Interaction of Chlorpromazine, Fluphenazine and Trifluoperazine with Ocular and Synthetic Melanin in Vitro
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
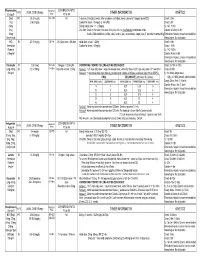
Medication Conversion Chart
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Schizophrenia Care Guide
August 2015 CCHCS/DHCS Care Guide: Schizophrenia SUMMARY DECISION SUPPORT PATIENT EDUCATION/SELF MANAGEMENT GOALS ALERTS Minimize frequency and severity of psychotic episodes Suicidal ideation or gestures Encourage medication adherence Abnormal movements Manage medication side effects Delusions Monitor as clinically appropriate Neuroleptic Malignant Syndrome Danger to self or others DIAGNOSTIC CRITERIA/EVALUATION (PER DSM V) 1. Rule out delirium or other medical illnesses mimicking schizophrenia (see page 5), medications or drugs of abuse causing psychosis (see page 6), other mental illness causes of psychosis, e.g., Bipolar Mania or Depression, Major Depression, PTSD, borderline personality disorder (see page 4). Ideas in patients (even odd ideas) that we disagree with can be learned and are therefore not necessarily signs of schizophrenia. Schizophrenia is a world-wide phenomenon that can occur in cultures with widely differing ideas. 2. Diagnosis is made based on the following: (Criteria A and B must be met) A. Two of the following symptoms/signs must be present over much of at least one month (unless treated), with a significant impact on social or occupational functioning, over at least a 6-month period of time: Delusions, Hallucinations, Disorganized Speech, Negative symptoms (social withdrawal, poverty of thought, etc.), severely disorganized or catatonic behavior. B. At least one of the symptoms/signs should be Delusions, Hallucinations, or Disorganized Speech. TREATMENT OPTIONS MEDICATIONS Informed consent for psychotropic -

Appendix 18: References to Studies from the Previous Guideline
Appendix 18: References to studies from the previous guideline This appendix contains references to the studies from the previous guideline, as they appeared in that guideline. References to studies added to the guideline update are in Appendix 17a-d. Contents Service references…………………………………………………………………..1 Psychology references……………………………………………………………..15 Pharmacology references………………………………………………………….28 Service references Araya2003 {published data only} Araya R, Rojas G, Fritsch R, Gaete J, Rojas M, Simon G, Peters TJ. Treating depression in primary care in low-income women in Santiago, Chile: A randomised controlled trial. Lancet 2003;361(9362):995-1000. Arthur2002 {published data only} Arthur AJ, Jagger C, Lindesay J, Matthews RJ. Evaluating a mental health assessment for older people with depressive symptoms in general practice: a randomised controlled trial. British Journal of General Practice 2002;52(476):202-207. Austin-Los Angeles Austin NK, Liberman RP, King LW, DeRisi WJ. A comparative evaluation of two day hospitals. Goal attainment scaling of behaviour therapy vs. milieu therapy. Journal of Nervous and Mental Disease 1976;163:253-62. Azim-Alberta Azim HF, Weiden TD, Ratcliffe WD, Nutter RW, Dyck RJ, Howarth BG. Current utilization of day hospitalization. Canadian Psychiatric Association Journal 1978;23:557-66 Baker2001 {published data only} Baker R, Reddish S, Robertson N, Hearnshaw H, Jones B. Randomised controlled trial of tailored strategies to implement guidelines for the management of patients with depression in general practice. British Journal of General Practice 2001;51:737-741. Barkley-Ontario Barkley AL, Fagen K, Lawson JS. Day care: can it prevent readmission to a psychiatric hospital? Psychiatric Journal of the University of Ottawa 1989;14:536-41. -

Medications to Be Avoided Or Used with Caution in Parkinson's Disease
Medications To Be Avoided Or Used With Caution in Parkinson’s Disease This medication list is not intended to be complete and additional brand names may be found for each medication. Every patient is different and you may need to take one of these medications despite caution against it. Please discuss your particular situation with your physician and do not stop any medication that you are currently taking without first seeking advice from your physician. Most medications should be tapered off and not stopped suddenly. Although you may not be taking these medications at home, one of these medications may be introduced while hospitalized. If a hospitalization is planned, please have your neurologist contact your treating physician in the hospital to advise which medications should be avoided. Medications to be avoided or used with caution in combination with Selegiline HCL (Eldepryl®, Deprenyl®, Zelapar®), Rasagiline (Azilect®) and Safinamide (Xadago®) Medication Type Medication Name Brand Name Narcotics/Analgesics Meperidine Demerol® Tramadol Ultram® Methadone Dolophine® Propoxyphene Darvon® Antidepressants St. John’s Wort Several Brands Muscle Relaxants Cyclobenzaprine Flexeril® Cough Suppressants Dextromethorphan Robitussin® products, other brands — found as an ingredient in various cough and cold medications Decongestants/Stimulants Pseudoephedrine Sudafed® products, other Phenylephrine brands — found as an ingredient Ephedrine in various cold and allergy medications Other medications Linezolid (antibiotic) Zyvox® that inhibit Monoamine oxidase Phenelzine Nardil® Tranylcypromine Parnate® Isocarboxazid Marplan® Note: Additional medications are cautioned against in people taking Monoamine oxidase inhibitors (MAOI), including other opioids (beyond what is mentioned in the chart above), most classes of antidepressants and other stimulants (beyond what is mentioned in the chart above). -

Long-Acting Injectable (LAI) Dosing Chart
Long-Acting Injectable (LAI) Dosing Chart Drug FDA-Approved Dosing Earliest Time to Missed Dose Indications Next Dose ABILIFY • Schizophrenia Initiation No sooner than 26 days If 2nd or 3rd dose missed: MAINTENA® • Maintenance 400 mg IM q month + aripiprazole 10-20 mg after last injection Timing of Missed Dose Dosing (aripiprazole)1 Monotherapy of PO daily x14 days OR if stable on another PO >4 weeks and <5 weeks • Administer missed dose ASAP. Bipolar 1 Disorder antipsychotic, + overlap x14 days >5 weeks • Restart next injection with oral Dosage Forms aripiprazole x 14 days. 300 mg and 400 mg Maintenance pre-filled syringe or 400 mg IM q month If 4th or subsequent doses missed: single-use vial Timing of Missed Dose Dosing Dose Adjustments >4 weeks and <6 weeks • Administer missed dose ASAP. Adverse reactions or known CYP2D6 poor >5 weeks • Restart next injection with oral metabolizers: aripiprazole x 14 days. 300 mg IM q month ARISTADA INITIO®, • Schizophrenia Initiation No sooner than 14 days ARISTADA® Option #1: ARISTADA INITIO® 675 mg IM x1 after last injection Management of a Missed Maintenance Dose (aripiprazole dose + aripiprazole 30 mg PO x1 dose + first Last Time Since Last Injection lauroxil)2,3 dose of *ARISTADA® IM (441 mg, 662 mg, 882 ARISTADA® mg, or 1064 mg) Dose Dosage Forms *first dose of ARISTADA® may be given 441 mg ≤6 weeks >6 and ≤7 weeks >7 weeks ARISTADA INITIO® 675 together or within 10 days* 662 mg ≤8 weeks >8 and ≤12 weeks >12 weeks mg pre-filled syringe 882 mg ≤8 weeks >8 and ≤12 weeks >12 weeks ARISTADA® 441 -

Aripiprazole Long-Acting Injection(Abilify Maintena
Aripiprazole Long-acting Injection Aripiprazole Long-acting Injection (Abilify Maintena, ARISTADA) National Drug Monograph October 2013; Addendum March 2016 VA Pharmacy Benefits Management Services, Medical Advisory Panel, and VISN Pharmacist Executives The purpose of VA PBM Services drug monographs is to provide a comprehensive drug review for making formulary decisions. These documents will be updated when new clinical data warrant additional formulary discussion. Documents will be placed in the Archive section when the information is deemed to be no longer current. Executive Summary: Aripiprazole long-acting intramuscular injection (LAIM) is sixth antipsychotic and the fourth second generation antipsychotic to be marketed in a long-acting injection formulation. LAIM antipsychotics on the VA National Formulary: haloperidol and fluphenazine decanoate; risperidone; and paliperidone. Aripiprazole LAIM has FDA label indication for the treatment of schizophrenia. The initial and usual maintenance dose of aripiprazole is 400 mg once a month. The dose can be reduced to 300 mg or 200 mg monthly based on drug interactions or tolerability. Patients should have established tolerability to aripiprazole before receipt of the LAIM formulation. Oral aripiprazole, 10-30 mg/day, or another oral antipsychotic must be continued for 2-weeks after the initial dose, and then discontinued. Aripiprazole LAIM’s efficacy and safety are based on experience with the oral formulation as well as pharmacokinetic trials and a 52-week randomized, double-blind, placebo-controlled trial. The randomized trial was terminated early because the difference in time to relapse met a predetermined statistically significant threshold (p=0.001) favoring aripiprazole LAIM [HR 5.03, 95% CI 3.15-8.02]. -

207533Orig1s000
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 207533Orig1s000 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 207553 Priority or Standard Standard Submit Date(s) 8/22/2014 Received Date(s) 8/22/2014 PDUFA Goal Date 8/22/2015 Division / Office DPP/ODE1 Reviewer Name(s) Lucas Kempf, MD Review Completion Date October 2, 2015 Established Name Aripiprazole lauroxil (Proposed) Trade Name Aristada Therapeutic Class Antipsychotic Applicant Alkermes Inc. Formulation(s) Extended release injection Dosing Regimen Suspension/ IM up to 6 weeks Indication(s) Schizophrenia Intended Population(s) Schizophrenia Template Version: March 6, 2009 Reference ID: 3806701 Clinical Review Lucas Kempf, MD NDA 207533 Aristada, Aripiprazole lauroxil Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 7 1.1 Recommendation on Regulatory Action ............................................................. 7 1.2 Risk Benefit Assessment .................................................................................... 7 1.3 Recommendations for Postmarket Risk Evaluation and Mitigation Strategies ... 8 1.4 Recommendations for Postmarket Requirements and Commitments ................ 8 2 INTRODUCTION AND REGULATORY BACKGROUND ........................................ 8 2.1 Product Information ............................................................................................ 8 2.2 Currently Available Treatments for Proposed Indications .................................. -

Fluphenazine Hydrochloride Tablets, USP
FLUPHENAZINE HYDROCHLORIDE- fluphenazine hydrochloride tablet, film coated Lannett Company, Inc. ---------- Fluphenazine Hydrochloride Tablets, USP Rx only WARNING Increased Mortality in Elderly Patients with Dementia-Related Psychosis: Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug- treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. Fluphenazine hydrochloride is not approved for the treatment of patients with dementia-related psychosis (see WARNINGS). DESCRIPTION Fluphenazine hydrochloride is a trifluoromethyl phenothiazine derivative intended for the management of schizophrenia. The chemical designation is 4-[3-[2-(Trifluoromethyl) phenothiazin-10-yl] propyl]-1- piperazineethanol dihydrochloride. The structural formula is represented below: Fluphenazine Hydrochloride Tablets, USP, for oral administration, contain 1 mg, 2.5 mg, 5 mg, or 10 mg fluphenazine hydrochloride, USP per tablet. -

Adverse Side Effects in Horses Following the Administration of Fluphenazine Decanoate
MEDICINE I Adverse Side Effects in Horses Following the Administration of Fluphenazine Decanoate John D. Baird, BVSc, PhD*; and George A. Maylin, DVM, MS, PhD Adverse behavioral side effects may occur in some horses after the intramuscular administration of fluphenazine decanoate. In severe cases, these side effects are difficult to manage and may be life-threatening to the horse, stable personnel, and treating veterinarians. The drug is not approved for use in the horse, and testing for fluphenazine is now conducted by various regulatory organizations involved in racing and equestrian competitions. Authors’ addresses: Department of Clinical Stud- ies, Ontario Veterinary College, University of Guelph, Guelph, ON Canada N1G 2W1 (Baird); and Equine Drug Testing and Research Program, New York Drug Testing and Research Program Mor- risville State College, 777 Warren Road, Ithaca, NY 14850 (Maylin); e-mail: jbaird@ovc. uoguelph.ca. *Corresponding author. © 2011 AAEP. 1. Introduction more commonly used clinically than fluphenazine Fluphenazine is a trifluoromethyl phenothiazine enanthate. In humans, the most common dosage of derivative (Fig. 1). The systematic International fluphenazine decanoate is 25 mg (range, 12.5 to 50 Union of Pure and Applied Chemistry (IUPAC) mg), with patients injected once every 2 weeks name of fluphenazine is 2-[4-[3-[2-(trifluoromethyl) (range, 1 to 4 weeks).1,5,6 After the IM injection of phenothiazin-10-yl]propyl]-piperazin-1-yl]ethanol fluphenazine decanoate, the drug gradually diffuses and it has the following structural formula. from the oily vehicle into the surrounding lymph nodes and tissues. Once the drug enters the tissue, 2. Clinical Pharmacology it is rapidly hydrolyzed by ubiquitous esterases, and Fluphenazine is a highly potent antipsychotic drug the parent compound (fluphenazine) is released. -

Long-Acting Injectable Antipsychotic Medications and Conversion Dosing
Long-Acting Injectable Antipsychotic Medications and Conversion Dosing Practical Tips for Practice Long-Acting Injectable Antipsychotic Medications and Conversion Dosing: Practical Tips for Practice Loading Usual Starting Dose Target Dose Max Dose Dosing Interval Dose Oral or Other Overlap* Injection Site Special Notes Option 12.5-50 mg Continue oral, decreasing dose by half after Not Gluteal Z-track recommended4 Fluphenazine Decanoate1,2 12.5-25 mg IM/SQ (interval determined by 100 mg/dose Q 2-4 weeks first injection, d/c oral therapy after second established Deltoid Use dry syringe/needle patient response) injection1,3 100 mg/1st Z-track recommended4 10-15X daily PO dose Continue oral for the first 2 to 3 injections if not Gluteal Haloperidol Decanoate5 10-20X daily PO dose IM injection; 450 mg/ Q 4 weeks Yes Do not administer > 3 ml per IM Q 4 weeks using loading dose3 Deltoid 4 weeks injection site Risperidone Long-acting 25-50 mg IM Q 2 Oral risperidone (or other oral antipsychotic) 25 mg IM Q 2 weeks + PO Gluteal injection weeks (range 12.5-50 50 mg Q 2 weeks Q 2 weeks No should be given with 1st injection and None dose for 3 weeks Deltoid (Risperdal Consta®)6 mg) continued for 3 weeks Risperidone “extended release” 90 or 120 mg SQ Q 120 mg SQ Q Establishment of tolerability by prior oral Abdomen only 90 or 120 mg SQ Q month Q Monthly n/a Abdomen- SQ (Perseris®)7 month month administration is recommended Inject slow and steady Deltoid Deltoid Day 1 and 8 Inj.; 234 mg IM day 1, then 156 Gluteal Paliperidone Palmitate monthly 117 -

Perphenazine (Trilafon)
Perphenazine (Trilafon) Generic name: Perphenazine Available strengths: 2 mg, 4 mg, 8 mg, 16 mg tablets; 16 mg/5 mL oral concentrate Available in generic: Yes Drug class: First-generation (conventional) antipsychotic General Information Perphenazine (Trilafon) belongs to the class of antipsychotics known as the first-generation antipsychotics, sometimes referred to as conventional or typical antipsychotics. Perphenazine was sold under the brand Trilafon, but Trilafon has since been discontinued. Currently, only generic perphenazine is available. The first-generation antipsychotics represent an older class of antipsychotics that have been the standard for treating psychotic dis- orders for many decades. When compared with a newer class of second-generation antipsychotics, these ear- lier antipsychotics are referred to as typical or conventional because they lack the wider spectrum of therapeutic activity. The first-generation antipsychotics are also more likely to induce side effects that cause movement dis- orders, such as extrapyramidal symptoms (EPS) and tardive dyskinesia (TD), than the newer antipsychotics. Perphenazine is an intermediate-potency antipsychotic relative to other first-generation antipsychotics such as chlorpromazine and thioridazine, which are low-potency agents, and fluphenazine and Haldol (halo- peridol), which are high-potency antipsychotics. Perphenazine is moderately sedating and less likely to lower blood pressure than the lower-potency agents. Perphenazine produces fewer EPS than the high-potency an- tipsychotics. Perphenazine was approved by the U.S. Food and Drug Administration for treatment of psychotic disor- ders, including schizophrenia, schizoaffective disorder, and drug-induced psychosis. The use of a medication for its approved indications is called its labeled use. In clinical practice, however, physicians often prescribe medications for unlabeled (“off-label”) uses when published clinical studies, case reports, or their own clinical experiences support the efficacy and safety of those treatments. -

Profiles of the Affinity of Antipsychotic Drugs for Neurotransmitter Receptors
Kitakanto Med.J. 87 48( 2 ) : 87•`102, 1998 PROFILES OF THE AFFINITY OF ANTIPSYCHOTIC DRUGS FOR NEUROTRANSMITTER RECEPTORS AND THEIR CLINICAL IMPLICATION Kimie Yonemura, Kazuo Miyanaga, and Yukiteru Machiyama Departmentof Neuropsychiatry,Gunma UniversitySchool of Medicine Abstract : The binding affinities of 12 different neurotransmitter receptors were measured using a radio-labeled receptor assay. The profiles of the pharmacological affinities of 20 antipsychotic drugs were assessed. Based on the Ki values for the D2 receptor, 5HT2 receptor, and ƒ¿1 receptor, the antipsychotic drugs were classified into five groups (i.e., three anti-DA activity predominant groups and two anti-5HT activity predominant groups). A theoretical basis for drug choice in clinical settings is also proposed. We recommend that the groups of the drugs having predominantly anti-DA activity be used in the treatment of hallucinations and delusions and that the drugs in the anti-5HT activity predominant group be used to treat hypobulia. If it is necessary to administer two or more of these drugs together, we propose that they should be drugs with different profiles. Extrapyramidal symptoms, side effects of antipsychotic drugs, were interpreted in terms of interaction among the anti-D, activity, anti-5HT2 activity, and anti-Ach activity. Key words : Antipsychotic drugs, Anti-DA action, Anti-5HT action, Anti-adrenergic action, Extrapyramidal symptoms. (Kitakanto Med.J. 48 (2) : 87•`102, 1998) number of reports on the affinity of antipsychotic INTRODUCTION agents for receptors have been published both in Japan A variety of antipsychotic agents have been devel- and other countrieso. The reports, however, have often oped for clinical use since 1952, when the effectiveness dealt with drugs that are seldom used or not used at all of chlorpromazine in treating schizophrenia was first at present in Japan.