of Bioscience, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia 3 Institute of Biological Sciences, Faculty of Science, University of Malaya, 50603 Kuala Lumpur, Malaysia 4 Department of Bioprocess Technology, Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia Correspondence should be addressed to Arbakariya Bin Ariff, [email protected] Received 26 March 2012; Revised 12 June 2012; Accepted 12 June 2012 Academic Editor: Joaquim Cabral Copyright © 2012 Siew Ling Hii et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. The use of pullulanase (EC 3.2.1.41) has recently been the subject of increased applications in starch-based industries especially those aimed for glucose production. Pullulanase, an important debranching enzyme, has been widely utilised to hydrolyse the α-1,6 glucosidic linkages in starch, amylopectin, pullulan, and related oligosaccharides, which enables a complete and efficient conversion of the branched polysaccharides into small fermentable sugars during saccharification process. The industrial manufacturing of glucose involves two successive enzymatic steps: liquefaction, carried out after gelatinisation by the action of α- amylase; saccharification, which results in further transformation of maltodextrins into glucose. -

Function and Structure Studies of GH Family 31 and 97 \Alpha
110610 (RV-17) Biosci. Biotechnol. Biochem., 75 (12), 110610-1–9, 2011 Award Review Function and Structure Studies of GH Family 31 and 97 -Glycosidases Masayuki OKUYAMA Research Faculty of Agriculture, Hokkaido University, Kita-9, Nishi-9, Kita-ku, Sapporo 060-8589, Japan Online Publication, December 7, 2011 [doi:10.1271/bbb.110610] A huge number of glycoside hydrolases are classified an evolutionary relationship of proteins in the family, into the glycoside hydrolase family (GH family) based from which it is often possible to extract information on on their amino-acid sequence similarity. The glycoside function and structure.3) Classification in a GH family hydrolases acting on -glucosidic linkage are in GH has, accordingly, become indispensable for research on family 4, 13, 15, 31, 63, 97, and 122. This review deals glycoside hydrolases. Glycoside hydrolases are also mainly with findings on GH family 31 and 97 enzymes. divided into two mechanistic classes, inverting and Research on two GH family 31 enzymes is described: retaining enzymes: with inversion or net retention of clarification of the substrate recognition of Escherichia anomeric configuration during the catalytic reaction.4) coli -xylosidase, and glycosynthase derived from Schiz- The stereochemical outcome is generally conserved in a osaccharomyces pombe -glucosidase. GH family 97 is GH family. The inverting mechanism proceeds via a an aberrant GH family, containing inverting and simple single displacement. Two functional groups, retainingAdvance glycoside hydrolases. The inverting View enzyme usually carboxyl groups, act as general acid and general in GH family 97 displays significant similarity to base catalysts. The general acid catalyst donates a proton retaining -glycosidases, including GH family 97 retain- to the departure aglycon, and the general base catalyst ing -glycosidase, but the inverting enzyme has no simultaneously deprotonates the incoming water mole- catalytic nucleophile residue. -

(12) Patent Application Publication (10) Pub. No.: US 2007/0143878 A1 Bhat Et Al
US 20070143878A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2007/0143878 A1 Bhat et al. (43) Pub. Date: Jun. 21, 2007 (54) NUCLEC ACID MOLECULES AND OTHER of application No. 09/198.779, filed on Nov. 24, 1998, MOLECULES ASSOCATED WITH THE now abandoned. TOCOPHEROL PATHWAY Said application No. 09/233,218 is a continuation-in part of application No. 09/227,586, filed on Jan. 8, (76) Inventors: Barkur G. Bhat, St. Louis, MO (US); 1999, now abandoned. Sekhar S. Boddupalli, Manchester, MO Said application No. 09/233,218 is a continuation-in (US); Ganesh M. Kishore, Creve part of application No. 09/229,413, filed on Jan. 12, Coeur, MO (US); Jingdong Liu, 1999, now abandoned. Ballwin, MO (US); Shaukat H. Rangwala, Ballwin, MO (US); (60) Provisional application No. 60/067,000, filed on Nov. Mylavarapu Venkatramesh, Ballwin, 24, 1997. Provisional application No. 60/066,873, MO (US) filed on Nov. 25, 1997. Provisional application No. 60/069.472, filed on Dec. 9, 1997. Provisional appli Correspondence Address: cation No. 60/074,201, filed on Feb. 10, 1998. Pro ARNOLD & PORTER, LLP visional application No. 60/074.282, filed on Feb. 10, 555 TWELFTH STREET, N.W. 1998. Provisional application No. 60/074,280, filed ATTN IP DOCKETING on Feb. 10, 1998. Provisional application No. 60/074, WASHINGTON, DC 20004 (US) 281, filed on Feb. 10, 1998. Provisional application No. 60/074,566, filed on Feb. 12, 1998. Provisional (21) Appl. No.: 11/329,160 application No. 60/074,567, filed on Feb. 12, 1998. -

102 4. Biosynthesis of Natural Products Derived from Shikimic Acid
102 4. Biosynthesis of Natural Products Derived from Shikimic Acid 4.1. Phenyl-Propanoid Natural Products (C6-C3) The biosynthesis of the aromatic amino acids occurs through the shikimic acid pathway, which is found in plants and microorganisms (but not in animals). We (humans) require these amino acids in our diet, since we are unable to produce them. For this reason, molecules that can inhibit enzymes on the shikimate pathway are potentially useful as antibiotics or herbicides, since they should not be toxic for humans. COO COO NH R = H Phenylalanine 3 R = OH Tyrosine R NH3 N Tryptophan H The aromatic amino acids also serve as starting materials for the biosynthesis of many interesting natural products. Here we will focus on the so-called phenyl-propanoide (C6-C3) natural products, e.g.: OH OH OH HO O HO OH HO O Chalcone OH O a Flavone OH O OH O a Flavonone OH OH Ar RO O O O HO O O OH O OR OH Anthocyanine OH O a Flavonol Podophyllotoxin MeO OMe OMe OH COOH Cinnamyl alcohol HO O O Cinnamic acid OH (Zimtsäure) Umbellierfone OH a Coumarin) MeO OH O COOH HO Polymerization OH Wood OH HO OH O OH MeO OMe Shikimic acid O HO 4.2. Shikimic acid biosynthesis The shikimic acid pathway starts in carbohydrate metabolism. Given the great social and industrial significance of this pathway, the enzymes have been intensively investigated. Here we will focus on the mechanisms of action of several key enzymes in the pathway. The following Scheme shows the pathway to shikimic acid: 103 COO- COO- Phosphoenolpyruvate HO COO- 2- O O3P-O 2- O3P-O DHQ-Synthase -

8.2 Shikimic Acid Pathway
CHAPTER 8 © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FORAromatic SALE OR DISTRIBUTION and NOT FOR SALE OR DISTRIBUTION Phenolic Compounds © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION CHAPTER OUTLINE Overview Synthesis and Properties of Polyketides 8.1 8.5 Synthesis of Chalcones © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC 8.2 Shikimic Acid Pathway Synthesis of Flavanones and Derivatives NOT FOR SALE ORPhenylalanine DISTRIBUTION and Tyrosine Synthesis NOT FOR SALESynthesis OR DISTRIBUTION and Properties of Flavones Tryptophan Synthesis Synthesis and Properties of Anthocyanidins Synthesis and Properties of Isofl avonoids Phenylpropanoid Pathway 8.3 Examples of Other Plant Polyketide Synthases Synthesis of Trans-Cinnamic Acid Synthesis and Activity of Coumarins Lignin Synthesis Polymerization© Jonesof Monolignols & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC Genetic EngineeringNOT FOR of Lignin SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION Natural Products Derived from the 8.4 Phenylpropanoid Pathway Natural Products from Monolignols © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION © Jones & Bartlett Learning, LLC © Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION NOT FOR SALE OR DISTRIBUTION 119 © Jones & Bartlett Learning, LLC. -

Elimination of Competing Hydrolysis and Coupling Side Reactions of A
Elimination of competing hydrolysis and coupling side reactions of a cyclodextrin glucanotransferase by directed evolution Ronan M Kelly, Hans Leemhuis, Henriëtte J Rozeboom, Niels van Oosterwijk, Bauke W. Dijkstra, Lubbert Dijkhuizen To cite this version: Ronan M Kelly, Hans Leemhuis, Henriëtte J Rozeboom, Niels van Oosterwijk, Bauke W. Dijkstra, et al.. Elimination of competing hydrolysis and coupling side reactions of a cyclodextrin glucan- otransferase by directed evolution. Biochemical Journal, Portland Press, 2008, 413 (3), pp.517-525. 10.1042/BJ20080353. hal-00478977 HAL Id: hal-00478977 https://hal.archives-ouvertes.fr/hal-00478977 Submitted on 30 Apr 2010 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Biochemical Journal Immediate Publication. Published on 21 Apr 2008 as manuscript BJ20080353 Elimination of competing hydrolysis and coupling side reactions of a cyclodextrin glucanotransferase by directed evolution Ronan M. Kelly1, Hans Leemhuis1, Henriëtte J. Rozeboom2, Niels van Oosterwijk2, Bauke W. Dijkstra2 and Lubbert Dijkhuizen1* 1Microbial Physiology, Groningen Biomolecular Sciences and Biotechnology Institute, University of Groningen, Kerklaan 30, 9751 NN Haren, The Netherlands, and Centre for Carbohydrate Bioprocessing, TNO-University of Groningen, Kerklaan 30, 9751 NN Haren, The Netherlands. 2Laboratory of Biophysical Chemistry, Groningen Biomolecular Sciences and Biotechnology Institute, University of Groningen, Nijenborgh 4, 9747 AG Groningen, The Netherlands. -

Review Article Pullulanase: Role in Starch Hydrolysis and Potential Industrial Applications
Hindawi Publishing Corporation Enzyme Research Volume 2012, Article ID 921362, 14 pages doi:10.1155/2012/921362 Review Article Pullulanase: Role in Starch Hydrolysis and Potential Industrial Applications Siew Ling Hii,1 Joo Shun Tan,2 Tau Chuan Ling,3 and Arbakariya Bin Ariff4 1 Department of Chemical Engineering, Faculty of Engineering and Science, Universiti Tunku Abdul Rahman, 53300 Kuala Lumpur, Malaysia 2 Institute of Bioscience, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia 3 Institute of Biological Sciences, Faculty of Science, University of Malaya, 50603 Kuala Lumpur, Malaysia 4 Department of Bioprocess Technology, Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia Correspondence should be addressed to Arbakariya Bin Ariff, [email protected] Received 26 March 2012; Revised 12 June 2012; Accepted 12 June 2012 Academic Editor: Joaquim Cabral Copyright © 2012 Siew Ling Hii et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. The use of pullulanase (EC 3.2.1.41) has recently been the subject of increased applications in starch-based industries especially those aimed for glucose production. Pullulanase, an important debranching enzyme, has been widely utilised to hydrolyse the α-1,6 glucosidic linkages in starch, amylopectin, pullulan, and related oligosaccharides, which enables a complete and efficient conversion of the branched polysaccharides into small fermentable sugars during saccharification process. The industrial manufacturing of glucose involves two successive enzymatic steps: liquefaction, carried out after gelatinisation by the action of α- amylase; saccharification, which results in further transformation of maltodextrins into glucose. -
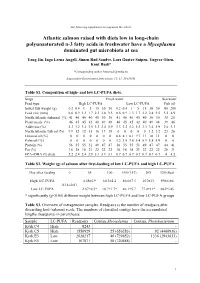
Q 297 Suppl USE
The following supplement accompanies the article Atlantic salmon raised with diets low in long-chain polyunsaturated n-3 fatty acids in freshwater have a Mycoplasma dominated gut microbiota at sea Yang Jin, Inga Leena Angell, Simen Rød Sandve, Lars Gustav Snipen, Yngvar Olsen, Knut Rudi* *Corresponding author: [email protected] Aquaculture Environment Interactions 11: 31–39 (2019) Table S1. Composition of high- and low LC-PUFA diets. Stage Fresh water Sea water Feed type High LC-PUFA Low LC-PUFA Fish oil Initial fish weight (g) 0.2 0.4 1 5 15 30 50 0.2 0.4 1 5 15 30 50 80 200 Feed size (mm) 0.6 0.9 1.3 1.7 2.2 2.8 3.5 0.6 0.9 1.3 1.7 2.2 2.8 3.5 3.5 4.9 North Atlantic fishmeal (%) 41 40 40 40 40 30 30 41 40 40 40 40 30 30 35 25 Plant meals (%) 46 45 45 42 40 49 48 46 45 45 42 40 49 48 39 46 Additives (%) 3.3 3.2 3.2 3.5 3.3 3.4 3.9 3.3 3.2 3.2 3.5 3.3 3.4 3.9 2.6 3.3 North Atlantic fish oil (%) 9.9 12 12 15 16 17 18 0 0 0 0 0 1.2 1.2 23 26 Linseed oil (%) 0 0 0 0 0 0 0 6.8 8.1 8.1 9.7 11 10 11 0 0 Palm oil (%) 0 0 0 0 0 0 0 3.2 3.8 3.8 5.4 5.9 5.8 5.9 0 0 Protein (%) 56 55 55 51 49 47 47 56 55 55 51 49 47 47 44 41 Fat (%) 16 18 18 21 22 22 22 16 18 18 21 22 22 22 28 31 EPA+DHA (% diet) 2.2 2.4 2.4 2.9 3.1 3.1 3.1 0.7 0.7 0.7 0.7 0.7 0.7 0.7 4 4.2 Table S2. -

Biosynthesis and Engineering of Cyclomarin and Cyclomarazine: Prenylated, Non-Ribosomal Cyclic Peptides of Marine Actinobacterial Origin
UC San Diego Research Theses and Dissertations Title Biosynthesis and Engineering of Cyclomarin and Cyclomarazine: Prenylated, Non-Ribosomal Cyclic Peptides of Marine Actinobacterial Origin Permalink https://escholarship.org/uc/item/21b965z8 Author Schultz, Andrew W. Publication Date 2010 Peer reviewed eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA, SAN DIEGO Biosynthesis and Engineering of Cyclomarin and Cyclomarazine: Prenylated, Non-Ribosomal Cyclic Peptides of Marine Actinobacterial Origin A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Oceanography by Andrew William Schultz Committee in charge: Professor Bradley Moore, Chair Professor Eric Allen Professor Pieter Dorrestein Professor William Fenical Professor William Gerwick 2010 Copyright Andrew William Schultz, 2010 All rights reserved. The Dissertation of Andrew William Schultz is approved, and it is acceptable in quality and form for publication on microfilm and electronically: ________________________________________________________________ ________________________________________________________________ ________________________________________________________________ Chair University of California, San Diego 2010 iii DEDICATION To my wife Elizabeth and our son Orion and To my parents Dale and Mary Thank you for your never ending love and support iv TABLE OF CONTENTS Signature Page .................................................................................................... -

STUDY of STARCH DEBRANCHING ENZYMES in DEVELOPING WHEAT KERNELS a Thesis Submitted to the College of Graduate Studies and Resear
STUDY OF STARCH DEBRANCHING ENZYMES IN DEVELOPING WHEAT KERNELS A Thesis Submitted to the College of Graduate Studies and Research in Partial Fulfilment of the Requirements for the Degree of Doctor of Philosophy in the Department of Biochemistry University of Saskatchewan Saskatoon By Supatcharee Netrphan Spring 2002 © Copyright Supatcharee Netrphan, 2002. All rights reserved. PERMISSION TO USE In presenting this thesis in partial fulfilment of the requirements for a Postgraduate degree from the University of Saskatchewan, I agree that the Libraries of this University may make it freely available for inspection. I further agree that permission for copying of this thesis in any manner, in whole or in part, for scholarly purposes may be granted by the professor or professors who supervised my thesis work or, in their absence, by the Head of the Department or the Dean of the College in which my thesis work was done. It is understood that any copying or publication or use of this thesis or parts thereof for financial gain shall not be allowed without my written permission. It is also understood that due recognition shall be given to me and to the University of Saskatchewan in any scholarly use which may be made of any material in my thesis. Requests for permission to copy or to make other use of material in this thesis in whole or in part should be addressed to: Head of the Department of Biochemistry University of Saskatchewan 107 Wiggins Road Saskatoon, Saskatchewan S7N 5E5 i ABSTRACT Starch debranching enzymes, which specifically hydrolyse a-1,6 glucosidic bonds in glucans containing both a-1,4 and a-1,6 linkages, are classified into two types: isoamylase (EC. -

Neopullulanase 13A, Geobacillus Stearothermophilus Gsnep13a
Neopullulanase 13A, Geobacillus stearothermophilus Gs Nep13A (CBM34-GH13) Catalogue number: CZ08671 , 0,5 mg CZ08672 , 3 × 0,5 mg Description Storage temperature Gs Nep13A (CBM34-GH13), E.C. number 3.2.1.135, is an enzyme This enzyme should be stored at -20 °C. that participates in the hydrolysis of pullulan to panose (6-α-D- glucosylmaltose) from Geobacillus stearothermophilus . Recombinant Gs Nep13A (CBM34-GH13), purified from Escherichia Substrate specificity coli , is a modular family 13 Glycoside Hydrolase (GH13) with an N- terminal family 34 Carbohydrate Binding Module (CBM34) Gs Nep13A (CBM34-GH13) hydrolyses β-CD, starch and pullulan. (www.cazy.org). The enzyme is provided in 35 mM NaHepes buffer, pH 7.5, 750 mM NaCl, 200 mM imidazol, 3.5 mM CaCl 2 and 25% (v/v) glycerol, at a 0,5 mg/mL concentration. Bulk quantities Temperature and pH optima of this product are available on request. The pH optimum for enzymatic activity is 6 while temperature optimum is 55 °C. Electrophoretic Purity Gs Nep13A (CBM34-GH13) purity was determined by sodium Enzyme activity dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) followed by BlueSafe staining (MB15201) (Figure 1). Substrate specificity and kinetic properties of Gs Nep13A (CBM34- GH13) are described in the reference provided below. Follow the instructions described in the paper for the implementation of enzyme assays and to obtain values of specific activity. To measure catalytic activity of GHs, quantify reducing sugars released from polysaccharides through the method described by Miller (1959; Anal. Chem. 31, 426-428). Reference Cheong et al. Biotechnol Appl Biochem. 2002 Feb.35(Pt 1):27-34.