GRAS Notice 668, Sodium Formate
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Food Allergen Management in Foodservice – a Best Practice Guideline
national allergy strategy Food Allergen Management in Foodservice A BEST PRACTICE GUIDELINE Developed by Statewide Foodservices Qld. Health This work is licensed under a Creative Commons Attribution Non-Commercial V4.0 International licence. To view a copy of this Block 7 Level 7 Royal Brisbane & Women’s Hospital, licence, visit https://creativecommons.org/licenses/by-nc/4.0/deed.en Butterfield St., Herston Qld 4029 You are free to copy, communicate and adapt the work for non- Ph. (07) 3646 2288 commercial purposes, as long as you attribute the State of Queensland (Queensland Health) and comply with the licence terms. [email protected] For copyright permissions beyond the scope of this licence contact: Intellectual Property Officer, Queensland Health, © State of Queensland (Queensland Health) 2018 email [email protected], phone (07) 3708 5069. PAGE 1 FOOD ALLERGEN MANAGEMENT IN FOODSERVICE – A BEST PRACTICE GUIDELINE Background Standards Added Sulphites in concentrations of 10 mg/kg or more In managing food allergies in health care foodservices there Cereals containing gluten and their products, namely, wheat, rye, barley, oats and spelt and their hybridised strains are overarching practices that are required as per the Food Crustacea and their products Standards Code (FSANZ) that will inform and support the Egg and egg products process of identifying, assessing, managing and auditing Fish and fish products, except for isinglass derived from swim bladders and the risk of food allergies in the food service. used as a clarifying agent in beer and wine Milk and milk products Peanuts and peanut products These include – Food Standards Australia & New Zealand (Chapter 1 – Food Allergen Labelling - A food allergy occurs when a person’s immune system Sesame seeds and sesame seed products reacts to allergens that are harmless to other people. -

Trimethylacetic Formic Anhydride Precipitation from Ethanol and Diethyl Ether, M.P
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/16408 Please be advised that this information was generated on 2021-09-24 and may be subject to change. 460 Edward J. Vlietsira et al. / Trimethylacetic formic anhydride precipitation from ethanol and diethyl ether, m.p. 240°C (dec.); Acknowledgements [a]5 5 —140° (c 1.0, water). MS: M* 700. We thank Mr. P. Kranenburg for valuable technical as N-(6,14-endo-Etheno-7,8-dihydromorphine-7ai-carbonyl)-L- sistance and Messrs. J. A. de Groot, L. J. M. Helvensteijn -phenylalanyl-L-leucinol (14) and E. F. Lameijer for carrying out preliminary experi The hydrochloride of 12 (1.63 g, 2.4 mmol) was converted into the ments. We are grateful to the Management of Diosynth base and dissolved in 30 ml of anhydrous 2-propanol. To this B. V., Apeldoorn, The Netherlands, for gifts of chemicals. solution, 1.5 g (15 mmol) of anhydrous calcium chloride and We thank the U.S.A. Committee on Problems of Drug 1.14 g (30 mmol) of sodium tetrahydroborate were added. The Dependence and Dr. A. E. Jacobson, Biological Coordi conversion was complete (TLC) after 6 days at 35°C. Water nator, for the results of the pharmacological studies. We (50 ml) was then added and the mixture acidified with 2 N hydro gen chloride to pH 2-3. Extraction with a mixture of chloroform are indebted to Dr. -

Outline What Are What Are Processing a Processing Aids?
The Issue of Undeclared Ingredients in Halal ﻗﺿﻳﺔ اﻟﻣﻛوﻧﺎت ﻏﻳر اﻟﻣﻌﻠﻧﺔ ﻓﻲ اﻷﻏذﻳﺔ ا ﻟ ﻣ ﺟ ﻬ زة اﻟﺣﻼﻝ؟ Food Processing اﻟﺗرﻛﻳز ﻋﻠﻰ ﻣﻌﺎﻟﺟﺎت اﻟﺗﺻﻧﻳﻌﻳﺔ A Focus on Processing Aids أ.د. ﻣﻳﺎن ﻣﺣﻣد ﻧدﻳم رﻳﺎض .Mian N. Riaz, Ph.D ﻣدﻳر أﺑﺣﺎث اﻟﺑروﺗﻳن، ﺟﺎﻣﻌﺔ ﺗﻛﺳﺎس أﻳﻪ أﻧد أم، ﺗﺎﻣو، اﻟوﻻﻳﺎت Food Protein R&D Center; Texas A&M University اﻟﻣﺗﺣدة اﻷﻣرﻳﻛﻳﺔ College Station, Texas USA ورشة الحﻻل اﻷولى First Halal Workshop 11 مايو May, 2014 2014 11 معھد الكويت لﻷبحاث العلمية Kuwait Institute for Scientific دولة الكويت Research, State of Kuwait Outline What are Processing AAids?ids? Processing aids and Hidden Ingredients Processing aids and Hidden Ingredients What are processing aids?aids? Processing aids are substances that are approved by both the Sources of processing aidsaids Food and Drug Administration Why use processing aids and hidden (FDA) and the U.S. Department ingredients? of Agriculture (USDA) Are All hidden ingredients are Halal? They are used in the production Examples of hidden ingredients of a variety of foods – meat, Control of Halal food processing aid poultry, produce, etc., and are and hidden ingredients not present in any significant amount in the finished product What are Processing Aids? What are Processing aids? Processing Aids and Hidden Ingredients Processing Aids and Hidden Ingredients The use of food Both the FDA and USDA recognize three situations in which a processing aid has manufacturing substance is deemed to be a processing aid: become more prominent in recent 11.. When substances are added to a food during processing years, due to the but subsequently removed before the food reaches its finished increased production form (example activated charcoals which filter out impurities) of prepared, processed, and convenience foods What are Processing aids? What are Processing aids? Processing Aids and Hidden Ingredients Processing Aids and Hidden Ingredients 2. -

SODIUM FORMATE, Hydrate
CXSO045 - SODIUM FORMATE, hydrate SODIUM FORMATE, hydrate Safety Data Sheet CXSO045 Date of issue: 04/04/2017 Version: 1.0 SECTION 1: Identification 1.1. Product identifier Product name : SODIUM FORMATE, hydrate Product code : CXSO045 Product form : Substance Physical state : Solid Formula : CHNaO2 Synonyms : FORMIC ACID, ZINC SALT, DIHYDRATE DIFORMATOZINC DIHYDRATE ZINC DIFORMATE DIHYDRATE Chemical family : METAL COMPOUND 1.2. Recommended use of the chemical and restrictions on use Recommended use : Chemical intermediate For research and industrial use only 1.3. Details of the supplier of the safety data sheet GELEST, INC. 11 East Steel Road Morrisville, PA 19067 USA T 215-547-1015 - F 215-547-2484 - (M-F): 8:00 AM - 5:30 PM EST [email protected] - www.gelest.com 1.4. Emergency telephone number Emergency number : CHEMTREC: 1-800-424-9300 (USA); +1 703-527-3887 (International) SECTION 2: Hazard(s) identification 2.1. Classification of the substance or mixture GHS-US classification Not classified 2.2. Label elements GHS-US labeling No labeling applicable 2.3. Hazards not otherwise classified (HNOC) No additional information available 2.4. Unknown acute toxicity (GHS US) No data available SECTION 3: Composition/Information on ingredients 3.1. Substances Substance type : Mono-constituent Name : SODIUM FORMATE, hydrate CAS No : 141-53-7 Name Product identifier % GHS-US classification Sodium formate (CAS No) 141-53-7 95 - 100 Not classified Full text of hazard classes and H-statements : see section 16 3.2. Mixtures Not applicable 4.1. Description of first aid measures First-aid measures general : Remove contaminated clothing and shoes. -

The Food Wars Thesis
.CHAPTER 1 THE FOOD WARS THESIS 'Ifyouknow before you look, you cannot see for knowing.' Sir Terry Frost RA (British artist 1915-2003) CORE ARGUMENTS Different visions for the future of food are shaping the potential for how food will be produced and marketed. Inevitably, there will be policy choices - for the state, the corporate sector and civil society. Human and environ mental health needs to be at the heart of these choices. Three broad conceptual frameworks or 'paradigms' pro pose the way forward for food policy, the food economy and health itself. All make claims to raise production and to deliver health benefits through food. The challenge for policy-makers is how to sift through the evidence and to give a fair hearing to a range of choices. This process is sometimes difficult because the relationship betweenevid ence and policy is not what it seems. The world of food is on the cusp of a far-reaching transition. INTRODUCTION The world is producingmore food than ever to feed more mouths than ever,' For the better off there are more food and beverage product choices than it is possible to imagine - globally 25,000 products in the average supermarket and more than 20,000 new packaged foods and beverages in 2002 alone.? Yet for many people there is a general feeling of unease and mistrust about the 12 FOOD WARS future of our food supply. Food and problems associated with producing and consuming food generate political and policy crises and are regular fodder for media coverage. In addition, along with the food production successes of the past 40 years in reducing famine, hunger continues hand in hand with excess. -

Australian Pilot Survey of Gm Food Labelling of Corn and Soy Food Products
AUSTRALIAN PILOT SURVEY OF GM FOOD LABELLING OF CORN AND SOY FOOD PRODUCTS by The TAG Working Group on GM Food Labelling June 2003 Table of Contents 1. SUMMARY............................................................................... 3 2. INTRODUCTION...................................................................... 4 2.1 Regulation of Food Produced Using Gene Technology.....................4 2.2 Australian Pilot Survey for GM Food Labelling ..................................5 2.3 Product Selection for the Survey .......................................................5 3. METHODS ............................................................................... 7 3.1 Sampling Programme........................................................................7 3.2 Testing Programme ...........................................................................7 3.3 Document Survey Methodology.........................................................8 4. RESULTS............................................................................... 10 4.1 Test Results.....................................................................................10 4.2 Documentation Survey Results .......................................................12 5. CONCLUSIONS..................................................................... 15 5.1 PCR Results ....................................................................................15 5.2 Documentation Survey Conclusions................................................19 6. ACKNOWLEDGEMENT ....................................................... -

Other Ingredients"
This is a work product of the National Organic Standards Board (NOSB). It does not represent official National Organic Program (NOP) positions or policy. National Organic Standards Board Handling Subcommittee Proposal Auxiliary/"Other Ingredients" January 29, 2013 Introduction On Nov. 23, 2011, National Organic Program (NOP) Deputy Administrator Miles McEvoy sent a Memorandum to the National Organic Standards Board (NOSB) requesting clarification of “other ingredients” contained within handling materials on the National List of Allowed and Prohibited substance used in processed organic products. Since OFPA requires that each non-agricultural ingredient be specifically listed, and because the National List does not specifically list “other ingredients” commonly found in formulated products, the NOP identified the need for clarity and requested that the NOSB develop a policy that specifies whether these “other ingredients” are allowed. In the memo to NOSB, NOP requested the following: The NOP is requesting that the NOSB develop a policy on “other ingredients” in § 205.605 substances that is comparable to the comprehensive policy for crop and livestock materials. From this point forward, NOP is requesting that NOSB consider the presence of any “other ingredients” as part of its processes. As substances on the National List come up for sunset review, or as new petitions are considered, NOP requests that NOSB clarify whether any restrictions are warranted for “other ingredients” in § 205.605 substances. Any third-party technical report that NOP provides will include information on any “other ingredients” commonly found in the substance under review. NOP is requesting that NOSB specify any allowed “other ingredients” in the background section of its recommendations for substances recommended for listing on § 205.605, so that these allowances are clear to the organic trade, certifying agents, and NOP. -

Chapter 8: Food Additives
8 Food Additives Tanya Louise Ditschun and Carl K. Winter CONTENTS Introduction Food Additive Functionality Food Additive Regulations Generally Recognized as Safe (GRAS) The Delaney Clause Unintentional Additives Assessment of Food Safety` Specific Food Additives Under Scrutiny Saccharin Aspartame Hydrolysis Products of Aspartame Aspartic Acid Phenylalanine Methanol Diketopiperazine Marketing of Aspartame Erythrosine (FD & C Red #3) Olestra Anectodal Reports of Health Effects Due to Olestra Effects of Olestra on Nutrient Absorption Vitamin A Vitamin E Vitamin D Vitamin K Triglycerides Dietary Phytochemicals References © 2000 by CRC Press LLC Introduction Food additives have been used for centuries in food processing practices such as smoking and salting meat. Prior to the advent of refrigeration, food grown in the summer had to be preserved for the winter; salt, sugar, and vinegar were commonly used preservatives. The pursuits of explorers such as Marco Polo were often for food additives. Additives serve many roles and common uses include maintaining product consistency and pal- atability, providing leavening or control pH, enhancing flavor, and impart- ing color. A food additive can be defined in many ways. The Codex Alimentarius Commission, which develops international regulatory guidelines for food additives, provides the following definition of a food additive: Any substance not normally consumed as a food by itself, and not normal- ly used as a typical ingredient of the food, whether or not it has nutritive value, the intentional addition of which to food for a technological (includ- ing organoleptic) purpose in the manufacture, processing, preparation, treatment, packing, packaging, transport or holding of such food results, or may reasonably be expected to result, directly or indirectly, in it or its by-products becoming a component of or otherwise affecting the charac- teristics of such food. -
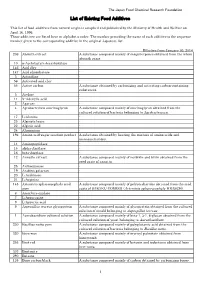
List of Existing Food Additives
The Japan Food Chemical Research Foundation List of Existing Food Additives This list of food additives from natural origin is complied and published by the Ministry of Health and Welfare on April 16, 1996. These additives are listed here in alphabetic order. The number preceding the name of each additive is the sequence number given to the corresponding additive in the original Japanese list. Effective from January 30, 2014 236 Absinth extract A substance composed mainly of sesquiterpenes obtained from the whole absinth grass. 10 α-Acetolactate decarboxylase - 146 Acid clay - 147 Acid phosphatase - 3 Actinidine - 56 Activated acid clay - 55 Active carbon A substance obtained by carbonizing and activating carbon-containing substances. 5 Acylase - 11 5'-Adenylic acid - 2 Agarase - 4 Agrobacterium succinoglycan A substance composed mainly of succinoglycan obtained from the cultured solution of bacteria belonging to Agrobacteriurn. 17 L-Alanine - 23 Alginate lyase - 22 Alginic acid - 24 Aluminium - 196 Amino acid-sugar reaction product A substance obtained by heating the mixture of amino acids and monosaccharides. 14 Aminopeptidase - 15 alpha-Amylase - 16 beta-Amylase - 12 Annatto extract A substance composed mainly of norbixin and bixin obtained from the seed coats of annatto. 25 Anthocyanase - 19 Arabino galactan - 20 L-Arabinose - 21 L-Arginine - 145 Artemisia sphaerocephala seed A substance composed mainly of polysaccharides obtained from the seed gum coats of SABAKU-YOMOGI (Artemisia sphaerocephala KRASCH). 6 Ascorbate oxidase - 7 L-Asparagine - 8 L-Aspartic acid - 9 Aspergillus terreus glycoprotein A substance composed mainly of glycoprotein obtained from the cultured solution of mould belonging to Aspergillus terreus . 1 Aureobasidium cultured solution A substance composed mainly of beta-1, 3-1, 6-glucan obtained from the cultured solution of yeast belonging to Aureobasidium . -

The Activation of Surface Films of Lecithin by Amphipathic Molecules
Vol. 72 493 The Activation of Surface Films of Lecithin by Amphipathic Molecules BY R. M. C. DAWSON AND A. D. BANGHAM Agricultural Research Council Institute of Animcw Physiology,, Babraham, Cambridge (Received, 31 December 1958) In the preceding paper it was shown that a phos- methanol-chloroform (1:1, v/v) and the auspension pholipase prepared,from Penicllium notatum will filtered. The filtrates were pooled and evaporated to hydrolyse lecithin only after the, addition,, of a dryness in vacuo. The,lipid residue was then dissolved in minimum proportion of anionic amphipathic 50 ml. of ethanol and the ethanol removed in vacuo to complete dehydration. The,residue was next thoroughly molecules1 e.g. dicetylphosphoric acid, cardiolipin, extracted with 100 ml. of-diethyl, ether; insoluble matter sodium dodecyl sulphate (Bangham & Dawson, was removed by filtration and the filtrate evaporated to 1959), The onset of hydrolysis occurred, only when dryness. The residue was dissolved in 4 ml. of ether and the emulsion particles carried a minimum net 40 ml. of acetone was added;-after allowing the solution to negative charge, independent of the .species of stand for 1 hr. at 0°, the precipitate of phospholipids was anion added. recovered by centrifuging. The, acetone-damp precipitate The question arose whether the negative charge was extracted, first with,8 ml. of chloroform -methanol on the surface of the emulsion particle was ,directly (1:1, v/v), and then onc,e more with 4 ml. of the same concerned with the reaction between enzyme,and solvent, any insoluble material-beAg"remroved by centri- substrate at the or whether it fuging. -

Effects of Sodium Formate and Calcium Propionate Additives On
animals Article Effects of Sodium Formate and Calcium Propionate Additives on the Fermentation Quality and Microbial Community of Wet Brewers Grains after Short-Term Storage Jingyi Lv, Xinpeng Fang, Guanzhi Feng, Guangning Zhang , Chao Zhao, Yonggen Zhang and Yang Li * College of Animal Science and Technology, Northeast Agricultural University, Harbin 150030, China; [email protected] (J.L.); [email protected] (X.F.); [email protected] (G.F.); [email protected] (G.Z.); [email protected] (C.Z.); [email protected] (Y.Z.) * Correspondence: [email protected]; Tel./Fax: +86-0451-5519-0840 Received: 27 July 2020; Accepted: 7 September 2020; Published: 9 September 2020 Simple Summary: The objective of this study was to examine the effect of sodium formate and calcium propionate on the fermentation quality and microbial community of wet brewers grains (WBG) after short-term storage. Both additives improved the silage quality of WBG ensiled for 20 days to different extents. However, ensiled WBG treated with sodium formate had higher contents of dry matter, water-soluble carbohydrates, and neutral detergent fibers and better fermentation quality, rumen degradation, and microbial composition. The addition of sodium formate enhances the abundance of desirable Lactobacillus and reduces the abundance of undesirable microorganisms, including Clostridium. In summary, during short-term storage of high-moisture feed, sodium formate has a more beneficial preservation effect than an equivalent dose of calcium propionate. Abstract: The objective of this research was to examine the effect of sodium formate (SF) and calcium propionate (CAP) on the fermentation characteristics and microbial community of wet brewers grains (WBG) after short-term storage. -

FSSAI to Notify Processing Aids to Bring Clarity to Food Processing
FSSAI to notify processing aids to bring clarity to food processing Friday, 27 July, 2018, 08 : 00 AM [IST] Ashwani Maindola, New Delhi FSSAI, the country’s apex food regulator, has decided to list and notify processing aids, in a bid to bring in more clarity with respect to processing of food. In this regard, an appendix is being added to the Food Safety and Standards (Food Products Standards and Food Additives) Amendment Regulations, 2018. FSSAI has issued a notice seeking comments on the subject. The draft notification consists of definitions of the different processing aids used by food manufacturers. “There are enzymes that speed up the process, while there are cooling agents that aid in rapidly freezing the products. Also present in the notification are the names of these processing substances (under different categories) that can be used by the manufacturers with their maximum residue levels (MRLs). To top it all, it also mentions food products, wherein these substances can be added,” said Ashwin Bhadri, chief executive officer, Equinox Labs. Processing aids are the minute substances that play an important role in bringing out the finished product in its best version. These are approved by the Food and Drug Administration (FDA) and are used in a variety of foods, including meat and poultry, and are present in negligible amounts in the finished product. However, experts state that the existence of such processing aids in any finished product does not affect the appearance or taste, and most importantly, they have no major impact on public health, if consumed in acceptable daily intake (ADI).