Download Download
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Vernacular Name AMPHIUMA, ONE-TOED (Aka: Congo Eel, Congo Snake, Ditch Eel, Fish Eel and Lamprey Eel)
1/6 Vernacular Name AMPHIUMA, ONE-TOED (aka: Congo Eel, Congo Snake, Ditch Eel, Fish Eel and Lamprey Eel) GEOGRAPHIC RANGE Eastern Gulf coast. HABITAT Wetlands: slow moving or stagnant freshwater rivers/streams/creeks and bogs, marshes, swamps, fens and peat lands. CONSERVATION STATUS IUCN: Near Threatened (2016). Population Trend: Decreasing. Because of the limited extent of its occurrence and because of the declining extent and quality of its habitat, this species is close to qualifying for Vulnerable. COOL FACTS Amphiumas are commonly known as "Congo eels," a misnomer. First of all, amphiumas are amphibians, rather than fish (which eels are). This notwithstanding, amphiumas bear resemblance to the elongate fishes. It is easy to overlook the diminutive legs, and the lack of any external gills adds to the similarity between the amphiumas and eels. Amphiumas are adapted for digging and tunneling. They seem to spend most of the time in muddy burrows and are rarely observed in the wild. They never fully metamorphose and retain larval characteristics in varying degrees into adulthood: one pair of the larval gill slits is retained and never disappears, no eyelids, no tongue and the presence of 4 gill arches with a single respiratory opening between the 3 rd--4th arches. Amphiumas have two pairs of limbs, and the three species, all of which occur in the S.E. U.S, differ in regard to the number of toes at the ends of these limbs: one, two or three. These amphiumas possess tiny, single-toed limbs, one pair just behind the small gill opening at each side of the neck and another pair just ahead of the longitudinal anal slit . -

Missouri State Wildlife Action Plan Missouri Department of Conservation Conserving Healthy Fish, Forests, and Wildlife 2015
Missouri State Wildlife Action Plan Missouri Department of Conservation CONSERVING HEALTHY FISH, FORESTS, AND WILDLIFE 2015 Missouri State Wildlife Action Plan 2015 Missouri is a national leader in fish, forest, and wildlife conservation due to Missouri citizens’ unique and proactive support of conservation efforts. The Conservation Department continues to build on our 79- year legacy of citizen-led conservation by outlining strategic priorities for the future to help us successfully manage fish, forest, and wildlife. Each of these priorities ties directly back to the heart of our mission: to manage and protect the fish, forest, and wildlife resources of the state and to provide opportunities for all citizens to use, enjoy, and learn about those resources. - Robert L. Ziehmer, Director Missouri Department of Conservation Missouri State Wildlife Action Plan 2015 FOREWORD issouri supports an abundant natural heri- examples of the state’s original natural communities tage, ranking 21st in the nation in terms of and outstanding biological diversity. The Depart- Mits numbers of native animal and plant spe- ment’s science-based efforts, aimed at understand- cies. There are over 180 native fish species, includ- ing life-history needs and habitat system dynamics, ing the endemic Niangua darter, that ply the state’s have benefited a variety of Missouri species, includ- aquatic habitats. More than 100 species of native ing recovery efforts of the American burying beetle, amphibians and reptiles occur in a myriad of habitats Ozark hellbender, eastern hellbender, eastern collared from mountain-top glades to lowland swamps. Mis- lizard, prairie massasauga rattlesnake, greater prai- souri supports nationally significant river and stream rie-chicken, bald eagle, peregrine falcon, pallid and systems, some of the largest forested tracts left in the lake sturgeons, Niangua darter, Topeka shiner, Virgin- Midwest, a high density of cave and karst features, ia sneezeweed, geocarpon, and Missouri bladderpod. -

Summary Report of Freshwater Nonindigenous Aquatic Species in U.S
Summary Report of Freshwater Nonindigenous Aquatic Species in U.S. Fish and Wildlife Service Region 4—An Update April 2013 Prepared by: Pam L. Fuller, Amy J. Benson, and Matthew J. Cannister U.S. Geological Survey Southeast Ecological Science Center Gainesville, Florida Prepared for: U.S. Fish and Wildlife Service Southeast Region Atlanta, Georgia Cover Photos: Silver Carp, Hypophthalmichthys molitrix – Auburn University Giant Applesnail, Pomacea maculata – David Knott Straightedge Crayfish, Procambarus hayi – U.S. Forest Service i Table of Contents Table of Contents ...................................................................................................................................... ii List of Figures ............................................................................................................................................ v List of Tables ............................................................................................................................................ vi INTRODUCTION ............................................................................................................................................. 1 Overview of Region 4 Introductions Since 2000 ....................................................................................... 1 Format of Species Accounts ...................................................................................................................... 2 Explanation of Maps ................................................................................................................................ -

AMPHIBIANS of OHIO F I E L D G U I D E DIVISION of WILDLIFE INTRODUCTION
AMPHIBIANS OF OHIO f i e l d g u i d e DIVISION OF WILDLIFE INTRODUCTION Amphibians are typically shy, secre- Unlike reptiles, their skin is not scaly. Amphibian eggs must remain moist if tive animals. While a few amphibians Nor do they have claws on their toes. they are to hatch. The eggs do not have are relatively large, most are small, deli- Most amphibians prefer to come out at shells but rather are covered with a jelly- cately attractive, and brightly colored. night. like substance. Amphibians lay eggs sin- That some of these more vulnerable spe- gly, in masses, or in strings in the water The young undergo what is known cies survive at all is cause for wonder. or in some other moist place. as metamorphosis. They pass through Nearly 200 million years ago, amphib- a larval, usually aquatic, stage before As with all Ohio wildlife, the only ians were the first creatures to emerge drastically changing form and becoming real threat to their continued existence from the seas to begin life on land. The adults. is habitat degradation and destruction. term amphibian comes from the Greek Only by conserving suitable habitat to- Ohio is fortunate in having many spe- amphi, which means dual, and bios, day will we enable future generations to cies of amphibians. Although generally meaning life. While it is true that many study and enjoy Ohio’s amphibians. inconspicuous most of the year, during amphibians live a double life — spend- the breeding season, especially follow- ing part of their lives in water and the ing a warm, early spring rain, amphib- rest on land — some never go into the ians appear in great numbers seemingly water and others never leave it. -

TRAPPING SUCCESS and POPULATION ANALYSIS of Siren Lacertina and Amphiuma Means
TRAPPING SUCCESS AND POPULATION ANALYSIS OF Siren lacertina AND Amphiuma means By KRISTINA SORENSEN A THESIS PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE UNIVERSITY OF FLORIDA 2003 ACKNOWLEDGMENTS I thank my committee members Lora Smith, Franklin Percival, and Dick Franz for all their support and advice. The Department of Interior's Student Career Experience Program and the U.S. Geological Survey's Amphibian Research and Monitoring Initiative provided funding for this project. I thank those involved with these programs who have helped me over the last three years: David Trauger, Ken Dodd, Jamie Barichivich, Jennifer Staiger, Kevin Smith, and Steve Johnson. Numerous people helped with field work: Audrey Owens, Maya Zacharow, Chris Gregory, Matt Chopp, Amanda Rice, Paul Loud, Travis Tuten, Steve Johnson, and Jennifer Staiger, Lora Smith, and the UF Wildlife Field Techniques Courses of2001-2002. Paul Moler and John Jensen provided advice and shared their wealth of herpetological knowledge. I thank the staff of Okefenokee National Wildlife Refuge and Steve Coates, manager of the Ordway Preserve, for their assistance on numerous occasions and for permission to conduct research on their property. Marinela Capanu of the IFAS Statistical Consulting Unit assisted with statistical analysis. Julien Martin, Bob Dorazio, Rob Bennets, and Cathy Langtimm provided advice on population analysis. I also thank the administrative staff of the Florida Caribbean Science Center and the Florida Cooperative Fish and Wildlife Research Unit. I am much indebted to all of these people, without whom this thesis would not have been possible. -

The Salamanders of Tennessee
Salamanders of Tennessee: modified from Lisa Powers tnwildlife.org Follow links to Nongame The Salamanders of Tennessee Photo by John White Salamanders are the group of tailed, vertebrate animals that along with frogs and caecilians make up the class Amphibia. Salamanders are ectothermic (cold-blooded), have smooth glandular skin, lack claws and must have a moist environment in which to live. 1 Amphibian Declines Worldwide, over 200 amphibian species have experienced recent population declines. Scientists have reports of 32 species First discovered in 1967, the golden extinctions, toad, Bufo periglenes, was last seen mainly species of in 1987. frogs. Much attention has been given to the Anurans (frogs) in recent years, however salamander populations have been poorly monitored. Photo by Henk Wallays Fire Salamander - Salamandra salamandra terrestris 2 Why The Concern For Salamanders in Tennessee? Their key role and high densities in many forests The stability in their counts and populations Their vulnerability to air and water pollution Their sensitivity as a measure of change The threatened and endangered status of several species Their inherent beauty and appeal as a creature to study and conserve. *Possible Factors Influencing Declines Around the World Climate Change Habitat Modification Habitat Fragmentation Introduced Species UV-B Radiation Chemical Contaminants Disease Trade in Amphibians as Pets *Often declines are caused by a combination of factors and do not have a single cause. Major Causes for Declines in Tennessee Habitat Modification -The destruction of natural habitats is undoubtedly the biggest threat facing amphibians in Tennessee. Housing, shopping center, industrial and highway construction are all increasing throughout the state and consequently decreasing the amount of available habitat for amphibians. -
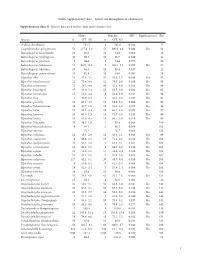
I Online Supplementary Data – Sexual Size Dimorphism in Salamanders
Online Supplementary data – Sexual size dimorphism in salamanders Supplementary data S1. Species data used in this study and references list. Males Females SSD Significant test Ref Species n SVL±SD n SVL±SD Andrias davidianus 2 532.5 8 383.0 -0.280 12 Cryptobranchus alleganiensis 53 277.4±5.2 52 300.9±3.4 0.084 Yes 61 Batrachuperus karlschmidti 10 80.0 10 84.8 0.060 26 Batrachuperus londongensis 20 98.6 10 96.7 -0.019 12 Batrachuperus pinchonii 5 69.6 5 74.6 0.070 26 Batrachuperus taibaiensis 11 92.9±12.1 9 102.1±7.1 0.099 Yes 27 Batrachuperus tibetanus 10 94.5 10 92.8 -0.017 12 Batrachuperus yenyuadensis 10 82.8 10 74.8 -0.096 26 Hynobius abei 24 57.8±2.1 34 55.0±1.2 -0.048 Yes 92 Hynobius amakusaensis 22 75.4±4.8 12 76.5±3.6 0.014 No 93 Hynobius arisanensis 72 54.3±4.8 40 55.2±4.8 0.016 No 94 Hynobius boulengeri 37 83.0±5.4 15 91.5±3.8 0.102 Yes 95 Hynobius formosanus 15 53.0±4.4 8 52.4±3.9 -0.011 No 94 Hynobius fuca 4 50.9±2.8 3 52.8±2.0 0.037 No 94 Hynobius glacialis 12 63.1±4.7 11 58.9±5.2 -0.066 No 94 Hynobius hidamontanus 39 47.7±1.0 15 51.3±1.2 0.075 Yes 96 Hynobius katoi 12 58.4±3.3 10 62.7±1.6 0.073 Yes 97 Hynobius kimurae 20 63.0±1.5 15 72.7±2.0 0.153 Yes 98 Hynobius leechii 70 61.6±4.5 18 66.5±5.9 0.079 Yes 99 Hynobius lichenatus 37 58.5±1.9 2 53.8 -0.080 100 Hynobius maoershanensis 4 86.1 2 80.1 -0.069 101 Hynobius naevius 72.1 76.7 0.063 102 Hynobius nebulosus 14 48.3±2.9 12 50.4±2.1 0.043 Yes 96 Hynobius osumiensis 9 68.4±3.1 15 70.2±3.0 0.026 No 103 Hynobius quelpaertensis 41 52.5±3.8 4 61.3±4.1 0.167 Yes 104 Hynobius -

Amphiumidae Cryptobranchidae Proteidae
Amphiumidae Amphiuma means – Two-toed Amphiuma cryptobranchidae Cryptobranchus alleganiensis – Hellbender Proteidae Necturus maculosus – Mudpuppy Sirenidae Siren intermedia – Lesser Siren Ambystomatidae Ambystoma gracile – Northwestern Salamander Ambystoma tigrinum – Tiger Salamander Ambystoma macrodactylum – Long-toed Salamander Ambystoma californiense – California Tiger Salamander Dicamptodontidae Dicamptodon ensatus – California Giant Salamander Dicamptodon tenebrosus – Pacific Giant Salamander Rhyacotritonidae Rhyacotriton variegatus – Southern Torrent Salamander Salamandridae Taricha torosa – California Newt Taricha Sierrae – Sierra Newt Taricha rivularis – Red-bellied Newt Taricha granulosa – Rough-skinned Newt Plethodontidae Aneides flavipunctatus – Black Salamander Aneides vagrans – Wandering Salamander Aneides ferreus – Clouded Salamander Aneides lugubris – Arboreal Salamander Plethodontidae Ensatina eschscholtzii – Ensatina Plethodontidae Batrachoseps attenuatus – California Slender Salamander Batrachoseps gavilanensis – Gabilan Mountains Slender Salamander Plethodontidae Plethodon dunni – Dunn’s Salamander Plethodon stormi – Siskiyou Mountains Salamander Plethodon elongatus – Del Norte Salamander Plethodon asupak – Scott Bar Salamander Plethodontidae Hydromantes brunus – Limestone Salamander Hydromantes platycephalus – Mount Lyell Salamander Hydromantes shastae – Shasta Salamander Ascaphidae Ascaphus truei – Tailed Frog Pelobatidae Spea hammondii – Western Spadefoot Spea intermontana – Great Basin Spadefoot Scaphiopus couchi -

Key to the Identification of Streamside Salamanders
Key to the Identification of Streamside Salamanders Ambystoma spp., mole salamanders (Family Ambystomatidae) Appearance : Medium to large stocky salamanders. Large round heads with bulging eyes . Larvae are also stocky and have elaborate gills. Size: 3-8” (Total length). Spotted salamander, Ambystoma maculatum Habitat: Burrowers that spend much of their life below ground in terrestrial habitats. Some species, (e.g. marbled salamander) may be found under logs or other debris in riparian areas. All species breed in fishless isolated ponds or wetlands. Range: Statewide. Other: Five species in Georgia. This group includes some of the largest and most dramatically patterned terrestrial species. Marbled salamander, Ambystoma opacum Amphiuma spp., amphiuma (Family Amphiumidae) Appearance: Gray to black, eel-like bodies with four greatly reduced, non-functional legs (A). Size: up to 46” (Total length) Habitat: Lakes, ponds, ditches and canals, one species is found in deep pockets of mud along the Apalachicola River floodplains. A Range: Southern half of the state. Other: One species, the two-toed amphiuma ( A. means ), shown on the right, is known to occur in A. pholeter southern Georgia; a second species, ,Two-toed amphiuma, Amphiuma means may occur in extreme southwest Georgia, but has yet to be confirmed. The two-toed amphiuma (shown in photo) has two diminutive toes on each of the front limbs. Cryptobranchus alleganiensis , hellbender (Family Cryptobranchidae) Appearance: Very large, wrinkled salamander with eyes positioned laterally (A). Brown-gray in color with darker splotches Size: 12-29” (Total length) A Habitat: Large, rocky, fast-flowing streams. Often found beneath large rocks in shallow rapids. Range: Extreme northern Georgia only. -

Streamside Salamander Identification Guide
Key to the Identification of Streamside Salamanders 1 Ambystoma spp., mole salamanders (Family Ambystomatidae) Appearance: Medium to large stocky salamanders. Large round heads with bulging eyes. Larvae are also stocky and have elaborate gills. Size: 3-8” (Total length). Spotted salamander, Ambystoma maculatum Habitat: Burrowers that spend much of their life below ground in terrestrial habitats. Some species, (e.g. marbled salamander) may be found under logs or other debris in riparian areas. All species breed in fishless isolated ponds or wetlands. Range: Statewide. Other: Five species in Georgia. This group includes some of the largest and most dramatically patterned terrestrial species. Marbled salamander, Ambystoma opacum Amphiuma spp., amphiuma (Family Amphiumidae) Appearance: Gray to black, eel-like bodies with four greatly reduced, non-functional legs (A). Size: up to 46” (Total length) Habitat: Lakes, ponds, ditches and canals, one species is found in deep pockets of mud along the Apalachicola River floodplains. A Range: Southern half of the state. A Other: One species, the two-toed amphiuma (A. means), shown on the right, is known to occur in A. pholeter southern Georgia; a second species, ,Two-toed amphiuma, Amphiuma means may occur in extreme southwest Georgia, but has yet to be confirmed. The two-toed amphiuma (shown in photo) has two diminutive toes on each of the front limbs. 2 Cryptobranchus alleganiensis, hellbender (Family Cryptobranchidae) Appearance: Very large, wrinkled salamander with eyes positioned laterally (A). Brown-gray in color with darker splotches Size: 12-29” (Total length) A Habitat: Large, rocky, fast-flowing streams. Often found beneath large rocks in shallow rapids. -

Sperm Storage in Spermathecae of the Great Lamper Eel, Amphiuma Tridacfyhm (Caudata: Amp Hi U Midae)
JOURNAL OF MORPHOLOGY 230:79-97 (1996) Sperm Storage in Spermathecae of the Great Lamper Eel, Amphiuma tridacfyhm (Caudata: Amp hi u midae) DAVID M. SEVER, J. SEAN DOODY, COURTNEY A. REDDISH, MICHELLE M. WENNER, AND DON R. CHURCH Department of Biology, Saint Mary's College, Notre Dame, Indiana 46556 (D.M.S., C.A.R., M.M. W.); Department of Biological Sciences, Southeastern Louisiana University, Hammond, Louisiana 70402 (J.S.D., D.R.C.) ABSTRACT The spermathecae of ten female Amphiuma tridactylum were examined by light and electron microscopy during the presumed mating and ovipository seasons (March-August) in Louisiana. Spermathecae were simple tubuloalveolar glands in the dorsal wall of the cloaca. Six of the ten specimens were vitellogenic, and all of these specimens contained sperm in their sperma- thecae and had secretory activity in the spermathecal epithelium. Two nonvitel- logenic females also had sperm in their spermathecae and active epithelial cells, whereas the other nonvitellogenic females lacked stored sperm and secretory activity in the spermathecae. In specimens storing sperm from March-May, the sperm were normal in cytology, and secretory vacuoles were contained within the epithelium. In the August sample, however, evidence of sperm degradation was present, and secretory material had been released into the lumen by an apocrine process. We therefore hypothesize that the spermathecal secretions function in sperm degeneration. Q 1996 Wiley-Liss, Inc. The Amphiumidae consists of three spe- of A. tridactylum and described these glands cies of Amphiuma, elongate, fossorial sala- in A. means and A. phloleter. In addition, manders with reduced limbs and one exter- Sever ('gla, '94) reported on the phylogeny nal gill slit, a paedomorphic character of the spermathecae and other cloacal glands (Duellman and Trueb, '86). -

The Giant Salamanders (Cryptobranchidae): Part B
Copyright: © This is an open-access article distributed under the terms of the Creative Commons Attribution–NonCommercial– NoDerivs 3.0 Unported License, which permits conditional use for non-commercial and education purposes, provided the Amphibian and Reptile Conservation 5(4): 30-50. original author and source are credited, and prohibits the deposition of material from www.redlist-arc.org, including PDFs, images, or text onto other websites without permission of the International Chapter at www.redlist-arc.org. The giant salamanders (Cryptobranchidae): Part B. Biogeography, ecology and reproduction 1Robert K Browne, 2Hong Li, 3Zhenghuan Wang, 4Sumio Okada, 5Paul Hime, 6Amy McMillan, 7Minyao Wu, 8Raul Diaz, 9Dale McGinnity, 10Jeffrey T. Briggler 1Sustainability America, Sarteneja, Belize. 2Polytechnic Institute of New York University, NYC, USA. 3School of Life Sciences, East China Normal University, 200062, Shanghai, Peoples Republic of China. 4Laboratory of Biology, Department of Regional Environment, Tottori University, Tottori 680-8551, Japan. 5Department of Biology, University of Kentucky, Department of Biology, Lexington, Kentucky 40506, USA. 6SUNY Buffalo State, Buffalo, New York 14222, USA. 7Shaanxi Normal University, Xi’an, 710062, Shaanxi Province, Peoples Republic of China. 8La Sierra University, Department of Biology, 4500 Riverwalk Parkway, Riverside, California 92515, USA 9Nashville Zoo, Nashville, Tennessee 37189, USA. 10Missouri Department of Conservation, Jefferson City, Missouri 65109, USA. Abstract - The Chinese (Andrias davidianus) and Japanese (A. japonicus) giant salamanders far exceed any other living amphibians in size, with the North American giant salamander (Hellbender; Cryptobranchus alleganiensis) also being one of the world’s largest amphibians. The sustainable management of cryptobranchids requires knowledge of cryptobranchid biogeography, ecology and reproduction in concert with other scientific fields.