Collagen and Calcium-Binding EGF Domains 1 Is Frequently Inactivated in Ovarian Cancer by Aberrant Promoter Hypermethylation and Modulates Cell Migration and Survival
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Proteolytic Activation Defines Distinct Lymphangiogenic Mechanisms for VEGFC and VEGFD
Proteolytic activation defines distinct lymphangiogenic mechanisms for VEGFC and VEGFD Hung M. Bui, … , Kari Alitalo, Mark L. Kahn J Clin Invest. 2016;126(6):2167-2180. https://doi.org/10.1172/JCI83967. Research Article Vascular biology Lymphangiogenesis is supported by 2 homologous VEGFR3 ligands, VEGFC and VEGFD. VEGFC is required for lymphatic development, while VEGFD is not. VEGFC and VEGFD are proteolytically cleaved after cell secretion in vitro, and recent studies have implicated the protease a disintegrin and metalloproteinase with thrombospondin motifs 3 (ADAMTS3) and the secreted factor collagen and calcium binding EGF domains 1 (CCBE1) in this process. It is not well understood how ligand proteolysis is controlled at the molecular level or how this process regulates lymphangiogenesis, because these complex molecular interactions have been difficult to follow ex vivo and test in vivo. Here, we have developed and used biochemical and cellular tools to demonstrate that an ADAMTS3-CCBE1 complex can form independently of VEGFR3 and is required to convert VEGFC, but not VEGFD, into an active ligand. Consistent with these ex vivo findings, mouse genetic studies revealed that ADAMTS3 is required for lymphatic development in a manner that is identical to the requirement of VEGFC and CCBE1 for lymphatic development. Moreover, CCBE1 was required for in vivo lymphangiogenesis stimulated by VEGFC but not VEGFD. Together, these studies reveal that lymphangiogenesis is regulated by two distinct proteolytic mechanisms of ligand activation: one in which VEGFC activation by ADAMTS3 and CCBE1 spatially and temporally patterns developing lymphatics, and one in which VEGFD activation by a distinct […] Find the latest version: https://jci.me/83967/pdf The Journal of Clinical Investigation RESEARCH ARTICLE Proteolytic activation defines distinct lymphangiogenic mechanisms for VEGFC and VEGFD Hung M. -

Study of the Requirement of the CCBE1 Growth Factor in the Generation of Cardiac Myocytes from Hes Cells
Study of the requirement of the CCBE1 growth factor in the generation of cardiac myocytes from hES cells MS.c Thesis in Biomedical Science Rita Catarina Vaz Drago Study of the requirement of the CCBE1 growth factor in the generation of cardiac myocytes from hES cells MS.c Thesis in Biomedical Science Rita Catarina Vaz Drago Orientador: Professor Doutor José António Belo Co-orientador: Doutora Andreia Bernardo Faro, 2012 ii MS.c Thesis proposal in Biomedical Science Area of Developmental Biology by the Universidade do Algarve Study of the requirement of the CCBE1 growth factor in the generation of cardiac myocytes from hES cells. Dissertação para obtenção do Grau de Mestre em Ciências Biomédicas Área de Biologia do Desenvolvimento pela Universidade do Algarve Estudo da função do factor de crescimento CCBE1 na diferenciação de miócitos cardíacos a partir de células estaminais embrionárias humanas Declaro ser a autora deste trabalho, que é original e inédito. Autores e trabalhos consultados estão devidamente citados no texto e constam da listagem de referências incluída. Copyright. A Universidade do Algarve tem o direito, perpétuo e sem limites geográficos, de arquivar e publicitar este trabalho através de exemplares impressos reproduzidos em papel ou de forma digital, ou por qualquer outro meio conhecido ou que venha a ser inventado, de o divulgar através de repositórios científicos e de admitir a sua cópia e distribuição com objetivos educacionais ou de investigação, não comerciais, desde que seja dado crédito ao autor e editor. iii ACKNOWLEDGEMENTS I thank my supervisor, Prof. José Belo, for the support he provided me during this long journey and for the opportunity of working at his laboratory. -

Mutations in CCBE1 Cause Generalized Lymph Vessel Dysplasia
BRIEF COMMUNICATIONS Mutations in CCBE1 cause (Fig. 1a–f)8. Subsequently, subjects with lymphangiectasias in pleura, pericardium, thyroid gland and kidney and with hydrops fetalis were generalized lymph vessel dysplasia described9,10. The entity was designated lymphedema-lymphangiectasia– mental retardation or Hennekam syndrome (MIM 235510). Occurrence in humans of affected siblings, equal occurrence among sexes and frequent consanguinity indicated autosomal recessive inheritance9. 1 2 2 1 Marielle Alders , Benjamin M Hogan , Evisa Gjini , Faranak Salehi , We collected blood samples from a series of 27 subjects with Hennekam 3 4 5 Lihadh Al-Gazali , Eric A Hennekam , Eva E Holmberg , syndrome born to 22 families. None of the subjects carried mutations 1 6 7,8 Marcel M A M Mannens , Margot F Mulder , G Johan A Offerhaus , in FLT4, FOXC2 or SOX18. We performed homozygosity mapping in 5 9 10 Trine E Prescott , Eelco J Schroor , Joke B G M Verheij , three unpublished subjects (A, B and C) originating from a small isolate 2 11,14 12 Merlijn Witte , Petra J Zwijnenburg , Mikka Vikkula , in The Netherlands. Pedigree analysis had shown the parents of subject 2,15 13–15 Stefan Schulte-Merker & Raoul C Hennekam A to be consanguineous and all three subjects to be related (Fig. 1g). We reasoned that occurrence of three cases of a rare disorder in a small Lymphedema, lymphangiectasias, mental retardation and isolate suggested homozygosity for a founder mutation. Homozygosity unusual facial characteristics define the autosomal recessive mapping identified a 5.7-Mb homozygous region on chromosome 18q21 Hennekam syndrome. Homozygosity mapping identified a with identical haplotypes in the three affected individuals (Fig. -

Prioritization of Genes Involved in Endothelial Cell Apoptosis by Their Implication in Lymphedema Using an Analysis of Associative Gene Networks with Andsystem Olga V
Saik et al. BMC Medical Genomics 2019, 12(Suppl 2):47 https://doi.org/10.1186/s12920-019-0492-9 RESEARCH Open Access Prioritization of genes involved in endothelial cell apoptosis by their implication in lymphedema using an analysis of associative gene networks with ANDSystem Olga V. Saik1,3*, Vadim V. Nimaev2,3, Dilovarkhuja B. Usmonov3,4, Pavel S. Demenkov1,3, Timofey V. Ivanisenko1,3, Inna N. Lavrik1,5 and Vladimir A. Ivanisenko1,3 From 11th International Multiconference “Bioinformatics of Genome Regulation and Structure\Systems Biology” - BGRS\SB- 2018 Novosibirsk, Russia. 20-25 August 2018 Abstract Background: Currently, more than 150 million people worldwide suffer from lymphedema. It is a chronic progressive disease characterized by high-protein edema of various parts of the body due to defects in lymphatic drainage. Molecular-genetic mechanisms of the disease are still poorly understood. Beginning of a clinical manifestation of primary lymphedema in middle age and the development of secondary lymphedema after treatment of breast cancer can be genetically determined. Disruption of endothelial cell apoptosis can be considered as one of the factors contributing to the development of lymphedema. However, a study of the relationship between genes associated with lymphedema and genes involved in endothelial apoptosis, in the associative gene network was not previously conducted. Methods: In the current work, we used well-known methods (ToppGene and Endeavour), as well as methods previously developed by us, to prioritize genes involved in endothelial apoptosis and to find potential participants of molecular-genetic mechanisms of lymphedema among them. Original methods of prioritization took into account the overrepresented Gene Ontology biological processes, the centrality of vertices in the associative gene network, describing the interactions of endothelial apoptosis genes with genes associated with lymphedema, and the association of the analyzed genes with diseases that are comorbid to lymphedema. -

Genetic Epidemiology of Hypertension in Populations
GENETIC EPIDEMIOLOGY OF HYPERTENSION IN POPULATIONS: APPLICATIONS OF MODIFIED METHODS by PRIYA BHATIA SHETTY, M.S. Submitted in partial fulfillment of the requirements For the degree of Doctor of Philosophy Dissertation Adviser: Xiaofeng Zhu, Ph.D. Department of Epidemiology and Biostatistics CASE WESTERN RESERVE UNIVERSITY January, 2014 CASE WESTERN RESERVE UNIVERSITY SCHOOL OF GRADUATE STUDIES We hereby approve the dissertation of Priya Bhatia Shetty, candidate for the Doctor of Philosophy degree*. Xiaofeng Zhu, PhD Robert C. Elston, PhD Jing Li, PhD Nathan Morris, PhD September 6, 2013 *We also certify that written approval has been obtained for any proprietary material contained therein. 2 Dedication This dissertation is dedicated to my husband Manju and our children Suvan, Poppy, Jujubee, and Sayali. 3 TABLE OF CONTENTS List of Tables………………………………………………………………….…………..5 List of Figures…………………………………………………………………………….6 Abstract……………………………………………………………………….…………..7 Chapter 1. Background……………………………………………………...….…………9 Chapter 2. Specific Aims………..……………………………………………………….53 Chapter 3. Variants in CXADR and F2RL1 are associated with blood pressure and obesity in African-Americans in regions identified through admixture mapping………………..60 Chapter 4. Novel variants for HDL-C, LDL-C and triglycerides identified from admixture mapping and fine-mapping analysis in families…...…………………………………….85 Chapter 5. Identification of admixture regions associated with risk of apparent treatment- resistant hypertension in African-Americans………..………………………………….117 Chapter 6. Discussion………………………………..………………………………....137 References………………………………………………………………………………142 4 List of Tables Chapter 1. Table 1. Genome-wide Association Studies of Hypertension………...……...40 Chapter 1. Table 2. Other Analysis Methods in the Genetic Analysis of Hypertension in Unrelated Subjects………………………………………………………………...……..51 Chapter 3. Table 1. Summary statistics…………………………………………...……..80 Chapter 3. Table 2. Results of gene-based multivariable regression models……..….….81 Chapter 3. -

Mouse Ccbe1 Conditional Knockout Project (CRISPR/Cas9)
https://www.alphaknockout.com Mouse Ccbe1 Conditional Knockout Project (CRISPR/Cas9) Objective: To create a Ccbe1 conditional knockout Mouse model (C57BL/6J) by CRISPR/Cas-mediated genome engineering. Strategy summary: The Ccbe1 gene (NCBI Reference Sequence: NM_178793 ; Ensembl: ENSMUSG00000046318 ) is located on Mouse chromosome 18. 11 exons are identified, with the ATG start codon in exon 1 and the TAG stop codon in exon 11 (Transcript: ENSMUST00000130300). Exon 3 will be selected as conditional knockout region (cKO region). Deletion of this region should result in the loss of function of the Mouse Ccbe1 gene. To engineer the targeting vector, homologous arms and cKO region will be generated by PCR using BAC clone RP23-138G22 as template. Cas9, gRNA and targeting vector will be co-injected into fertilized eggs for cKO Mouse production. The pups will be genotyped by PCR followed by sequencing analysis. Note: Mice homozygous for a knock-out allele exhibit prenatal lethality associated with edema and absence of lymphatic vessels. Exon 3 starts from about 17.65% of the coding region. The knockout of Exon 3 will result in frameshift of the gene. The size of intron 2 for 5'-loxP site insertion: 197081 bp, and the size of intron 3 for 3'-loxP site insertion: 9125 bp. The size of effective cKO region: ~553 bp. The cKO region does not have any other known gene. Page 1 of 8 https://www.alphaknockout.com Overview of the Targeting Strategy Wildtype allele gRNA region 5' gRNA region 3' 1 3 11 Targeting vector Targeted allele Constitutive KO allele (After Cre recombination) Legends Exon of mouse Ccbe1 Homology arm cKO region loxP site Page 2 of 8 https://www.alphaknockout.com Overview of the Dot Plot Window size: 10 bp Forward Reverse Complement Sequence 12 Note: The sequence of homologous arms and cKO region is aligned with itself to determine if there are tandem repeats. -

Loss of ADAMTS3 Activity Causes Hennekam Lymphangiectasia–Lymphedema Syndrome 3 Pascal Brouillard1,‡, Laura Dupont2,‡, Raphael Helaers1, Richard Coulie1, George E
Human Molecular Genetics, 2017, Vol. 26, No. 21 4095–4104 doi: 10.1093/hmg/ddx297 Advance Access Publication Date: 2 August 2017 Original Article ORIGINAL ARTICLE Loss of ADAMTS3 activity causes Hennekam lymphangiectasia–lymphedema syndrome 3 Pascal Brouillard1,‡, Laura Dupont2,‡, Raphael Helaers1, Richard Coulie1, George E. Tiller3,†, Joseph Peeden4, Alain Colige2,‡ and Miikka Vikkula 1,5,*,‡ 1Human Molecular Genetics, de Duve Institute, University of Louvain, 1200 Brussels, Belgium, 2Laboratory of Connective Tissues Biology, University of Lie`ge, 4000 Lie`ge, Belgium, 3Pediatric Medical Genetics, Vanderbilt University Medical Center, Nashville 37232, TN, USA, 4East Tennessee Children’s Hospital, University of Tennessee Medical Center, Knoxville, TN 37916, USA and 5Walloon Excellence in Life Sciences and Biotechnology (WELBIO), de Duve Institute, University of Louvain, 1200 Brussels, Belgium *To whom correspondence should be addressed at: Human Molecular Genetics, de Duve Institute, University of Louvain, Avenue Hippocrate 74, Box B1.74.06, B-1200 Brussels, Belgium. Tel: þ32 27647496; Fax: þ32 27647460; Email: [email protected] Abstract Primary lymphedema is due to developmental and/or functional defects in the lymphatic system. It may affect any part of the body, with predominance for the lower extremities. Twenty-seven genes have already been linked to primary lymph- edema, either isolated, or as part of a syndrome. The proteins that they encode are involved in VEGFR3 receptor signaling. They account for about one third of all primary lymphedema cases, underscoring the existence of additional genetic factors. We used whole-exome sequencing to investigate the underlying cause in a non-consanguineous family with two children af- fected by lymphedema, lymphangiectasia and distinct facial features. -

CCBE1 Mutation in Two Siblings, One Manifesting Lymphedema-Cholestasis Syndrome, and the Other, Fetal Hydrops
CCBE1 Mutation in Two Siblings, One Manifesting Lymphedema-Cholestasis Syndrome, and the Other, Fetal Hydrops Sohela Shah1, Laura K. Conlin2, Luis Gomez3, Øystein Aagenaes4, Kristin Eiklid5, A. S. Knisely6, Michael T. Mennuti3, Randolph P. Matthews7, Nancy B. Spinner2, Laura N. Bull1,8* 1 Liver Center Laboratory, Department of Medicine, University of California San Francisco, San Francisco, California, United States of America, 2 Department of Pathology and Laboratory Medicine, Children’s Hospital of Philadelphia and Perelman School of Medicine, University of Pennsylvania, Philadelphia, Pennsylvania, United States of America, 3 Department of Obstetrics and Gynecology, University of Pennsylvania, Philadelphia, Pennsylvania, United States of America, 4 Oslo University Hospital, Oslo, Norway, 5 Department of Medical Genetics, Oslo University Hospital, Ullevål, Oslo, Norway, 6 Institute of Liver Studies, King’s College Hospital, London, United Kingdom, 7 Division of Gastroenterology, Hepatology,and Nutrition, Children’s Hospital of Philadelphia and Department of Pediatrics, Perelman School of Medicine, University of Pennsylvania, Philadelphia, Pennsylvania, United States of America, 8 Institute for Human Genetics, Department of Medicine, University of California San Francisco, San Francisco, California, United States of America Abstract Background: Lymphedema-cholestasis syndrome (LCS; Aagenaes syndrome) is a rare autosomal recessive disorder, characterized by 1) neonatal intrahepatic cholestasis, often lessening and becoming intermittent with age, and 2) severe chronic lymphedema, mainly lower limb. LCS was originally described in a Norwegian kindred in which a locus, LCS1, was mapped to a 6.6cM region on chromosome 15. Mutations in CCBE1 on chromosome 18 have been reported in some cases of lymphatic dysplasia, but not in LCS. Methods: Consanguineous parents of Mexican ancestry had a child with LCS who did not exhibit extended homozygosity in the LCS1 region. -

Comprehensive Analysis Reveals Novel Gene Signature in Head and Neck Squamous Cell Carcinoma: Predicting Is Associated with Poor Prognosis in Patients
5892 Original Article Comprehensive analysis reveals novel gene signature in head and neck squamous cell carcinoma: predicting is associated with poor prognosis in patients Yixin Sun1,2#, Quan Zhang1,2#, Lanlin Yao2#, Shuai Wang3, Zhiming Zhang1,2 1Department of Breast Surgery, The First Affiliated Hospital of Xiamen University, School of Medicine, Xiamen University, Xiamen, China; 2School of Medicine, Xiamen University, Xiamen, China; 3State Key Laboratory of Cellular Stress Biology, School of Life Sciences, Xiamen University, Xiamen, China Contributions: (I) Conception and design: Y Sun, Q Zhang; (II) Administrative support: Z Zhang; (III) Provision of study materials or patients: Y Sun, Q Zhang; (IV) Collection and assembly of data: Y Sun, L Yao; (V) Data analysis and interpretation: Y Sun, S Wang; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors. #These authors contributed equally to this work. Correspondence to: Zhiming Zhang. Department of Surgery, The First Affiliated Hospital of Xiamen University, Xiamen, China. Email: [email protected]. Background: Head and neck squamous cell carcinoma (HNSC) remains an important public health problem, with classic risk factors being smoking and excessive alcohol consumption and usually has a poor prognosis. Therefore, it is important to explore the underlying mechanisms of tumorigenesis and screen the genes and pathways identified from such studies and their role in pathogenesis. The purpose of this study was to identify genes or signal pathways associated with the development of HNSC. Methods: In this study, we downloaded gene expression profiles of GSE53819 from the Gene Expression Omnibus (GEO) database, including 18 HNSC tissues and 18 normal tissues. -
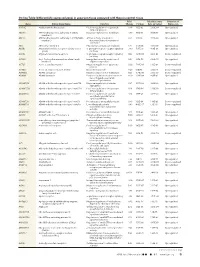
Differentially Expressed Genes in Aneurysm Tissue Compared With
On-line Table: Differentially expressed genes in aneurysm tissue compared with those in control tissue Fold False Discovery Direction of Gene Entrez Gene Name Function Change P Value Rate (q Value) Expression AADAC Arylacetamide deacetylase Positive regulation of triglyceride 4.46 1.33E-05 2.60E-04 Up-regulated catabolic process ABCA6 ATP-binding cassette, subfamily A (ABC1), Integral component of membrane 3.79 9.15E-14 8.88E-12 Up-regulated member 6 ABCC3 ATP-binding cassette, subfamily C (CFTR/MRP), ATPase activity, coupled to 6.63 1.21E-10 7.33E-09 Up-regulated member 3 transmembrane movement of substances ABI3 ABI family, member 3 Peptidyl-tyrosine phosphorylation 6.47 2.47E-05 4.56E-04 Up-regulated ACKR1 Atypical chemokine receptor 1 (Duffy blood G-protein–coupled receptor signaling 3.80 7.95E-10 4.18E-08 Up-regulated group) pathway ACKR2 Atypical chemokine receptor 2 G-protein–coupled receptor signaling 0.42 3.29E-04 4.41E-03 Down-regulated pathway ACSM1 Acyl-CoA synthetase medium-chain family Energy derivation by oxidation of 9.87 1.70E-08 6.52E-07 Up-regulated member 1 organic compounds ACTC1 Actin, ␣, cardiac muscle 1 Negative regulation of apoptotic 0.30 7.96E-06 1.65E-04 Down-regulated process ACTG2 Actin, ␥2, smooth muscle, enteric Blood microparticle 0.29 1.61E-16 2.36E-14 Down-regulated ADAM33 ADAM domain 33 Integral component of membrane 0.23 9.74E-09 3.95E-07 Down-regulated ADAM8 ADAM domain 8 Positive regulation of tumor necrosis 4.69 2.93E-04 4.01E-03 Up-regulated factor (ligand) superfamily member 11 production ADAMTS18 -

Defective Synaptic Connectivity and Axonal Neuropathology in a Human
Defective synaptic connectivity and axonal PNAS PLUS neuropathology in a human iPSC-based model of familial Parkinson’s disease Georgia Kouroupia, Era Taoufika, Ioannis S. Vlachosb, Konstantinos Tsiorasa, Nasia Antonioua, Florentia Papastefanakia, Dafni Chroni-Tzartoua,c, Wolfgang Wrasidlod, Delphine Bohle,1, Dimitris Stellasf, Panagiotis K. Politisg, Kostas Vekrellish, Dimitra Papadimitrioui, Leonidas Stefanish,j, Piotr Bregestovskik, Artemis G. Hatzigeorgioub, Eliezer Masliahd, and Rebecca Matsasa,2 aDepartment of Neurobiology, Hellenic Pasteur Institute, 11521 Athens, Greece; bDIANA-Lab, Hellenic Pasteur Institute, 11521 Athens, Greece; cDepartment of Neurology, Aeginition Hospital, University of Athens Medical School, 11528 Athens, Greece; dDepartment of Neurosciences, University of California, San Diego, La Jolla, CA 92093; eInstitut Pasteur, 75015 Paris, France; fDepartment of Cancer Biology, Biomedical Research Foundation of the Academy of Athens, 11527 Athens, Greece; gCenter for Basic Research, Biomedical Research Foundation of the Academy of Athens, 11527 Athens, Greece; hCenter of Clinical Research, Experimental Surgery and Translational Research, Biomedical Research Foundation of the Academy of Athens, 11527 Athens, Greece; iNeurology Clinic, Henry Dunant Hospital Center, 11526 Athens, Greece; jSecond Department of Neurology, University of Athens Medical School, 11527 Athens, Greece; and kAix-Marseille Université, INSERM, Institut de Neurosciences des Systèmes, 13005 Marseille, France Edited by Solomon H. Snyder, Johns Hopkins University School of Medicine, Baltimore, MD, and approved March 24, 2017 (received for review October 18, 2016) α-Synuclein (αSyn) is the major gene linked to sporadic Parkinson’s number of p.A53T-based in vitro and in vivo animal models have disease (PD), whereas the G209A (p.A53T) αSyn mutation causes a been created for understanding the mechanisms of PD patho- familial form of PD characterized by early onset and a generally genesis and progression and for assisting in drug development. -
CCBE1 Enhances Lymphangiogenesis Via ADAMTS3-Mediated VEGF-C Processing
CCBE1 enhances lymphangiogenesis via ADAMTS3-mediated VEGF-C processing Master’s Thesis Sawan Kumar Jha University of Helsinki Department of Biosciences 2014 Abstract Lymphangiogenesis is the process that leads to the formation of lymphatic vessels from pre-existing vessels. Vascular endothelial growth factor C (VEGF-C), the ma- jor lymphangiogenic growth factor, is produced as an inactive precursor and needs to be proteolytically processed into a mature form in order to activate its receptors VEGFR-3 and VEGFR-2. A deficiency of VEGF-C during embryonic lymphan- giogenesis results in embryonic lethality due to the lack of lymphatic vasculature. Hennekam lymphangiectasia-lymphedema syndrome (OMIM 235510) is in a subset of patients associated with mutations in the collagen- and calcium-binding EGF domains 1 (CCBE1 ) gene. CCBE1 and VEGF-C act at the same stage during em- bryonic lymphangiogenesis and their deficiency results in similar lymphatic defects. The mechanism behind the lymphatic phenotype caused by CCBE1 mutations is un- known. The aim of this study was to investigate the potential link between VEGF-C and CCBE1 that could contribute to the lymphatic phenotype. In this study, 293T cells were used to observe the e↵ect of CCBE1 on VEGF-C pro- cessing. The co-transfection of constructs coding for CCBE1 and VEGF-C showed processing of the inactive pro-VEGF-C into the active, mature form. However, this processing was efficient only in 293T cells. When CCBE1 from 293T supernatant was purified, A disintegrin and metalloproteinase with thrombospondin type 1 mo- tif 3 (ADAMTS3) co-purified with CCBE1. The levels of pro-VEGF-C and active VEGF-C were monitored by immunoblotting or immunoprecipitating metabolically labeled supernatant with specific antibodies or receptors followed by autoradiogra- phy.