Soluble Urokinase Plasminogen Activator Receptor Predicts
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Urokinase Receptor Induces a Mesenchymal Gene Expression Signature in Glioblastoma Cells and Promotes Tumor Cell Survival In
www.nature.com/scientificreports OPEN The Urokinase Receptor Induces a Mesenchymal Gene Expression Signature in Glioblastoma Cells and Received: 20 October 2017 Accepted: 2 February 2018 Promotes Tumor Cell Survival in Published: xx xx xxxx Neurospheres Andrew S. Gilder1, Letizia Natali1, Danielle M. Van Dyk1, Cristina Zalfa1, Michael A. Banki1, Donald P. Pizzo1, Huawei Wang 1, Richard L. Klemke1, Elisabetta Mantuano 1,2 & Steven L. Gonias1 PLAUR encodes the urokinase receptor (uPAR), which promotes cell survival, migration, and resistance to targeted cancer therapeutics in glioblastoma cells in culture and in mouse model systems. Herein, we show that patient survival correlates inversely with PLAUR mRNA expression in gliomas of all grades, in glioblastomas, and in the subset of glioblastomas that demonstrate the mesenchymal gene expression signature. PLAUR clusters with genes that defne the more aggressive mesenchymal subtype in transcriptome profles of glioblastoma tissue and glioblastoma cells in neurospheres, which are enriched for multipotent cells with stem cell-like qualities. When PLAUR was over-expressed or silenced in glioblastoma cells, neurosphere growth and expression of mesenchymal subtype biomarkers correlated with uPAR abundance. uPAR also promoted glioblastoma cell survival in neurospheres. Constitutively-active EGF Receptor (EGFRvIII) promoted neurosphere growth; however, unlike uPAR, EGFRvIII did not induce the mesenchymal gene expression signature. Immunohistochemical analysis of human glioblastomas showed that uPAR is typically expressed by a small sub-population of the cancer cells; it is thus reasonable to conclude that this subpopulation of cells is responsible for the efects of PLAUR on patient survival. We propose that uPAR-expressing glioblastoma cells demonstrate a mesenchymal gene signature, an increased capacity for cell survival, and stem cell-like properties. -

Cómo Citar El Artículo Número Completo Más Información Del
Revista de Biología Tropical ISSN: 0034-7744 ISSN: 0034-7744 Universidad de Costa Rica Alpízar-Alpízar, Warner; Malespín-Bendaña, Wendy; Une, Clas; Ramírez-Mayorga, Vanessa Relevance of the plasminogen activation system in the pathogenesis and progression of gastric cancer Revista de Biología Tropical, vol. 66, núm. 1, 2018, pp. 28-47 Universidad de Costa Rica DOI: 10.15517/rbt.v66i1.29014 Disponible en: http://www.redalyc.org/articulo.oa?id=44955366003 Cómo citar el artículo Número completo Sistema de Información Científica Redalyc Más información del artículo Red de Revistas Científicas de América Latina y el Caribe, España y Portugal Página de la revista en redalyc.org Proyecto académico sin fines de lucro, desarrollado bajo la iniciativa de acceso abierto Relevance of the plasminogen activation system in the pathogenesis and progression of gastric cancer Warner Alpízar-Alpízar1,2*, Wendy Malespín-Bendaña3, Clas Une3 & Vanessa Ramírez-Mayorga3,4 1. Centro de Investigación en Estructuras Microscópicas (CIEMic), Universidad de Costa Rica, San José, Costa Rica; [email protected] 2. Departamento de Bioquímica, Escuela de Medicina, Universidad de Costa Rica, San José, Costa Rica. 3. Instituto de Investigaciones en Salud (INISA), Universidad de Costa Rica, San José, Costa Rica; [email protected], [email protected], [email protected] 4. Sección de Nutrición Pública, Escuela de Nutrición, Universidad de Costa Rica, San José, Costa Rica. * Correspondence Received 14-VII-2017. Corrected 18-X-2017. Accepted 16-XI-2017. Abstract: Gastric cancer is ranked as the third death-causing cancer and one of the most incident malignancies worldwide. -

Receptor-Like 1 and -Like 2 Is an Endogenous Ligand for Formyl Peptide Through a Urokinase Receptor Epitope That Urokinase Induc
Urokinase Induces Basophil Chemotaxis through a Urokinase Receptor Epitope That Is an Endogenous Ligand for Formyl Peptide Receptor-Like 1 and -Like 2 This information is current as of May 10, 2019. Amato de Paulis, Nunzia Montuori, Nella Prevete, Isabella Fiorentino, Francesca Wanda Rossi, Valeria Visconte, Guido Rossi, Gianni Marone and Pia Ragno J Immunol 2004; 173:5739-5748; ; doi: 10.4049/jimmunol.173.9.5739 http://www.jimmunol.org/content/173/9/5739 Downloaded from References This article cites 65 articles, 33 of which you can access for free at: http://www.jimmunol.org/content/173/9/5739.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on May 10, 2019 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2004 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Urokinase Induces Basophil Chemotaxis through a Urokinase Receptor Epitope That Is an Endogenous Ligand for Formyl Peptide Receptor-Like 1 and -Like 21 Amato de Paulis,* Nunzia Montuori,† Nella Prevete,* Isabella Fiorentino,* Francesca Wanda Rossi,* Valeria Visconte,‡ Guido Rossi,‡ Gianni Marone,2* and Pia Ragno† Basophils circulate in the blood and are able to migrate into tissues at sites of inflammation. -

The Urokinase Receptor: a Multifunctional Receptor in Cancer Cell Biology
International Journal of Molecular Sciences Review The Urokinase Receptor: A Multifunctional Receptor in Cancer Cell Biology. Therapeutic Implications Anna Li Santi 1,†, Filomena Napolitano 2,†, Nunzia Montuori 2 and Pia Ragno 1,* 1 Department of Chemistry and Biology, University of Salerno, Fisciano, 84084 Salerno, Italy; [email protected] 2 Department of Translational Medical Sciences, “Federico II” University, 80135 Naples, Italy; fi[email protected] (F.N.); [email protected] (N.M.) * Correspondence: [email protected] † Equal contribution. Abstract: Proteolysis is a key event in several biological processes; proteolysis must be tightly con- trolled because its improper activation leads to dramatic consequences. Deregulation of proteolytic activity characterizes many pathological conditions, including cancer. The plasminogen activation (PA) system plays a key role in cancer; it includes the serine-protease urokinase-type plasminogen activator (uPA). uPA binds to a specific cellular receptor (uPAR), which concentrates proteolytic activity at the cell surface, thus supporting cell migration. However, a large body of evidence clearly showed uPAR involvement in the biology of cancer cell independently of the proteolytic activity of its ligand. In this review we will first describe this multifunctional molecule and then we will discuss how uPAR can sustain most of cancer hallmarks, which represent the biological capabilities acquired during the multistep cancer development. Finally, we will illustrate the main data available in the literature on uPAR as a cancer biomarker and a molecular target in anti-cancer therapy. Citation: Li Santi, A.; Napolitano, F.; Montuori, N.; Ragno, P. The Keywords: urokinase receptor; uPAR; cancer hallmarks Urokinase Receptor: A Multifunctional Receptor in Cancer Cell Biology. -

Folate Receptor Β Regulates Integrin Cd11b/CD18 Adhesion of a Macrophage Subset to Collagen
Folate Receptor β Regulates Integrin CD11b/CD18 Adhesion of a Macrophage Subset to Collagen This information is current as Christian Machacek, Verena Supper, Vladimir Leksa, Goran of September 24, 2021. Mitulovic, Andreas Spittler, Karel Drbal, Miloslav Suchanek, Anna Ohradanova-Repic and Hannes Stockinger J Immunol 2016; 197:2229-2238; Prepublished online 17 August 2016; doi: 10.4049/jimmunol.1501878 Downloaded from http://www.jimmunol.org/content/197/6/2229 Supplementary http://www.jimmunol.org/content/suppl/2016/08/17/jimmunol.150187 Material 8.DCSupplemental http://www.jimmunol.org/ References This article cites 49 articles, 23 of which you can access for free at: http://www.jimmunol.org/content/197/6/2229.full#ref-list-1 Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision by guest on September 24, 2021 • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2016 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Folate Receptor b Regulates Integrin CD11b/CD18 Adhesion of a Macrophage Subset to Collagen Christian Machacek,* Verena Supper,* Vladimir Leksa,*,† Goran Mitulovic,‡ Andreas Spittler,x Karel Drbal,{,1 Miloslav Suchanek,{ Anna Ohradanova-Repic,* and Hannes Stockinger* Folate, also known as vitamin B9, is necessary for essential cellular functions such as DNA synthesis, repair, and methylation. -

Treatmentupdate 223 Vol
TreatmentUpdate 223 Vol. 29, No. 5 · December 2017 Available online at www.catie.ca/en/treatmentupdate Contents G. Canakinumab’s anti-cancer potential 10 I INFLAMMATION AND HIV H. Issues to consider with canakinumab 12 A. Exploring HIV and inflammation 2 I. Canakinumab in HIV reduces B. Inflammation in arteries vs. inflammation 13 lymph nodes 5 J. The potential of Ixolaris 15 C. Pitavastatin found to reduce levels of bad cholesterol and inflammation 6 K. Clinical trials in Canada to explore reducing inflammation in HIV 16 D. Inflammation-related illness among HIV-positive people 7 L. suPAR—an early warning signal 16 E. Why is there renewed interest in the antibody canakinumab? 8 F. Canakinumab—effect on reducing inflammation and heart attacks in the Cantos study 9 produced by 555 Richmond Street West, Suite 505 Box 1104 Toronto, Ontario M5V 3B1 Canada phone: 416.203.7122 toll-free: 1.800.263.1638 fax: 416.203.8284 www.catie.ca charitable registration number: 13225 8740 RR Page 2 TreatmentUpdate 223 — Vol. 29 No. 5 __________________________________________________________ I INFLAMMATION AND HIV Why is persistent immune activation and inflammation important? A. Exploring HIV and inflammation The activation of the immune system and inflammation are important responses used by Chronic HIV infection is associated with relatively the immune system to help control infections and high levels of inflammation and a growing body tumours. However, researchers are concerned that of evidence suggests that inflammation may prolonged immune activation and inflammation increase the risk for a range of health problems. could slowly degrade vital organ-systems in the body. -

Utility of the Plasma Level of Supar in Monitoring Risk of Mortality During TB Treatment
Utility of the Plasma Level of suPAR in Monitoring Risk of Mortality during TB Treatment Paulo Rabna1, Andreas Andersen1, Christian Wejse1,2,3, Ines Oliveira4, Victor Francisco Gomes1, Maya Bonde Haaland3, Peter Aaby1,5, Jesper Eugen-Olsen1* 1 Bandim Health Project, Indepth Network, Bissau, Guinea-Bissau, 2 Department of Infectious Diseases, Aarhus University Hospital, Aarhus, Denmark, 3 Center of Global Health, School of Public Health, Aarhus University, Aarhus, Denmark, 4 Clinical Research Centre, Copenhagen University Hospital Hvidovre, Hvidovre, Denmark, 5 Bandim Health Project, Statens Serum Institute, Copenhagen, Denmark Abstract Objective: To investigate whether changes in the plasma level of soluble urokinase plasminogen activator receptor (suPAR) can be used to monitor tuberculosis (TB) treatment efficacy. Design: This prospective cohort study included 278 patients diagnosed with active pulmonary TB and followed throughout the 8-month treatment period. Results: Mortality during treatment was higher in the highest inclusion quartile of suPAR (23%) compared to the lowest three quartiles (7%), the risk ratio being 3.1 (95% CI 1.65–6.07). No association between early smear conversion and subsequent mortality or inclusion suPAR was observed. After 1 and 2 months of treatment, an increase in suPAR compared to at diagnosis was associated with a Mortality Rate Ratio (MRR) of 4.5 (95%CI: 1.45–14.1) and 2.1 (95%CI 0.62–6.82), respectively, for the remaining treatment period. Conclusions: The present study confirmed that elevated suPAR level at time of initiation of TB treatment is associated with increased risk of mortality. Furthermore, increased suPAR levels after one month of treatment was associated with increased risk of mortality during the remaining 7-month treatment period. -

1 Instructions for Use Suparnostic® Turbilatex Reagents
1 Instructions for Use suPARnostic® TurbiLatex Reagents INTERPRETATION OF suPAR suPAR level Clinical decision soluble urokinase Plasminogen Activator Receptor <3 ng/mL Supports the decision of discharge. T005 The underlying health condition is good, and the prognosis for surviving is very high. suPARnostic® and the ViroGates A/S ViroGates logo are Banevaenget 13 3-6 ng/mL Some disease activity or co-morbidity is present. registered trademarks of Birkeroed 3460, Some readmissions and an elevated mortality are ViroGates A/S Denmark. Denmark expected after 6 months follow-up. ©2008 ViroGates. Tel: +45 2113 1336 www.virogates.com All rights reserved. >6 ng/mL Attention is needed. High risk of mortality. Further tests are needed to find the presence of severe disease. Table 1: Simplified suPAR clinical decision scheme (data from Danish hospital)3. Interpretation of results must be made considering the patient´s clinical history and results of other diagnostic tests. The decision to discharge This product is protected by one or more US, European, and/or foreign can, therefore, not be performed solely based on the patients suPAR patents. value. The T005 product is validated on the automated Roche Cobas c701/2 PRINCIPLES OF ASSAY PROCEDURE ® instrument (Cobas is a trademark of Roche), and this instruction for use The suPARnostic® TurbiLatex test is a turbidimetric immunoassay is dedicated for the Roche cobas c701/c702 biochemistry analyser. that quantitatively determines suPAR in human plasma samples. The first stage of testing is an incubation of human origin specimen (EDTA- Refer to the webpage http://www.virogates.com for instructions for use or Heparin plasma) with the R1 reagent. -
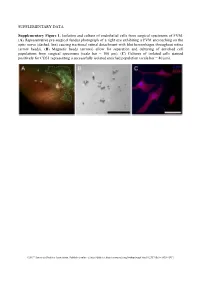
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Of Body Inflammation for Maxillofacial Surgery Purpose-To Make
applied sciences Article Index of Body Inflammation for Maxillofacial Surgery Purpose-to Make the Soluble Urokinase-Type Plasminogen Activator Receptor Serum Level Independent on Patient Age Marcin Kozakiewicz 1,*, Magdalena Trzci´nska-Kubik 1 and Rafał Nikodem Wlazeł 2 1 Department of Maxillofacial Surgery, Medical University of Lodz, 113 Zeromskiego˙ Str., 90-549 Lodz, Poland; [email protected] 2 Department of Laboratory Diagnostics and Clinical Biochemistry, Medical University of Lodz, 251 Pomorska Str., 92-213 Lodz, Poland; [email protected] * Correspondence: [email protected]; Tel.: +48-42-6393068 Featured Application: Inflammation is still a threat to patients of all age groups. Monitoring the infection is essential to control it. The soluble urokinase-type plasminogen activator receptor (suPAR) is a very sensitive marker. Maxillofacial surgery is one of the fields of medicine widely fighting against infections. The concentration of suPAR holds an independent information for risk stratification of morbidity and mortality in various acute and chronic diseases. The use of an ultra-sensitive measure of the development of inflammation and at the same time independent on the patient’s age would be clinically very beneficial. Abstract: Background: The serum suPAR level is affected in humans by it increases with age. Therefore it makes difficult interpretation and any comparison of age varied groups. The aim of this Citation: Kozakiewicz, M.; study is to find simple way to age independent presentation of suPAR serum level for maxillofacial Trzci´nska-Kubik,M.; Wlazeł, R.N. surgery purpose. Methods: In generally healthy patients from 15 to 59 y.o. suPAR level was tested Index of Body Inflammation for in serum before orthognathic or minor traumatologic procedures. -

Conformation Based Reagents for the Detection of Disease-Associated Prion Protein
CONFORMATION BASED REAGENTS FOR THE DETECTION OF DISEASE-ASSOCIATED PRION PROTEIN By KRISTEN-LOUISE HATCHER Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy Dissertation Advisor: Shu G. Chen, Ph.D. Department of Pathology CASE WESTERN RESERVE UNIVERSITY May, 2009 CASE WESTERN RESERVE UNIVERSITY SCHOOL OF GRADUATE STUDIES We hereby approve the thesis/dissertation of _____________________________________________________ candidate for the ______________________degree *. (signed)_______________________________________________ (chair of the committee) ________________________________________________ ________________________________________________ ________________________________________________ ________________________________________________ ________________________________________________ (date) _______________________ *We also certify that written approval has been obtained for any proprietary material contained therein. To Grandad Dee, for teaching me to love science, and Grandad Hatcher, for always believing I could do it Table of Contents List of Tables 3 List of Figures 4 Acknowledgements 5 List of Abbreviations 6 Abstract 7 Chapter 1: Introduction 1.1 The Prion Protein 9 1.1.1 The Codon 129 Polymorphism 11 1.2 The Pathogenic Prion Protein 12 1.2.1 Prion Conversion 12 1.2.2 PK Sensitive PrPSc 14 1.2.3 Prion Strains 15 1.3 Creutzfeldt-Jakob Disease 16 1.3.1 Sporadic Creutzfeldt-Jakob Disease 16 1.3.2 Familial Creutzfeldt-Jakob Disease 19 1.3.3 Acquired Creutzfeldt-Jakob Disease 22 1.3.3.1 Iatrogenic -

Predicts Microalbuminuria in Patients at Risk for Type 2 Diabetes Mellitus
www.nature.com/scientificreports OPEN Soluble urokinase receptor (suPAR) predicts microalbuminuria in patients at risk for type 2 Received: 09 August 2016 Accepted: 08 December 2016 diabetes mellitus Published: 16 January 2017 Martina Guthoff1,2,3,*, Robert Wagner1,2,3,*, Elko Randrianarisoa1,2,3, Erifili Hatziagelaki4, Andreas Peter1,2,3, Hans-Ulrich Häring1,2,3, Andreas Fritsche1,2,3 & Nils Heyne1,2,3 Early identification of patients at risk of developing diabetic nephropathy is essential. Elevated serum concentrations of soluble urokinase receptor (suPAR) associate with diabetes mellitus and predict onset and loss of renal function in chronic kidney disease. We hypothesize, that suPAR may be an early risk indicator for diabetic nephropathy, preceding microalbuminuria. The relationship of baseline suPAR and incident microalbuminuria was assessed in a prospective long-term cohort of subjects at increased risk for type 2 diabetes (TULIP, n = 258). Association with albuminuria at later stages of disease was studied in a cross-sectional cohort with manifest type 2 diabetes (ICEPHA, n = 266). A higher baseline suPAR was associated with an increased risk of new-onset microalbuminuria in subjects at risk for type 2 diabetes (hazard ratio 5.3 (95% CI 1.1–25.2, p = 0.03) for the highest vs. lowest suPAR quartile). The proportion of subjects with prediabetes at the end of observation was higher in subjects with new-onset microalbuminuria. suPAR consistently correlated with albuminuria in a separate cohort with manifest type 2 diabetes. Elevated baseline suPAR concentrations independently associate with new-onset microalbuminuria in subjects at increased risk of developing type 2 diabetes.