Lab #4 the Behavior of Two Families in the Periodic Table
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Federal Law and Vertebrate Pest Control
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Proceedings of the 1st Vertebrate Pest Vertebrate Pest Conference Proceedings Conference (1962) collection February 1962 FEDERAL LAW AND VERTEBRATE PEST CONTROL Justus C. Ward Director, Pesticides Regulation Division, Agricultural Research Service, U.S. Department of Agriculture Follow this and additional works at: https://digitalcommons.unl.edu/vpcone Part of the Environmental Health and Protection Commons Ward, Justus C., "FEDERAL LAW AND VERTEBRATE PEST CONTROL" (1962). Proceedings of the 1st Vertebrate Pest Conference (1962). 25. https://digitalcommons.unl.edu/vpcone/25 This Article is brought to you for free and open access by the Vertebrate Pest Conference Proceedings collection at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Proceedings of the 1st Vertebrate Pest Conference (1962) by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. FEDERAL LAW AND VERTEBRATE PEST CONTROL By: Justus C. Ward, Director, Pesticides Regulation Division, Agricultural Research Service, U.S. Department of Agriculture Presented at the Vertebrate Pest Control Conference, Sacramento, California, February 6 and 7, 1 962 Shortly after the passage of the Federal Insecticide Act of 1910> mammal control specialists in the Bureau of Biological Survey began to consider a similar law to cover the chemicals with which they were concerned. Work on the project went slowly a nd spasmodically, but reached the point of having a Federal Rodenticide Act available for study and possible revision in 1928. At this time, the mammal control chemicals in use were limited to strychnine -- alkaloid and sulphate -arsenic, barium carbonate, th allium sulphate, phosphorus, s odium and calcium cyanide, carbon disulphide, and red squill. -

Influence of Water on Conductivity of Aluminium Halide Solutions in Acetonitrile
Influence of water on conductivity of aluminium halide solutions in acetonitrile L. LUX Department of Chemistry, Faculty of Metallurgy, Technical University, CS-043 85 Košice Received 7 September 1981 Accepted for publication 8 February 1982 Equilibria of the substitution and association reactions of AICI3 and AlBr3 with acetonitrile were investigated by measuring the conductivity of solutions at varying concentrations of A1C13, AlBr3, and water in the acetonitrile solution. Both halides behave in acetonitrile in the absence of water as relatively strong electrolytes. It has been revealed that water displaces acetoni trile from the solvate sphere of cation of both halides. As for AlBr3, the bromide ion is also displaced from the coordination sphere of aluminium and replaced by a neutral water molecule, which simultaneously results in an increased number of ionized particles in solution. The hydrate A1X3 -6H20 was identified as final product which separates from the solution by adding water. Изучены равновесия реакций замещения и ассоциации А1С13 и А1Вг3 с ацетонитрилом посредством измерения электропроводности раствора в зависимости от концентрации А1С13, А1Вг3, как и в зависимости от концентрации воды в растворе ацетонитрила. Оба галогенида ведут себя в ацетонитриле как сравнительно сильные электролиты в отсутствии воды. Обнаружено, что вода вытесняет ацетонитрил из сольватной обо лочки катиона у обоих галогенидов. У А1Вг3 доходит и к вытеснению иона брома из координационной сферы алюминия и его замещению нейтраль ной молекулой воды, что одновременно приводит к увеличению числа ионизированных частиц в растворе. Конечным продуктом, выделенным из раствора при добавлении воды был идентифицированный гидрат А1Х3-6Н20. Acetonitrile is a typical aprotic solvent which is widely used in electrochemical research because of its profitable properties, i.e. -

Molecular Dynamics Simulation of Ion Mobility. 2. Alkali Metal and Halide Ions Using the SPC/E Model for Water at 25 °C†
+ + 1420 J. Phys. Chem. 1996, 100, 1420-1425 Molecular Dynamics Simulation of Ion Mobility. 2. Alkali Metal and Halide Ions Using the SPC/E Model for Water at 25 °C† Song Hi Lee Department of Chemistry, Kyungsung UniVersity, Pusan 608-736, Korea Jayendran C. Rasaiah* Department of Chemistry, UniVersity of Maine, Orono, Maine 04469 ReceiVed: October 16, 1995; In Final Form: NoVember 10, 1995X We present results of computer simulations of the mobilities of the alkali metal ions (Li+, Na+,K+, Rb+, and Cs+) and the halides (F-, Cl-, Br-, and I-) at 25 °C using the SPC/E model for water and ion-water parameters fitted to the binding energies of small clusters of ions. A simple truncation of the ion-water and water- water potentials was used, and the mobilities calculated from the mean square displacement and the velocity autocorrelation functions, respectively, were found to be in good agreement with each other. The calculations demonstrate, for the first time, cation and anion mobilities that fall on separate curves, as functions of ion size, with distinct maxima. This is in complete accord with experimental trends observed in water at 25 °C. The cation mobilities are also in better agreement with the measured values than the calculations done earlier (J. Chem. Phys. 1994, 101, 6964) using the TIP4P model. The mobilities of the halides calculated here for the SPC/E model are however slightly lower than the experimental results. The residence times of water in the hydration shells around an ion are found to decrease dramatically with its size. Stereoscopic pictures show that the structure of the solvent cage around an ion is qualitatively different for the larger ions, implicating both solvent dynamics and structure as important factors in explaining ion mobility in aqueous systems. -

Strontium Chromate from Austria and France
Strontium Chromate from Austria and France Investigation Nos. 731-TA-1422-1423 (Preliminary) Publication 4836 October 2018 U.S. International Trade Commission Washington, DC 20436 U.S. International Trade Commission COMMISSIONERS David S. Johanson, Chairman Irving A. Williamson Meredith M. Broadbent Rhonda K. Schmidtlein Jason E. Kearns Catherine DeFilippo Director of Operations Staff assigned Kristina Lara, Investigator Samantha DeCarlo, Industry Analyst Tana von Kessler Economist Jennifer Brinckhaus, Accountant KaDeadra McNealy, Statistician David Goldfine, Attorney Douglas Corkran, Supervisory Investigator Address all communications to Secretary to the Commission United States International Trade Commission Washington, DC 20436 U.S. International Trade Commission Washington, DC 20436 www.usitc.gov Strontium Chromate from Austria and France Investigation Nos. 731-TA-1422-1423 (Preliminary) Publication 4836 October 2018 CONTENTS Page ....................................................................................................................... 1 ........................................................................................................ 3 Introduction ................................................................................................................ I‐1 Background ................................................................................................................................ I‐1 Statutory criteria and organization of the report .................................................................... -

Step-By-Step Guide to Better Laboratory Management Practices
Step-by-Step Guide to Better Laboratory Management Practices Prepared by The Washington State Department of Ecology Hazardous Waste and Toxics Reduction Program Publication No. 97- 431 Revised January 2003 Printed on recycled paper For additional copies of this document, contact: Department of Ecology Publications Distribution Center PO Box 47600 Olympia, WA 98504-7600 (360) 407-7472 or 1 (800) 633-7585 or contact your regional office: Department of Ecology’s Regional Offices (425) 649-7000 (509) 575-2490 (509) 329-3400 (360) 407-6300 The Department of Ecology is an equal opportunity agency and does not discriminate on the basis of race, creed, color, disability, age, religion, national origin, sex, marital status, disabled veteran’s status, Vietnam Era veteran’s status or sexual orientation. If you have special accommodation needs, or require this document in an alternate format, contact the Hazardous Waste and Toxics Reduction Program at (360)407-6700 (voice) or 711 or (800) 833-6388 (TTY). Table of Contents Introduction ....................................................................................................................................iii Section 1 Laboratory Hazardous Waste Management ...........................................................1 Designating Dangerous Waste................................................................................................1 Counting Wastes .......................................................................................................................8 Treatment by Generator...........................................................................................................12 -

Halides and Halogens. What Do I Need to Know? John Vivari, Nordson EFD
Halides and Halogens. What do I need to know? John Vivari, Nordson EFD Abstract With halogen-containing substances in the public eye due to scrutiny by the European Union and a variety of non- governmental organizations (NGOs) as possible additions to the list of substances banned from electronics, we at EFD have received numerous inquiries from customers asking how this subject will affect them and their processes. Having just overcome the hurdle of RoHS (Restriction of Hazardous Substances), they want to know what halogens and halides are, and what changes they should be prepared for if required to stop using them. Halide-free materials are not new. Some segments of the electronics industry have been sensitive to halides and their significance for decades. This paper will give the reader a working knowledge of halogens and halides. Armed with this education, the reader will be able to make informed decisions when required to use halogen-free materials, either because regulations dictate it or social pressure makes acceptance preferable to resistance. Key Words: halide, halogen, bromine, chlorine, flame retardant, RoHS What are halogens and halides? damage. Brominated flame retardant use is not limited to electronics. It is also in common usage in furniture, At their most basic level, halogens are the electronegative construction materials and textiles. elements in column 17 of the periodic table, including fluorine (F), chlorine, (Cl), bromine (Br), iodine (I) and Other sources of halogens in circuit boards include astatine (At). In electronics fiberglass sizing, epoxy curing agents and accelerators, applications, iodine and resin wetting and de-foaming agents, flux residues, and astatine are rarely if ever contamination from handling. -

Strontium-90 and Strontium-89: a Review of Measurement Techniques in Environmental Media
STRONTIUM-90 AND STRONTIUM-89: A REVIEW OF MEASUREMENT TECHNIQUES IN ENVIRONMENTAL MEDIA Robert J. Budnitz Lawrence Berkeley Laboratory University of California Berkeley, CA 91*720 1. IntroiUiction 2. Sources of Enivronmental Radiostrontium 3. Measurement Considerations a. Introduction b. Counting c. •»Sr/"Sr Separation 4. Chemical Techniques a. Air b. Water c. Milk d. Other Media e. Yttrium Recovery after Ingrowth f. Interferences S- Calibration Techniques h. Quality Control 5. Summary and Conclusions 6. Acknowli'J.jnent 7. References -NOTICE- This report was prepared as an account of worK sponsored by the United States Government. Neither the United States nor the United Slates Atomic Energy Commission, nor any of their employees, nor any of their contractors, subcontractors, or their employees, makes any wananty, express or implied, or asjumes any legal liabili:y or responsibility for the accuracy, com pleteness or usefulness of any Information, apparatus, J product or process disclosed, or represents that its use I would not infringe privately owned rights. Mmh li\ -1- 1. INTRODUCTION There are only two radioactive isotopes SrflO of strontium of significance in radiological measurements in the environment: strontium-89 Avg.jS energy 196-1 k«V and strontium-90. 100 Strontium-90 is the more significant from the point of view of environmental impact. 80 Energy keV 644 This is due mostly to its long half-life % Emission 100 (28.1 years). It is a pure beta emitter with 60 Typ* of only one decay mode, leading to yttrium-90 •mission by emission of a negative beta with HUBX * 40 h S46 keV. -

PUBLIC HEALTH STATEMENT Strontium CAS#: 7440-24-6
PUBLIC HEALTH STATEMENT Strontium CAS#: 7440-24-6 Division of Toxicology April 2004 This Public Health Statement is the summary External exposure to radiation may occur from chapter from the Toxicological Profile for natural or man-made sources. Naturally occurring strontium. It is one in a series of Public Health sources of radiation are cosmic radiation from space Statements about hazardous substances and their or radioactive materials in soil or building materials. health effects. A shorter version, the ToxFAQs™, is Man-made sources of radioactive materials are also available. This information is important found in consumer products, industrial equipment, because this substance may harm you. The effects atom bomb fallout, and to a smaller extent from of exposure to any hazardous substance depend on hospital waste and nuclear reactors. the dose, the duration, how you are exposed, personal traits and habits, and whether other If you are exposed to strontium, many factors chemicals are present. For more information, call determine whether you’ll be harmed. These factors the ATSDR Information Center at 1-888-422-8737. include the dose (how much), the duration (how _____________________________________ long), and how you come in contact with it. You must also consider the other chemicals you’re This public health statement tells you about exposed to and your age, sex, diet, family traits, strontium and the effects of exposure. lifestyle, and state of health. The Environmental Protection Agency (EPA) identifies the most serious hazardous waste sites in 1.1 WHAT IS STRONTIUM? the nation. These sites make up the National Priorities List (NPL) and are the sites targeted for Strontium is a natural and commonly occurring long-term federal cleanup activities. -

Electrochemical Separation and Purification of Yttrium-86
Radiochim. Acta 90, 225–228 (2002) by Oldenbourg Wissenschaftsverlag, München Electrochemical separation and purification of yttrium-86 By G. Reischl1,F.Rösch2 and H.-J. Machulla1 ,∗ 1 PET-Center, Division of Radiopharmacy, University Hospital Tübingen, D-72076 Tübingen, Germany 2 Institute of Nuclear Chemistry, University of Mainz, D-55099 Mainz, Germany (Received July 5, 2001; accepted in revised form October 24, 2001) Yttrium-86 / Electrolytic purification / radioactive rare earth nuclides onto a small tungsten cath- Electrochemical separation / Y-86 labeling ode was described in 1974 [6]. Recently, the electrochemical deposition of radionuclides of superheavy elements in de- pendence on electrode material and deposition potential was Summary. For quantitative determination of in vivo dosime- investigated [7]. An electrochemical method, based on a pro- try of 90Y-labeled radiotherapeuticals by means of PET, the cedure developed at Mainz [8, 9] to isolate 90Sr from a mix- positron emitting analogue yttrium-86 was produced at a low ture of 90Yand90Sr (c.a.) electrochemically, appeared very 86 86 energy (“medical”) cyclotron via the known Sr(p, n) Y 86 86 promising for isolating and purifying Y. reaction. Using 200 mg of SrCO3 (enrichment 95.6%) and protons of 15.1 MeV energy, average yields of 86Y of 48 ± 8MBq/µA h were produced. After dissolution of 86 86 Experimental SrCO3 in 3 ml of 0.6 N HNO3, Y was deposited in a simple and highly efficient electrochemical two-step procedure onto Materials a platinum cathode at 450 mA (= 20 mA/cm2). The isotope was finally removed from the electrode by 100–300 µlof Strontium-86 was supplied by Chemotrade, Germany, with . -

Strontium Carbonate Solubility Data in Aqueous Mixtures of Monoethyleneglycol Under a Carbon Dioxide Atmosphere
Brazilian Journal ISSN 0104-6632 of Chemical Printed in Brazil www.scielo.br/bjce Engineering Vol. 35, No. 02, pp. 395 - 402, April - June, 2018 dx.doi.org/10.1590/0104-6632.20180352s20160355 STRONTIUM CARBONATE SOLUBILITY DATA IN AQUEOUS MIXTURES OF MONOETHYLENEGLYCOL UNDER A CARBON DIOXIDE ATMOSPHERE Deborah C. de Andrade1,*, Naíra S. de A. Maniçoba1, Rony O. Sant'ana1, Fedra A.V. Ferreira1, Camila S. Figueiredo2, Jailton F. Nascimento2, Leonardo S. Pereira2 and Osvaldo Chiavone-Filho1 1UFRN. Campus Universitário. Departamento de Engenharia Química. Lagoa Nova. Natal – RN – Brazil, ZIP 59066-800. 2PETROBRAS/CENPES/PDEP/TPP. Av. Horácio Macedo, 950 – Cidade Universitária - Ilha do Fundão. Rio de Janeiro – RJ – Brazil, ZIP 219141-915. (Submitted: June 1, 2016; Revised: March 20, 2017; Accepted: April 5, 2017) Abstract - In order to inhibit natural gas hydrate formation, monoethyleneglycol (MEG) is usually injected into producing well heads. The MEG regeneration process is continuously performed at the platform. Scaling problems usually occur due to the presence of chlorides and carbonates. This work presents salt solubility data for the aqueous system with strontium carbonate, MEG and carbon dioxide. A specific analytical method was developed. Thus, experimental data for strontium carbonate (SrCO3) solubility at various carbon dioxide pressures are reported. Solubilities of SrCO3 in water were measured from 760 to 1610 mmHg at 278.15. 288.15. 298.15 and 323.15 K. For the mixed solvent (ms) conditions, solubilities were measured at 298.15 K and four isobars, i.e., 760, 1210, 1410 and 1520 mmHg. Experimental data were correlated and demonstrated to be accurate for thermodynamic modeling and process simulation. -

Toxicological Profile for Barium and Barium Compounds
TOXICOLOGICAL PROFILE FOR BARIUM AND BARIUM COMPOUNDS U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Public Health Service Agency for Toxic Substances and Disease Registry August 2007 BARIUM AND BARIUM COMPOUNDS ii DISCLAIMER The use of company or product name(s) is for identification only and does not imply endorsement by the Agency for Toxic Substances and Disease Registry. BARIUM AND BARIUM COMPOUNDS iii UPDATE STATEMENT A Toxicological Profile for Barium and Barium Compounds, Draft for Public Comment was released in September 2005. This edition supersedes any previously released draft or final profile. Toxicological profiles are revised and republished as necessary. For information regarding the update status of previously released profiles, contact ATSDR at: Agency for Toxic Substances and Disease Registry Division of Toxicology and Environmental Medicine/Applied Toxicology Branch 1600 Clifton Road NE Mailstop F-32 Atlanta, Georgia 30333 BARIUM AND BARIUM COMPOUNDS iv This page is intentionally blank. v FOREWORD This toxicological profile is prepared in accordance with guidelines developed by the Agency for Toxic Substances and Disease Registry (ATSDR) and the Environmental Protection Agency (EPA). The original guidelines were published in the Federal Register on April 17, 1987. Each profile will be revised and republished as necessary. The ATSDR toxicological profile succinctly characterizes the toxicologic and adverse health effects information for the hazardous substance described therein. Each peer-reviewed profile identifies and reviews the key literature that describes a hazardous substance's toxicologic properties. Other pertinent literature is also presented, but is described in less detail than the key studies. The profile is not intended to be an exhaustive document; however, more comprehensive sources of specialty information are referenced. -
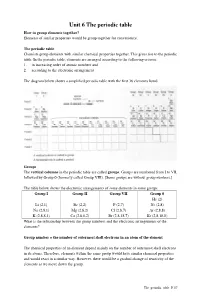
Unit 6 the Periodic Table How to Group Elements Together? Elements of Similar Properties Would Be Group Together for Convenience
Unit 6 The periodic table How to group elements together? Elements of similar properties would be group together for convenience. The periodic table Chemists group elements with similar chemical properties together. This gives rise to the periodic table. In the periodic table, elements are arranged according to the following criteria: 1. in increasing order of atomic numbers and 2. according to the electronic arrangement The diagram below shows a simplified periodic table with the first 36 elements listed. Groups The vertical columns in the periodic table are called groups . Groups are numbered from I to VII, followed by Group 0 (formerly called Group VIII). [Some groups are without group numbers.] The table below shows the electronic arrangements of some elements in some groups. Group I Group II Group VII Group 0 He (2) Li (2,1) Be (2,2) F (2,7) Ne (2,8) Na (2,8,1) Mg (2,8,2) Cl (2,8,7) Ar (2,8,8) K (2,8,8,1) Ca (2,8,8,2) Br (2,8,18,7) Kr (2,8,18,8) What is the relationship between the group numbers and the electronic arrangements of the elements? Group number = the number of outermost shell electrons in an atom of the element The chemical properties of an element depend mainly on the number of outermost shell electrons in its atoms. Therefore, elements within the same group would have similar chemical properties and would react in a similar way. However, there would be a gradual change of reactivity of the elements as we move down the group.