Medication Audit Criteria and Guidelines: Antipsychotics-Clozapine (Clozaril®, Fazaclo®)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Prochlorperazine 5Mg Tablets
Package leaflet: Information for the patient Prochlorperazine 5mg tablets Read all of this leaflet carefully before you start taking this • the person is a child. This is because children may develop unusual face and body medicine, because it contains important information for you. movements (dystonic reactions) • Keep this leaflet. You may need to read it again. • you are diabetic or have high levels of sugar in your blood (hyperglycaemia). Your doctor • If you have any further questions, ask your doctor or pharmacist. may want to monitor you more closely. • This medicine has been prescribed for you only. Do not pass it on to If you are not sure if any of the above apply to you, talk to your doctor or pharmacist before others. It may harm them, even if their signs of illness are the same taking Prochlorperazine Tablets. as yours. Other medicines and Prochlorperazine tablets • If you get any side effects, talk to your doctor or pharmacist. Tell your doctor or pharmacist if you are taking, have recently taken or might take any This includes any possible side effects not listed in this leaflet. other medicines. This includes medicines you buy without a prescription, including herbal See section 4. medicines. This is because Prochlorperazine Tablets can affect the way some other medicines work. What is in this leaflet: Also some medicines can affect the way Prochlorperazine Tablets work. 1 What Prochlorperazine tablets are and what they In particular, tell your doctor if you are taking any of the following: • medicines to help you sleep -
Medication Conversion Chart
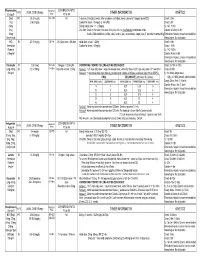
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Schizophrenia Care Guide
August 2015 CCHCS/DHCS Care Guide: Schizophrenia SUMMARY DECISION SUPPORT PATIENT EDUCATION/SELF MANAGEMENT GOALS ALERTS Minimize frequency and severity of psychotic episodes Suicidal ideation or gestures Encourage medication adherence Abnormal movements Manage medication side effects Delusions Monitor as clinically appropriate Neuroleptic Malignant Syndrome Danger to self or others DIAGNOSTIC CRITERIA/EVALUATION (PER DSM V) 1. Rule out delirium or other medical illnesses mimicking schizophrenia (see page 5), medications or drugs of abuse causing psychosis (see page 6), other mental illness causes of psychosis, e.g., Bipolar Mania or Depression, Major Depression, PTSD, borderline personality disorder (see page 4). Ideas in patients (even odd ideas) that we disagree with can be learned and are therefore not necessarily signs of schizophrenia. Schizophrenia is a world-wide phenomenon that can occur in cultures with widely differing ideas. 2. Diagnosis is made based on the following: (Criteria A and B must be met) A. Two of the following symptoms/signs must be present over much of at least one month (unless treated), with a significant impact on social or occupational functioning, over at least a 6-month period of time: Delusions, Hallucinations, Disorganized Speech, Negative symptoms (social withdrawal, poverty of thought, etc.), severely disorganized or catatonic behavior. B. At least one of the symptoms/signs should be Delusions, Hallucinations, or Disorganized Speech. TREATMENT OPTIONS MEDICATIONS Informed consent for psychotropic -

Management of Major Depressive Disorder Clinical Practice Guidelines May 2014
Federal Bureau of Prisons Management of Major Depressive Disorder Clinical Practice Guidelines May 2014 Table of Contents 1. Purpose ............................................................................................................................................. 1 2. Introduction ...................................................................................................................................... 1 Natural History ................................................................................................................................. 2 Special Considerations ...................................................................................................................... 2 3. Screening ........................................................................................................................................... 3 Screening Questions .......................................................................................................................... 3 Further Screening Methods................................................................................................................ 4 4. Diagnosis ........................................................................................................................................... 4 Depression: Three Levels of Severity ............................................................................................... 4 Clinical Interview and Documentation of Risk Assessment............................................................... -

Appendix 18: References to Studies from the Previous Guideline
Appendix 18: References to studies from the previous guideline This appendix contains references to the studies from the previous guideline, as they appeared in that guideline. References to studies added to the guideline update are in Appendix 17a-d. Contents Service references…………………………………………………………………..1 Psychology references……………………………………………………………..15 Pharmacology references………………………………………………………….28 Service references Araya2003 {published data only} Araya R, Rojas G, Fritsch R, Gaete J, Rojas M, Simon G, Peters TJ. Treating depression in primary care in low-income women in Santiago, Chile: A randomised controlled trial. Lancet 2003;361(9362):995-1000. Arthur2002 {published data only} Arthur AJ, Jagger C, Lindesay J, Matthews RJ. Evaluating a mental health assessment for older people with depressive symptoms in general practice: a randomised controlled trial. British Journal of General Practice 2002;52(476):202-207. Austin-Los Angeles Austin NK, Liberman RP, King LW, DeRisi WJ. A comparative evaluation of two day hospitals. Goal attainment scaling of behaviour therapy vs. milieu therapy. Journal of Nervous and Mental Disease 1976;163:253-62. Azim-Alberta Azim HF, Weiden TD, Ratcliffe WD, Nutter RW, Dyck RJ, Howarth BG. Current utilization of day hospitalization. Canadian Psychiatric Association Journal 1978;23:557-66 Baker2001 {published data only} Baker R, Reddish S, Robertson N, Hearnshaw H, Jones B. Randomised controlled trial of tailored strategies to implement guidelines for the management of patients with depression in general practice. British Journal of General Practice 2001;51:737-741. Barkley-Ontario Barkley AL, Fagen K, Lawson JS. Day care: can it prevent readmission to a psychiatric hospital? Psychiatric Journal of the University of Ottawa 1989;14:536-41. -

Louisiana Fee-For-Service Medicaid Antipsychotics
Louisiana Fee-for-Service Medicaid Antipsychotics The Louisiana Uniform Prescription Drug Prior Authorization Form should be utilized to request: Authorization for non-preferred agents for recipients 6 years of age and older; AND Authorization for all preferred and non-preferred agents for recipients younger than 6 years of age; AND Authorization to exceed maximum daily dose/quantity limit for all ages. See full prescribing information for individual agents for details on the information below: *These agents have Black Box Warnings †These agents are subject to Risk Evaluation and Mitigation Strategy (REMS) under FDA safety regulations ‡ For long-acting injectable agents, it is required that the previous 60-day period of pharmacy claims show one of the following: Established tolerance to the oral formulation (as evidenced by a paid pharmacy claim for the oral formulation); OR Established therapy with the requested injectable agent (as evidenced by a paid pharmacy claim for the requested injectable agent) NOTE: Diagnosis code requirements apply to both preferred and non-preferred agents (see Table 1). Maximum daily dose edits (see Table 2), quantity limits (see Table 3), and other requirements at Point-of-Sale for select agents in this category may apply to both preferred and non-preferred agents. For additional information, see http://www.lamedicaid.com/provweb1/Pharmacy/pharmacyindex.htm. Oral Antipsychotics – Generic Name (Brand Example) * Amitriptyline/Perphenazine * Aripiprazole ODT; Oral Solution (Abilify®); Tablet (Abilify®) -

Medications to Be Avoided Or Used with Caution in Parkinson's Disease
Medications To Be Avoided Or Used With Caution in Parkinson’s Disease This medication list is not intended to be complete and additional brand names may be found for each medication. Every patient is different and you may need to take one of these medications despite caution against it. Please discuss your particular situation with your physician and do not stop any medication that you are currently taking without first seeking advice from your physician. Most medications should be tapered off and not stopped suddenly. Although you may not be taking these medications at home, one of these medications may be introduced while hospitalized. If a hospitalization is planned, please have your neurologist contact your treating physician in the hospital to advise which medications should be avoided. Medications to be avoided or used with caution in combination with Selegiline HCL (Eldepryl®, Deprenyl®, Zelapar®), Rasagiline (Azilect®) and Safinamide (Xadago®) Medication Type Medication Name Brand Name Narcotics/Analgesics Meperidine Demerol® Tramadol Ultram® Methadone Dolophine® Propoxyphene Darvon® Antidepressants St. John’s Wort Several Brands Muscle Relaxants Cyclobenzaprine Flexeril® Cough Suppressants Dextromethorphan Robitussin® products, other brands — found as an ingredient in various cough and cold medications Decongestants/Stimulants Pseudoephedrine Sudafed® products, other Phenylephrine brands — found as an ingredient Ephedrine in various cold and allergy medications Other medications Linezolid (antibiotic) Zyvox® that inhibit Monoamine oxidase Phenelzine Nardil® Tranylcypromine Parnate® Isocarboxazid Marplan® Note: Additional medications are cautioned against in people taking Monoamine oxidase inhibitors (MAOI), including other opioids (beyond what is mentioned in the chart above), most classes of antidepressants and other stimulants (beyond what is mentioned in the chart above). -

Stemzine® (Prochlorperazine Maleate) Tablet
AUSTRALIAN PRODUCT INFORMATION – STEMZINE® (PROCHLORPERAZINE MALEATE) TABLET 1 NAME OF THE MEDICINE Prochlorperazine maleate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Stemzine contains 5mg prochlorperazine maleate as the active ingredient. Excipients of known effect: wheat starch (gluten). For the full list of excipients, see Section 6.1 List of excipients. 3 PHARMACEUTICAL FORM Stemzine 5 mg tablets are off-white to pale cream coloured circular tablets, not more than slightly mottled or specked, one side impressed with 'S' and reverse face plain. 4 CLINICAL PARTICULARS 4.1 THERAPEUTIC INDICATIONS Nausea and vomiting due to various causes including migraine; vertigo due to Meniere's syndrome, labyrinthitis and other causes. 4.2 DOSE AND METHOD OF ADMINISTRATION Nausea and vomiting Adults Dosage should be adjusted to suit the response of the individual, beginning with lowest recommended dosage. Oral: 5 or 10 mg two or three times daily. Acute: 20 mg at once, followed, if necessary by 10 mg two hours later. Children (See Section 4.4 Special warnings and precautions for use, Paediatric Use). stem-ccsiv5-piv19-04jun21 Page 1 of 16 If it is considered unavoidable to use prochlorperazine for a child, the dosage is 250 micrograms/kg bodyweight two or three times a day. Prochlorperazine has been associated with dystonic reactions particularly after a cumulative dosage of 500 micrograms/kg. It should therefore be used cautiously in children. Prochlorperazine is not recommended for children weighing less than 10 kg. When treating children, it is recommended that the 5 mg tablets are used. Vertigo and meniere’s disease Adults Oral: 5 to 10 mg three or four times daily. -

Long-Acting Injectable (LAI) Dosing Chart
Long-Acting Injectable (LAI) Dosing Chart Drug FDA-Approved Dosing Earliest Time to Missed Dose Indications Next Dose ABILIFY • Schizophrenia Initiation No sooner than 26 days If 2nd or 3rd dose missed: MAINTENA® • Maintenance 400 mg IM q month + aripiprazole 10-20 mg after last injection Timing of Missed Dose Dosing (aripiprazole)1 Monotherapy of PO daily x14 days OR if stable on another PO >4 weeks and <5 weeks • Administer missed dose ASAP. Bipolar 1 Disorder antipsychotic, + overlap x14 days >5 weeks • Restart next injection with oral Dosage Forms aripiprazole x 14 days. 300 mg and 400 mg Maintenance pre-filled syringe or 400 mg IM q month If 4th or subsequent doses missed: single-use vial Timing of Missed Dose Dosing Dose Adjustments >4 weeks and <6 weeks • Administer missed dose ASAP. Adverse reactions or known CYP2D6 poor >5 weeks • Restart next injection with oral metabolizers: aripiprazole x 14 days. 300 mg IM q month ARISTADA INITIO®, • Schizophrenia Initiation No sooner than 14 days ARISTADA® Option #1: ARISTADA INITIO® 675 mg IM x1 after last injection Management of a Missed Maintenance Dose (aripiprazole dose + aripiprazole 30 mg PO x1 dose + first Last Time Since Last Injection lauroxil)2,3 dose of *ARISTADA® IM (441 mg, 662 mg, 882 ARISTADA® mg, or 1064 mg) Dose Dosage Forms *first dose of ARISTADA® may be given 441 mg ≤6 weeks >6 and ≤7 weeks >7 weeks ARISTADA INITIO® 675 together or within 10 days* 662 mg ≤8 weeks >8 and ≤12 weeks >12 weeks mg pre-filled syringe 882 mg ≤8 weeks >8 and ≤12 weeks >12 weeks ARISTADA® 441 -

207533Orig1s000
( CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 207533Orig1s000 RISK ASSESSMENT and RISK MITIGATION REVIEW(S) Department of Health and Human Services Public Health Service Food and Drug Administration Center for Drug Evaluation and Research Office of Surveillance and Epidemiology Office of Medication Error Prevention and Risk Management Risk Evaluation and Mitigation Strategy (REMS) Review Date: October 2, 2015 Reviewer(s): Cathy A. Miller, MPH, B.SN Risk Management Analyst Division of Risk Management Team Leader Kimberly Lehrfeld, Pharm.D. Division of Risk Management Acting Deputy Division Reema Mehta, Pharm.D., MPH Director Division of Risk Management Drug Name(s): Aristada (aripiprazole lauroxil) extended-release injection Therapeutic Class: Atypical Antipsychotic Dosage and Route: 441 mg/1.6 mL; 662 mg/2.4 mL; 882 mg/3.2mL intramuscular injection Application Type/Number: NDA 207533 Submission Number: ORIG-1 Submission Seq. No. 0000 dated August 22, 2014 Applicant/Sponsor: Alkermes OSE RCM #: 2014-1849 ***This document contains proprietary and confidential information that should not be released to the public*** Reference ID: 3828483 CONTENTS 1 INTRODUCTION ....................................................................................................... 1 1.1 Product Background............................................................................................ 1 1.2 Disease Background............................................................................................ 2 1.3 Regulatory History ............................................................................................. -

Aripiprazole Long-Acting Injection(Abilify Maintena
Aripiprazole Long-acting Injection Aripiprazole Long-acting Injection (Abilify Maintena, ARISTADA) National Drug Monograph October 2013; Addendum March 2016 VA Pharmacy Benefits Management Services, Medical Advisory Panel, and VISN Pharmacist Executives The purpose of VA PBM Services drug monographs is to provide a comprehensive drug review for making formulary decisions. These documents will be updated when new clinical data warrant additional formulary discussion. Documents will be placed in the Archive section when the information is deemed to be no longer current. Executive Summary: Aripiprazole long-acting intramuscular injection (LAIM) is sixth antipsychotic and the fourth second generation antipsychotic to be marketed in a long-acting injection formulation. LAIM antipsychotics on the VA National Formulary: haloperidol and fluphenazine decanoate; risperidone; and paliperidone. Aripiprazole LAIM has FDA label indication for the treatment of schizophrenia. The initial and usual maintenance dose of aripiprazole is 400 mg once a month. The dose can be reduced to 300 mg or 200 mg monthly based on drug interactions or tolerability. Patients should have established tolerability to aripiprazole before receipt of the LAIM formulation. Oral aripiprazole, 10-30 mg/day, or another oral antipsychotic must be continued for 2-weeks after the initial dose, and then discontinued. Aripiprazole LAIM’s efficacy and safety are based on experience with the oral formulation as well as pharmacokinetic trials and a 52-week randomized, double-blind, placebo-controlled trial. The randomized trial was terminated early because the difference in time to relapse met a predetermined statistically significant threshold (p=0.001) favoring aripiprazole LAIM [HR 5.03, 95% CI 3.15-8.02]. -

Using Medications, Cosmetics, Or Eating Certain Foods Can Increase Sensitivity to Ultraviolet Radiation
USING MEDICATIONS, COSMETICS, OR EATING CERTAIN FOODS CAN INCREASE SENSITIVITY TO ULTRAVIOLET RADIATION. INDIVIDUALS SHOULD CONSULT A PHYSICIAN BEFORE USING A SUNLAMP, IF THEY ARE TAKING MEDICATIONS. THE ITEMS LISTED ARE POTENTIAL PHOTOSENSITIZING AGENTS THAT MAY INCREASE SENSITIVITY TO ULTRAVIOLET LIGHT THAT MAY RESULT IN A PHOTOTOXIC OR PHOTOALLERGIC RESPONSES. PHOTOSENSITIZING MEDICATIONS Acetazolamide Amiloride+Hydrochlorothizide Amiodarone Amitriptyline Amoxapine Astemizole Atenolol+Chlorthalidone Auranofin Azatadine (Optimine) Azatidine+Pseudoephedrine Bendroflumethiazide Benzthiazide Bromodiphenhydramine Bromopheniramine Captopril Captopril+Hydrochlorothiazide Carbaamazepine Chlordiazepoxide+Amitriptyline Chlorothiazide Chlorpheniramine Chlorpheniramin+DPseudoephedrine Chlorpromazine Chlorpheniramine+Phenylopropanolamine Chlorpropamide Chlorprothixene Chlorthalidone Chlorthalidone+Reserpine Ciprofloxacin Clemastine Clofazime ClonidineChlorthalisone+Coal Tar Coal Tar Contraceptive (oral) Cyclobenzaprine Cyproheptadine Dacarcazine Danazol Demeclocycline Desipramine Dexchlorpheniramine Diclofenac Diflunisal Ditiazem Diphenhydramine Diphenylpyraline Doxepin Doxycycline Doxycycline Hyclate Enalapril Enalapril+Hydrochlorothiazide Erythromycin Ethylsuccinate+Sulfisoxazole Estrogens Estrogens Ethionamide Etretinate Floxuridine Flucytosine Fluorouracil Fluphenazine Flubiprofen Flutamide Gentamicin Glipizide Glyburide Gold Salts (compounds) Gold Sodium Thiomalate Griseofulvin Griseofulvin Ultramicrosize Griseofulvin+Hydrochlorothiazide Haloperidol