Folate Receptor Β Regulates Integrin Cd11b/CD18 Adhesion of a Macrophage Subset to Collagen
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Human CD64 / FCGR1A Protein (His Tag), Biotinylated
Human CD64 / FCGR1A Protein (His Tag), Biotinylated Catalog Number: 10256-H08S-B General Information SDS-PAGE: Gene Name Synonym: CD64; Fc gamma RI Protein Construction: A DNA sequence encoding the human FCGR1A (NP_000557.1) (Met1- Pro288) was expressed with a polyhistidine tag at the C-terminus. The purified protein was biotinylated in vitro. Source: Human Expression Host: CHO Stable Cells QC Testing Purity: > 95 % as determined by SDS-PAGE. Bio Activity: Protein Description Measured by its binding ability in a functional ELISA.Immobilized High affinity immunoglobulin gamma Fc receptor I, also known as FCGR1 biotinylated Human CD64 Protein (Cat:10256-H08S-B)at 10 μg/mL can and CD64, is an integral membraneglycoprotein and a member of the bind human IgG1,The EC50 of human IgG1 is 6-14 ng/mL. immunoglobulin superfamily. CD64 is a high affinity receptor for the Fc region of IgG gamma and functions in both innate and adaptive immune Endotoxin: responses. Receptors that recognize the Fc portion of IgG function in the regulation of immune response and are divided into three classes < 1.0 EU per μg protein as determined by the LAL method. designated CD64, CD32, and CD16. CD64 is structurally composed of asignal peptidethat allows its transport to the surface of a cell, Stability: threeextracellularimmunoglobulin domainsof the C2-type that it uses to Samples are stable for up to twelve months from date of receipt at -70 ℃ bind antibody, a hydrophobictransmembrane domain, and a short cytoplasmic tail. CD64 isconstitutivelyfound on only macrophages and Predicted N terminal: Gln 16 monocytes, but treatment of polymorphonuclear leukocyteswith cytokines likeIFNγandG-CSFcan induce CD64 expression on these cells. -

The Urokinase Receptor Induces a Mesenchymal Gene Expression Signature in Glioblastoma Cells and Promotes Tumor Cell Survival In
www.nature.com/scientificreports OPEN The Urokinase Receptor Induces a Mesenchymal Gene Expression Signature in Glioblastoma Cells and Received: 20 October 2017 Accepted: 2 February 2018 Promotes Tumor Cell Survival in Published: xx xx xxxx Neurospheres Andrew S. Gilder1, Letizia Natali1, Danielle M. Van Dyk1, Cristina Zalfa1, Michael A. Banki1, Donald P. Pizzo1, Huawei Wang 1, Richard L. Klemke1, Elisabetta Mantuano 1,2 & Steven L. Gonias1 PLAUR encodes the urokinase receptor (uPAR), which promotes cell survival, migration, and resistance to targeted cancer therapeutics in glioblastoma cells in culture and in mouse model systems. Herein, we show that patient survival correlates inversely with PLAUR mRNA expression in gliomas of all grades, in glioblastomas, and in the subset of glioblastomas that demonstrate the mesenchymal gene expression signature. PLAUR clusters with genes that defne the more aggressive mesenchymal subtype in transcriptome profles of glioblastoma tissue and glioblastoma cells in neurospheres, which are enriched for multipotent cells with stem cell-like qualities. When PLAUR was over-expressed or silenced in glioblastoma cells, neurosphere growth and expression of mesenchymal subtype biomarkers correlated with uPAR abundance. uPAR also promoted glioblastoma cell survival in neurospheres. Constitutively-active EGF Receptor (EGFRvIII) promoted neurosphere growth; however, unlike uPAR, EGFRvIII did not induce the mesenchymal gene expression signature. Immunohistochemical analysis of human glioblastomas showed that uPAR is typically expressed by a small sub-population of the cancer cells; it is thus reasonable to conclude that this subpopulation of cells is responsible for the efects of PLAUR on patient survival. We propose that uPAR-expressing glioblastoma cells demonstrate a mesenchymal gene signature, an increased capacity for cell survival, and stem cell-like properties. -

Cómo Citar El Artículo Número Completo Más Información Del
Revista de Biología Tropical ISSN: 0034-7744 ISSN: 0034-7744 Universidad de Costa Rica Alpízar-Alpízar, Warner; Malespín-Bendaña, Wendy; Une, Clas; Ramírez-Mayorga, Vanessa Relevance of the plasminogen activation system in the pathogenesis and progression of gastric cancer Revista de Biología Tropical, vol. 66, núm. 1, 2018, pp. 28-47 Universidad de Costa Rica DOI: 10.15517/rbt.v66i1.29014 Disponible en: http://www.redalyc.org/articulo.oa?id=44955366003 Cómo citar el artículo Número completo Sistema de Información Científica Redalyc Más información del artículo Red de Revistas Científicas de América Latina y el Caribe, España y Portugal Página de la revista en redalyc.org Proyecto académico sin fines de lucro, desarrollado bajo la iniciativa de acceso abierto Relevance of the plasminogen activation system in the pathogenesis and progression of gastric cancer Warner Alpízar-Alpízar1,2*, Wendy Malespín-Bendaña3, Clas Une3 & Vanessa Ramírez-Mayorga3,4 1. Centro de Investigación en Estructuras Microscópicas (CIEMic), Universidad de Costa Rica, San José, Costa Rica; [email protected] 2. Departamento de Bioquímica, Escuela de Medicina, Universidad de Costa Rica, San José, Costa Rica. 3. Instituto de Investigaciones en Salud (INISA), Universidad de Costa Rica, San José, Costa Rica; [email protected], [email protected], [email protected] 4. Sección de Nutrición Pública, Escuela de Nutrición, Universidad de Costa Rica, San José, Costa Rica. * Correspondence Received 14-VII-2017. Corrected 18-X-2017. Accepted 16-XI-2017. Abstract: Gastric cancer is ranked as the third death-causing cancer and one of the most incident malignancies worldwide. -

Receptor-Like 1 and -Like 2 Is an Endogenous Ligand for Formyl Peptide Through a Urokinase Receptor Epitope That Urokinase Induc
Urokinase Induces Basophil Chemotaxis through a Urokinase Receptor Epitope That Is an Endogenous Ligand for Formyl Peptide Receptor-Like 1 and -Like 2 This information is current as of May 10, 2019. Amato de Paulis, Nunzia Montuori, Nella Prevete, Isabella Fiorentino, Francesca Wanda Rossi, Valeria Visconte, Guido Rossi, Gianni Marone and Pia Ragno J Immunol 2004; 173:5739-5748; ; doi: 10.4049/jimmunol.173.9.5739 http://www.jimmunol.org/content/173/9/5739 Downloaded from References This article cites 65 articles, 33 of which you can access for free at: http://www.jimmunol.org/content/173/9/5739.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on May 10, 2019 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2004 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Urokinase Induces Basophil Chemotaxis through a Urokinase Receptor Epitope That Is an Endogenous Ligand for Formyl Peptide Receptor-Like 1 and -Like 21 Amato de Paulis,* Nunzia Montuori,† Nella Prevete,* Isabella Fiorentino,* Francesca Wanda Rossi,* Valeria Visconte,‡ Guido Rossi,‡ Gianni Marone,2* and Pia Ragno† Basophils circulate in the blood and are able to migrate into tissues at sites of inflammation. -

FCGR1A Recombinant Protein
FCGR1A Recombinant Protein CATALOG NUMBER: 96-305 The purity of rh CD64 / FCGR1A was determined by DTT-reduced (+) SDS- PAGE and staining overnight with Coomassie Blue. Specifications SPECIES: Human SOURCE SPECIES: HEK293 cells SEQUENCE: Gln 16 - Pro 288 FUSION TAG: His Tag TESTED APPLICATIONS: WB APPLICATIONS: This recombinant protein can be used for WB. For research use only. BIOLOGICAL ACTIVITY: Measured by its ability to bind human IgG1 in the SPR assay (Biacore 2000) with the KD < 5 nM. Measured by its binding ability in a functional ELISA. Immobilized Human IgG4 at 10ug/mL (100 µl/well),can bind Human Fc gamma RI, His Tag (Cat# FCA-H52H2) with a linear of 0.1-2 ng/mL. Properties PURITY: >90% as determined by SDS-PAGE. PREDICTED MOLECULAR 32.5 kDa WEIGHT: PHYSICAL STATE: Lyopholized BUFFER: PBS, pH7.4 STORAGE CONDITIONS: Lyophilized Protein should be stored at -20˚C or lower for long term storage. Upon reconstitution, working aliquots should be stored at -20˚C or -70˚C. Avoid repeated freeze-thaw cycles. Additional Info ALTERNATE NAMES: FCGR1A, FCG1, FCGR1, IGFR1, CD64, CD64A, FCRI ACCESSION NO.: AAH32634 Background Receptors that recognize the Fc portion of IgG are divided into three groups designated Fc gamma RI, RII, and RIII, also known respectively as CD64, CD32, and CD16. Fc gamma RI binds IgG with high affinity and functions during early immune responses. Fc gamma RII and RIII are low affinity receptors that recognize IgG as aggregates surrounding multivalent antigens during late immune responses. High affinity immunoglobulin gamma Fc receptor I is also known as FCGR1A, FCG1, FCGR1, CD64 and IGFR1, is a type of integral membrane glycoprotein that binds monomeric IgG-type antibodies with high affinity, which belongs to the immunoglobulin superfamily or FCGR1 family. -

Supplementary Table 1: Adhesion Genes Data Set
Supplementary Table 1: Adhesion genes data set PROBE Entrez Gene ID Celera Gene ID Gene_Symbol Gene_Name 160832 1 hCG201364.3 A1BG alpha-1-B glycoprotein 223658 1 hCG201364.3 A1BG alpha-1-B glycoprotein 212988 102 hCG40040.3 ADAM10 ADAM metallopeptidase domain 10 133411 4185 hCG28232.2 ADAM11 ADAM metallopeptidase domain 11 110695 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 195222 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 165344 8751 hCG20021.3 ADAM15 ADAM metallopeptidase domain 15 (metargidin) 189065 6868 null ADAM17 ADAM metallopeptidase domain 17 (tumor necrosis factor, alpha, converting enzyme) 108119 8728 hCG15398.4 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 117763 8748 hCG20675.3 ADAM20 ADAM metallopeptidase domain 20 126448 8747 hCG1785634.2 ADAM21 ADAM metallopeptidase domain 21 208981 8747 hCG1785634.2|hCG2042897 ADAM21 ADAM metallopeptidase domain 21 180903 53616 hCG17212.4 ADAM22 ADAM metallopeptidase domain 22 177272 8745 hCG1811623.1 ADAM23 ADAM metallopeptidase domain 23 102384 10863 hCG1818505.1 ADAM28 ADAM metallopeptidase domain 28 119968 11086 hCG1786734.2 ADAM29 ADAM metallopeptidase domain 29 205542 11085 hCG1997196.1 ADAM30 ADAM metallopeptidase domain 30 148417 80332 hCG39255.4 ADAM33 ADAM metallopeptidase domain 33 140492 8756 hCG1789002.2 ADAM7 ADAM metallopeptidase domain 7 122603 101 hCG1816947.1 ADAM8 ADAM metallopeptidase domain 8 183965 8754 hCG1996391 ADAM9 ADAM metallopeptidase domain 9 (meltrin gamma) 129974 27299 hCG15447.3 ADAMDEC1 ADAM-like, -

The Urokinase Receptor: a Multifunctional Receptor in Cancer Cell Biology
International Journal of Molecular Sciences Review The Urokinase Receptor: A Multifunctional Receptor in Cancer Cell Biology. Therapeutic Implications Anna Li Santi 1,†, Filomena Napolitano 2,†, Nunzia Montuori 2 and Pia Ragno 1,* 1 Department of Chemistry and Biology, University of Salerno, Fisciano, 84084 Salerno, Italy; [email protected] 2 Department of Translational Medical Sciences, “Federico II” University, 80135 Naples, Italy; fi[email protected] (F.N.); [email protected] (N.M.) * Correspondence: [email protected] † Equal contribution. Abstract: Proteolysis is a key event in several biological processes; proteolysis must be tightly con- trolled because its improper activation leads to dramatic consequences. Deregulation of proteolytic activity characterizes many pathological conditions, including cancer. The plasminogen activation (PA) system plays a key role in cancer; it includes the serine-protease urokinase-type plasminogen activator (uPA). uPA binds to a specific cellular receptor (uPAR), which concentrates proteolytic activity at the cell surface, thus supporting cell migration. However, a large body of evidence clearly showed uPAR involvement in the biology of cancer cell independently of the proteolytic activity of its ligand. In this review we will first describe this multifunctional molecule and then we will discuss how uPAR can sustain most of cancer hallmarks, which represent the biological capabilities acquired during the multistep cancer development. Finally, we will illustrate the main data available in the literature on uPAR as a cancer biomarker and a molecular target in anti-cancer therapy. Citation: Li Santi, A.; Napolitano, F.; Montuori, N.; Ragno, P. The Keywords: urokinase receptor; uPAR; cancer hallmarks Urokinase Receptor: A Multifunctional Receptor in Cancer Cell Biology. -

Product Datasheet: OAAB02345
Aviva Systems Biology FCGR1A Antibody - middle region (OAAB02345) Product Number OAAB02345 Product Page http://www.avivasysbio.com/fcgr1a-antibody-center-region-oaab02345.html Product Name FCGR1A Antibody - middle region (OAAB02345) Size 400ul Gene Symbol FCGR1A Alias Symbols CD_antigen=CD64, Flags: Precursor, Fc-gamma RI, FcRI, Fc-gamma RIA, FcgammaRIa, High affinity immunoglobulin gamma Fc receptor I, IgG Fc receptor I,IGFR1, FCGR1, FCG1, FCGR1A Molecular Weight 42632 Da Product Format Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. Isotype Rabbit Ig NCBI Gene Id 2209 Host Rabbit Clonality Polyclonal Concentration Approximately 0.5mg/ml. Actual concentration varies with each lot. Reconstitution and Maintain refrigerated at 2-8 deg C for up to 6 months. For long term storage store at -20 Storage deg C in small aliquots to prevent freeze-thaw cycles. Application Info WB: 1:1000 IHC-P: 1:50-100 FC: 1:10-50 Lead Time Domestic: within 1 week delivery International: 1 week *** Required Wet/Dry Ice Surcharge will automatically be applied upon checkout for the shipment. See Surcharges Immunogen This FCGR1A antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between amino acid 217-245 from the Central region of human FCGR1A. Tissue Tool Find tissues and cell lines supported by DNA array analysis to express FCGR1A. Swissprot Id P12314 Protein Name High affinity immunoglobulin gamma Fc receptor I Sample Type FCGR1A is strongly supported by BioGPS gene expression data to be expressed in Confirmation NCI-H460 Protein Accession NP_000557.1 # RNA Seq Find tissues and cell lines supported by RNA-seq analysis to express FCGR1A. -

Gene List HTG Edgeseq Immuno-Oncology Assay
Gene List HTG EdgeSeq Immuno-Oncology Assay Adhesion ADGRE5 CLEC4A CLEC7A IBSP ICAM4 ITGA5 ITGB1 L1CAM MBL2 SELE ALCAM CLEC4C DST ICAM1 ITGA1 ITGA6 ITGB2 LGALS1 MUC1 SVIL CDH1 CLEC5A EPCAM ICAM2 ITGA2 ITGAL ITGB3 LGALS3 NCAM1 THBS1 CDH5 CLEC6A FN1 ICAM3 ITGA4 ITGAM ITGB4 LGALS9 PVR THY1 Apoptosis APAF1 BCL2 BID CARD11 CASP10 CASP8 FADD NOD1 SSX1 TP53 TRAF3 BCL10 BCL2L1 BIRC5 CASP1 CASP3 DDX58 NLRP3 NOD2 TIMP1 TRAF2 TRAF6 B-Cell Function BLNK BTLA CD22 CD79A FAS FCER2 IKBKG PAX5 SLAMF1 SLAMF7 SPN BTK CD19 CD24 EBF4 FASLG IKBKB MS4A1 RAG1 SLAMF6 SPI1 Cell Cycle ABL1 ATF1 ATM BATF CCND1 CDK1 CDKN1B NCL RELA SSX1 TBX21 TP53 ABL2 ATF2 AXL BAX CCND3 CDKN1A EGR1 REL RELB TBK1 TIMP1 TTK Cell Signaling ADORA2A DUSP4 HES1 IGF2R LYN MAPK1 MUC1 NOTCH1 RIPK2 SMAD3 STAT5B AKT3 DUSP6 HES5 IKZF1 MAF MAPK11 MYC PIK3CD RNF4 SOCS1 STAT6 BCL6 ELK1 HEY1 IKZF2 MAP2K1 MAPK14 NFATC1 PIK3CG RORC SOCS3 SYK CEBPB EP300 HEY2 IKZF3 MAP2K2 MAPK3 NFATC3 POU2F2 RUNX1 SPINK5 TAL1 CIITA ETS1 HEYL JAK1 MAP2K4 MAPK8 NFATC4 PRKCD RUNX3 STAT1 TCF7 CREB1 FLT3 HMGB1 JAK2 MAP2K7 MAPKAPK2 NFKB1 PRKCE S100B STAT2 TYK2 CREB5 FOS HRAS JAK3 MAP3K1 MEF2C NFKB2 PTEN SEMA4D STAT3 CREBBP GATA3 IGF1R KIT MAP3K5 MTDH NFKBIA PYCARD SMAD2 STAT4 Chemokine CCL1 CCL16 CCL20 CCL25 CCL4 CCR2 CCR7 CX3CL1 CXCL12 CXCL3 CXCR1 CXCR6 CCL11 CCL17 CCL21 CCL26 CCL5 CCR3 CCR9 CX3CR1 CXCL13 CXCL5 CXCR2 MST1R CCL13 CCL18 CCL22 CCL27 CCL7 CCR4 CCRL2 CXCL1 CXCL14 CXCL6 CXCR3 PPBP CCL14 CCL19 CCL23 CCL28 CCL8 CCR5 CKLF CXCL10 CXCL16 CXCL8 CXCR4 XCL2 CCL15 CCL2 CCL24 CCL3 CCR1 CCR6 CMKLR1 CXCL11 CXCL2 CXCL9 CXCR5 -
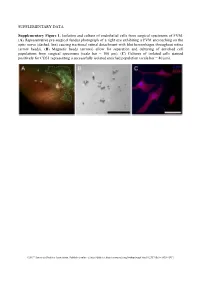
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Identi Cation of Key Survival Genes of the Tumor Microenvironment And
Identication of key survival genes of the tumor microenvironment and immune inltration in head and neck squamous cell carcinoma Min Wang Department of Otorhinolaryngology, The First Aliated Hospital of Chongqing Medical University Tao Lu Department of Otorhinolaryngology, The First Aliated Hospital of Chongqing Medical University Yanshi Li Department of Otorhinolaryngology, The First Aliated Hospital of Chongqing Medical University Min Pan Department of Otorhinolaryngology, The First Aliated Hospital of Chongqing Medical University Zhihai Wang Department of Otorhinolaryngology, The First Aliated Hospital of Chongqing Medical University Guohua Hu ( [email protected] ) Department of Otorhinolaryngology, The First Aliated Hospital of Chongqing Medical University Research Article Keywords: Head and neck cancer, Tumor microenvironment, Immune scores, Bioinformatics analysis, Survival Posted Date: March 13th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-283947/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/23 Abstract Background: Head and neck cancer (HNC) are highly aggressive solid tumors with poor prognoses. The tumor microenvironment (TME) plays a critical role in angiogenesis, invasion, and metastasis of HNC. In the TME, immune and stromal cells inuence tumor initiation, response, and therapy. Our study aimed to evaluate the progression and prognosis of HNC by analyzing the key genes involved in immunization and stromal cells. Methods: Gene expression proles, demographics, and survival data were downloaded from the TCGA database. Patients with HNC were divided into high immune/stromal score groupss or low immune/stromal score groups based on the ESTIMATE algorithm. Differentially expressed genes (DEGs) were identied via functional enrichment analysis and protein-protein interaction networks, and survival analysis based on DEGs was also performed. -

Soluble Urokinase Plasminogen Activator Receptor Predicts
1112 Diabetes Care Volume 42, June 2019 Viktor Rotbain Curovic,1 Simone Theilade,1 Soluble Urokinase Plasminogen Signe A. Winther,1 Nete Tofte,1 Jesper Eugen-Olsen,2 Frederik Persson,1 Activator Receptor Predicts Tine W. Hansen,1 Jørgen Jeppesen,3,4 and Cardiovascular Events, Kidney Peter Rossing1,4 Function Decline, and Mortality in Patients With Type 1 Diabetes Diabetes Care 2019;42:1112–1119 | https://doi.org/10.2337/dc18-1427 OBJECTIVE Soluble urokinase plasminogen activator receptor (suPAR) is an important in- flammatory biomarker implicated in endothelial and podocyte dysfunction. How- ever, suPAR’s predictive qualities for complications in type 1 diabetes have yet to be determined. We investigated the prognostic value of suPAR for the development of cardiovascularevents,declineinrenalfunction, andmortalityinpatientswith type1 diabetes. RESEARCH DESIGN AND METHODS We included 667 patients with type 1 diabetes with various degrees of albuminuria inaprospective study. End points were cardiovascular events (cardiovascular death, nonfatal acute myocardial infarction, nonfatal stroke, or coronary or peripheral arterial interventions), estimated glomerular filtration rate (eGFR) decline ‡30%, progressionfromlowertohigheralbuminuricstate,developmentofend-stagerenal disease (ESRD), and mortality. Follow-up was 5.2–6.2 years. Results were adjusted for known risk factors. Hazard ratios (HRs) are presented per doubling of suPAR with 95% CI. Relative integrated discrimination improvement (rIDI) was calculated. 1Steno Diabetes Center Copenhagen,