Unit 3.3 Metric System (System International)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Toolkit 1: Energy Units and Fundamentals of Quantitative Analysis
Energy & Society Energy Units and Fundamentals Toolkit 1: Energy Units and Fundamentals of Quantitative Analysis 1 Energy & Society Energy Units and Fundamentals Table of Contents 1. Key Concepts: Force, Work, Energy & Power 3 2. Orders of Magnitude & Scientific Notation 6 2.1. Orders of Magnitude 6 2.2. Scientific Notation 7 2.3. Rules for Calculations 7 2.3.1. Multiplication 8 2.3.2. Division 8 2.3.3. Exponentiation 8 2.3.4. Square Root 8 2.3.5. Addition & Subtraction 9 3. Linear versus Exponential Growth 10 3.1. Linear Growth 10 3.2. Exponential Growth 11 4. Uncertainty & Significant Figures 14 4.1. Uncertainty 14 4.2. Significant Figures 15 4.3. Exact Numbers 15 4.4. Identifying Significant Figures 16 4.5. Rules for Calculations 17 4.5.1. Addition & Subtraction 17 4.5.2. Multiplication, Division & Exponentiation 18 5. Unit Analysis 19 5.1. Commonly Used Energy & Non-energy Units 20 5.2. Form & Function 21 6. Sample Problems 22 6.1. Scientific Notation 22 6.2. Linear & Exponential Growth 22 6.3. Significant Figures 23 6.4. Unit Conversions 23 7. Answers to Sample Problems 24 7.1. Scientific Notation 24 7.2. Linear & Exponential Growth 24 7.3. Significant Figures 24 7.4. Unit Conversions 26 8. References 27 2 Energy & Society Energy Units and Fundamentals 1. KEY CONCEPTS: FORCE, WORK, ENERGY & POWER Among the most important fundamentals to be mastered when studying energy pertain to the differences and inter-relationships among four concepts: force, work, energy, and power. Each of these terms has a technical meaning in addition to popular or colloquial meanings. -

Lord Kelvin and the Age of the Earth.Pdf
ME201/MTH281/ME400/CHE400 Lord Kelvin and the Age of the Earth Lord Kelvin (1824 - 1907) 1. About Lord Kelvin Lord Kelvin was born William Thomson in Belfast Ireland in 1824. He attended Glasgow University from the age of 10, and later took his BA at Cambridge. He was appointed Professor of Natural Philosophy at Glasgow in 1846, a position he retained the rest of his life. He worked on a broad range of topics in physics, including thermody- namics, electricity and magnetism, hydrodynamics, atomic physics, and earth science. He also had a strong interest in practical problems, and in 1866 he was knighted for his work on the transtlantic cable. In 1892 he became Baron Kelvin, and this name survives as the name of the absolute temperature scale which he proposed in 1848. During his long career, Kelvin published more than 600 papers. He was elected to the Royal Society in 1851, and served as president of that organization from 1890 to 1895. The information in this section and the picture above were taken from a very useful web site called the MacTu- tor History of Mathematics Archive, sponsored by St. Andrews University. The web address is http://www-history.mcs.st-and.ac.uk/~history/ 2 kelvin.nb 2. The Age of the Earth The earth shows it age in many ways. Some techniques for estimating this age require us to observe the present state of a time-dependent process, and from that observation infer the time at which the process started. If we believe that the process started when the earth was formed, we get an estimate of the earth's age. -

Guide for the Use of the International System of Units (SI)
Guide for the Use of the International System of Units (SI) m kg s cd SI mol K A NIST Special Publication 811 2008 Edition Ambler Thompson and Barry N. Taylor NIST Special Publication 811 2008 Edition Guide for the Use of the International System of Units (SI) Ambler Thompson Technology Services and Barry N. Taylor Physics Laboratory National Institute of Standards and Technology Gaithersburg, MD 20899 (Supersedes NIST Special Publication 811, 1995 Edition, April 1995) March 2008 U.S. Department of Commerce Carlos M. Gutierrez, Secretary National Institute of Standards and Technology James M. Turner, Acting Director National Institute of Standards and Technology Special Publication 811, 2008 Edition (Supersedes NIST Special Publication 811, April 1995 Edition) Natl. Inst. Stand. Technol. Spec. Publ. 811, 2008 Ed., 85 pages (March 2008; 2nd printing November 2008) CODEN: NSPUE3 Note on 2nd printing: This 2nd printing dated November 2008 of NIST SP811 corrects a number of minor typographical errors present in the 1st printing dated March 2008. Guide for the Use of the International System of Units (SI) Preface The International System of Units, universally abbreviated SI (from the French Le Système International d’Unités), is the modern metric system of measurement. Long the dominant measurement system used in science, the SI is becoming the dominant measurement system used in international commerce. The Omnibus Trade and Competitiveness Act of August 1988 [Public Law (PL) 100-418] changed the name of the National Bureau of Standards (NBS) to the National Institute of Standards and Technology (NIST) and gave to NIST the added task of helping U.S. -

Chapter 1, Section
Chapter 1 Chemical Foundations 1.3 Units of Measurement Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 1 Measurement You make a measurement every time you • measure your height. • read your watch. • take your temperature. • weigh a cantaloupe. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 2 Measurement in Chemistry In chemistry we • measure quantities. • do experiments. • calculate results. • use numbers to report measurements. • compare results to standards. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 3 Measurement In a measurement • a measuring tool is used to compare some dimension of an object to a standard. • of the thickness of the skin fold at the waist, calipers are used. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 4 Stating a Measurement In every measurement, a number is followed by a unit. Observe the following examples of measurements: Number and Unit 35 m 0.25 L 225 lb 3.4 hr 5 The Metric System (SI) The metric system or SI (international system) is • a decimal system based on 10. • used in most of the world. • used everywhere by scientists. 6 The 7 Basic Fundamental SI Units Physical Quantity Name of Unit Abbreviation Mass kilogram kg Length meter m Time second s Temperature kelvin K Electric current ampere A Amount of substance mole mol Luminous intensity candela cd 7 Units in the Metric System In the metric and SI systems, one unit is used for each type of measurement: Measurement Metric SI Length meter (m) meter (m) Volume liter (L) cubic meter (m3) Mass gram (g) kilogram (kg) Time second (s) second (s) Temperature Celsius (°C) Kelvin (K) 8 Length Measurement Length • is measured using a meter stick. -

Measuring in Metric Units BEFORE Now WHY? You Used Metric Units
Measuring in Metric Units BEFORE Now WHY? You used metric units. You’ll measure and estimate So you can estimate the mass using metric units. of a bike, as in Ex. 20. Themetric system is a decimal system of measurement. The metric Word Watch system has units for length, mass, and capacity. metric system, p. 80 Length Themeter (m) is the basic unit of length in the metric system. length: meter, millimeter, centimeter, kilometer, Three other metric units of length are themillimeter (mm) , p. 80 centimeter (cm) , andkilometer (km) . mass: gram, milligram, kilogram, p. 81 You can use the following benchmarks to estimate length. capacity: liter, milliliter, kiloliter, p. 82 1 millimeter 1 centimeter 1 meter thickness of width of a large height of the a dime paper clip back of a chair 1 kilometer combined length of 9 football fields EXAMPLE 1 Using Metric Units of Length Estimate the length of the bandage by imagining paper clips laid next to it. Then measure the bandage with a metric ruler to check your estimate. 1 Estimate using paper clips. About 5 large paper clips fit next to the bandage, so it is about 5 centimeters long. ch O at ut! W 2 Measure using a ruler. A typical metric ruler allows you to measure Each centimeter is divided only to the nearest tenth of into tenths, so the bandage cm 12345 a centimeter. is 4.8 centimeters long. 80 Chapter 2 Decimal Operations Mass Mass is the amount of matter that an object has. The gram (g) is the basic metric unit of mass. -

About the Nature of Gravitational Constant and a Rational Metric System
See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/303862772 About the Nature of Gravitational Constant and a Rational Metric System. Working Paper · September 2002 DOI: 10.13140/RG.2.1.1992.9205 1 author: Andrew Wutke Thales Group 6 PUBLICATIONS 1 CITATION SEE PROFILE Available from: Andrew Wutke Retrieved on: 04 July 2016 DRAF"T About The Nature of G6pffi Constant and Rational Metric SYstem Andrew Wutke SYdneY Australia email andreww @ optus hame. c om. au 15 september,2002 "We are to adtnit no mare causes of natural things that such as are both true and sufficient to explain their appearance*" INewon] Abstract The gravitational constant G has been a sutrject of interest for more than two centuries. Precise measurements indicate #bqual to 6'673(I0)xl0'r m3i1tg s21 with relative standard uncertainty of 1.5x10-3[U. The need fcrr such constant is discussed. Various s)6tem of units of measure have emerged since Newton, and none of them is both piaaical, and usefid in thesetical rqsearch. The relevance ofa metric system that only Las length and time as base units is analysed and such system proposed. As a rssult much highm clarity of physical quantitie.s is achieved. Grmi$d;onstant and electric constant can be eliminated to become nondimensional, that results in elimination of of the sarre composite unit for both kilogram of mass and Coulomb 9f charge in fuvour physical quantities being m3/s2. The system offers a great advantage to General Relativity where only space-time units can be used. -

UNITS This Appendix Explains Some of the Abbreviations1•2 Used For
APPENDIX: UNITS This appendix explains some of the abbreviations1•2 used for units in this book and gives conversion factors to SI units and atomic units: length 1 a0 = 1 bohr = 0.5291771 X 10-10 m 1 A= 1 Angstrom= lo-10 m = 1.889727 ao mass 1 me = 1 atomic unit of mass = mass of an electron 9.109534 X 10-31 kg = 5.485803 X 10-4 U 1 u 1 universal atomic mass unit = one twelfth the mass of a 12c atom 1.6605655 x lo-27 kg = 1822.887 me time 1 t Eh 1 = 1 atomic unit of time = 2.418884 x l0-17 s 1 s = 1 second = 4.134137 x 1016 t/Eh temperature 1 K = 1 Kelvin amount of substance 1 mol = 1 mole 6.022045 x 1023 atoms, molecules, or formula units energy 1 cm-1 = 1 wavenumber 1 kayser 1.986477 x lo-23 J 4.556335 x 10-6 Eh 857 858 APPENDIX: UNITS 1 kcal/mol = 1 kilocalorie per mole 4.184 kJ/mol = 1.593601 x 10-3 Eh 1 eV 1 electron volt = 1.602189 x lo-19 J 3.674902 X 10-2 Eh 1 Eh 1 hartree = 4.359814 x lo-18 J Since so many different energy units are used in the book, it is helpful to have a conversion table. Such a table was calculated from the recommended values of Cohen and Taylor3 for the physical censtants and is given in Table 1. REFERENCES 1. "Standard for Metric Practice", American Society for Testing and Materials, Philadelphia (1976). -

The Kelvin and Temperature Measurements
Volume 106, Number 1, January–February 2001 Journal of Research of the National Institute of Standards and Technology [J. Res. Natl. Inst. Stand. Technol. 106, 105–149 (2001)] The Kelvin and Temperature Measurements Volume 106 Number 1 January–February 2001 B. W. Mangum, G. T. Furukawa, The International Temperature Scale of are available to the thermometry commu- K. G. Kreider, C. W. Meyer, D. C. 1990 (ITS-90) is defined from 0.65 K nity are described. Part II of the paper Ripple, G. F. Strouse, W. L. Tew, upwards to the highest temperature measur- describes the realization of temperature able by spectral radiation thermometry, above 1234.93 K for which the ITS-90 is M. R. Moldover, B. Carol Johnson, the radiation thermometry being based on defined in terms of the calibration of spec- H. W. Yoon, C. E. Gibson, and the Planck radiation law. When it was troradiometers using reference blackbody R. D. Saunders developed, the ITS-90 represented thermo- sources that are at the temperature of the dynamic temperatures as closely as pos- equilibrium liquid-solid phase transition National Institute of Standards and sible. Part I of this paper describes the real- of pure silver, gold, or copper. The realiza- Technology, ization of contact thermometry up to tion of temperature from absolute spec- 1234.93 K, the temperature range in which tral or total radiometry over the tempera- Gaithersburg, MD 20899-0001 the ITS-90 is defined in terms of calibra- ture range from about 60 K to 3000 K is [email protected] tion of thermometers at 15 fixed points and also described. -
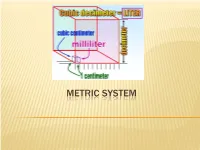
Metric System.Pdf
METRIC SYSTEM THE METRIC SYSTEM The metric system is much easier. All metric units are related by factors of 10. Nearly the entire world (95%), except the United States, now uses the metric system. Metric is used exclusively in science. Because the metric system uses units related by factors of ten and the types of units (distance, area, volume, mass) are simply-related, performing calculations with the metric system is much easier. METRIC CHART Prefix Symbol Factor Number Factor Word Kilo K 1,000 Thousand Hecto H 100 Hundred Deca Dk 10 Ten Base Unit Meter, gram, liter 1 One Deci D 0.1 Tenth Centi C 0.01 Hundredth Milli M 0.001 Thousandth The metric system has three units or bases. Meter – the basic unit used to measure length Gram – the basic unit used to measure weight Liter – the basic unit used to measure liquid capacity (think 2 Liter cokes!) The United States, Liberia and Burma (countries in black) have stuck with using the Imperial System of measurement. You can think of “the metric system” as a nickname for the International System of Units, or SI. HOW TO REMEMBER THE PREFIXES Kids Kilo Have Hecto Dropped Deca Over base unit (gram, liter, meter) Dead Deci Converting Centi Metrics Milli Large Units – Kilo (1000), Hecto (100), Deca (10) Small Units – Deci (0.1), Centi (0.01), Milli (0.001) Because you are dealing with multiples of ten, you do not have to calculate anything. All you have to do is move the decimal point, but you need to understand what you are doing when you move the decimal point. -

Kelvin Color Temperature
KELVIN COLOR TEMPERATURE William Thompson Kelvin was a 19th century physicist and mathematician who invented a temperature scale that had absolute zero as its low endpoint. In physics, absolute zero is a very cold temperature, the coldest possible, at which no heat exists and kinetic energy (movement) ceases. On the Celsius scale absolute zero is -273 degrees, and on the Fahrenheit scale it is -459 degrees. The Kelvin temperature scale is often used for scientific measurements. Kelvins, as the degrees are now called, are derived from the actual temperature of a black body radiator, which means a black material heated to that temperature. An incandescent filament is very dark, and approaches being a black body radiator, so the actual temperature of an incandescent filament is somewhat close to its color temperature in Kelvins. The color temperature of a lamp is very important in the television industry where the camera must be calibrated for white balance. This is often done by focusing the camera on a white card in the available lighting and tweaking it so that the card reads as true white. All other colors will automatically adjust so that they read properly. This is especially important to reproduce “normal” looking skin tones. In theatre applications, where it is only important for colors to read properly to the human eye, the exact color temperature of lamps is not so important. Incandescent lamps tend to have a color temperature around 3200 K, but this is true only if they are operating with full voltage. Remember that dimmers work by varying the voltage pressure supplied to the lamp. -

The International System of Units (SI) - Conversion Factors For
NIST Special Publication 1038 The International System of Units (SI) – Conversion Factors for General Use Kenneth Butcher Linda Crown Elizabeth J. Gentry Weights and Measures Division Technology Services NIST Special Publication 1038 The International System of Units (SI) - Conversion Factors for General Use Editors: Kenneth S. Butcher Linda D. Crown Elizabeth J. Gentry Weights and Measures Division Carol Hockert, Chief Weights and Measures Division Technology Services National Institute of Standards and Technology May 2006 U.S. Department of Commerce Carlo M. Gutierrez, Secretary Technology Administration Robert Cresanti, Under Secretary of Commerce for Technology National Institute of Standards and Technology William Jeffrey, Director Certain commercial entities, equipment, or materials may be identified in this document in order to describe an experimental procedure or concept adequately. Such identification is not intended to imply recommendation or endorsement by the National Institute of Standards and Technology, nor is it intended to imply that the entities, materials, or equipment are necessarily the best available for the purpose. National Institute of Standards and Technology Special Publications 1038 Natl. Inst. Stand. Technol. Spec. Pub. 1038, 24 pages (May 2006) Available through NIST Weights and Measures Division STOP 2600 Gaithersburg, MD 20899-2600 Phone: (301) 975-4004 — Fax: (301) 926-0647 Internet: www.nist.gov/owm or www.nist.gov/metric TABLE OF CONTENTS FOREWORD.................................................................................................................................................................v -

SI Base Units
463 Appendix I SI base units 1 THE SEVEN BASE UNITS IN THE INTERNatioNAL SYSTEM OF UNITS (SI) Quantity Name of Symbol base SI Unit Length metre m Mass kilogram kg Time second s Electric current ampere A Thermodynamic temperature kelvin K Amount of substance mole mol Luminous intensity candela cd 2 SOME DERIVED SI UNITS WITH THEIR SYMBOL/DerivatioN Quantity Common Unit Symbol Derivation symbol Term Term Length a, b, c metre m SI base unit Area A square metre m² Volume V cubic metre m³ Mass m kilogram kg SI base unit Density r (rho) kilogram per cubic metre kg/m³ Force F newton N 1 N = 1 kgm/s2 Weight force W newton N 9.80665 N = 1 kgf Time t second s SI base unit Velocity v metre per second m/s Acceleration a metre per second per second m/s2 Frequency (cycles per second) f hertz Hz 1 Hz = 1 c/s Bending moment (torque) M newton metre Nm Pressure P, F newton per square metre Pa (N/m²) 1 MN/m² = 1 N/mm² Stress σ (sigma) newton per square metre Pa (N/m²) Work, energy W joule J 1 J = 1 Nm Power P watt W 1 W = 1 J/s Quantity of heat Q joule J Thermodynamic temperature T kelvin K SI base unit Specific heat capacity c joule per kilogram degree kelvin J/ kg × K Thermal conductivity k watt per metre degree kelvin W/m × K Coefficient of heat U watt per square metre kelvin w/ m² × K 464 Rural structures in the tropics: design and development 3 MUltiples AND SUB MUltiples OF SI–UNITS COMMONLY USED IN CONSTRUCTION THEORY Factor Prefix Symbol 106 mega M 103 kilo k (102 hecto h) (10 deca da) (10-1 deci d) (10-2 centi c) 10-3 milli m 10-6 micro u Prefix in brackets should be avoided.