FORM 10-Q Johnson & Johnson
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Mouth Rinses
Mouth Rinses We may recommend a mouth rinse to you for one or more of the following reasons: Periodontal Disease, Gingivitis, Halitosis (bad breath), sensitive teeth, decay or increased risk of decay, dry mouth, minor mouth sore or irritation. Red= RX only Blue= Available through your dentist only Green= Over the counter *Common side effects found with many mouth rinses include the possibility of staining. We still encourage you to use the rinse that treats your problem because none of the stains these rinses cause are permanent. With good oral hygiene on your part, using your bleaching trays and bleach as needed, you can keep the staining to a minimum. If there is any staining that you can’t prevent, it can always be easily removed by your hygienist at your next cleaning. If you have periodontal disease or Gingivitis, we may recommend an antimicrobial/antiseptic type rinse. These rinses kill germs, reduce plaque, redness, swelling and bleeding when used in conjunction with proper brushing and flossing. Some common examples of antiseptic rinses are: Peridex or Perioguard oral rinses are available by prescription only. Active ingredient is chlorhexidine gluconate .12%. These also contain alcohol 11.6%. Can cause staining of the teeth*, taste alteration and dry mouth. Periomed Oral rinse is available through your dentist only. Active ingredient is stannous fluoride .63%. It is alcohol free. Periomed provides antimicrobial activity for up to 8 hours. Also promotes enamel remineralization, and helps reduce sensitivity. Can cause staining* and dry mouth. Listerine Antiseptic: Active ingredient is alcohol 26.9% in the original gold Listerine, and 21.6% in the other flavors. -

Responding to Mylan's Inadequate Tender Offer
Responding To Mylan’s Inadequate Tender Offer: Perrigo’s Board Recommends That You Reject the Offer and Do Not Tender September 2015 Important Information Forward Looking Statements Certain statements in this presentation are forward-looking statements. These statements relate to future events or the Company’s future financial performance and involve known and unknown risks, uncertainties and other factors that may cause the actual results, levels of activity, performance or achievements of the Company or its industry to be materially different from those expressed or implied by any forward-looking statements. In some cases, forward-looking statements can be identified by terminology such as “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “potential” or other comparable terminology. The Company has based these forward-looking statements on its current expectations, assumptions, estimates and projections. While the Company believes these expectations, assumptions, estimates and projections are reasonable, such forward-looking statements are only predictions and involve known and unknown risks and uncertainties, many of which are beyond the Company’s control, including future actions that may be taken by Mylan in furtherance of its unsolicited offer. These and other important factors, including those discussed under “Risk Factors” in the Perrigo Company’s Form 10-K for the year ended June 27, 2015, as well as the Company’s subsequent filings with the Securities and Exchange -

1 Brief Report: the Virucidal Efficacy of Oral Rinse Components Against SARS-Cov-2 in Vitro Evelina Statkute1†, Anzelika Rubin
bioRxiv preprint doi: https://doi.org/10.1101/2020.11.13.381079; this version posted November 13, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. Brief Report: The Virucidal Efficacy of Oral Rinse Components Against SARS-CoV-2 In Vitro Evelina Statkute1†, Anzelika Rubina1†, Valerie B O’Donnell1, David W. Thomas2† Richard J. Stanton1† 1Systems Immunity University Research Institute, Division of Infection & Immunity, School of Medicine, Heath Park, Cardiff, CF14 4XN 2Advanced Therapies Group, School of Dentistry, Cardiff University, Heath Park, Cardiff CF14 4XY, UK †These authors contributed equally * Correspondence: [email protected], [email protected] Running title: Virucidal Activity of Mouthwashes Keywords: SARS-CoV2, mouthwash, lipid, envelope Disclosure: Venture Life Group plc provided information on mouthwash formulations employed in the study, but had no role in funding, planning, execution, analysis or writing of this study. A separate study funded to Cardiff University by Venture Life Group is assessing in vivo efficacy of CPC in patients with COVID19. The investigators declare no direct conflicts exist. 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.11.13.381079; this version posted November 13, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. -

Recommendations for the Primary Analysis of Continuous Endpoints in Longitudinal Clinical Trials
02-DIJ42(4) 2348.qxd 6/9/08 1:46 PM Page 303 STATISTICS 303 Recommendations for the Primary Analysis of Continuous Endpoints in Longitudinal Clinical Trials Craig H. Mallinckrodt, PhD Research Advisor, Lilly This position paper summarizes relevant theo- simple methods in favor of joint analysis of Research Laboratories, Eli ry and current practice regarding the analysis data from all time points based on a multivari- Lilly and Company, of longitudinal clinical trials intended to sup- ate model (eg, of a mixed-effects type). One Indianapolis, Indiana port regulatory approval of medicinal prod- such newer method, a likelihood-based mixed- Peter W. Lane, MA, CStat ucts, and it reviews published research regard- effects model repeated measures (MMRM) ap- Director of Consultancy and Training, Research Statistics ing methods for handling missing data. It is proach, has received considerable attention in Unit, GlaxoSmithKline, one strand of the PhRMA initiative to improve the clinical trials literature. We discuss specif- Harlow, United Kingdom efficiency of late-stage clinical research and ic concerns raised by regulatory agencies with Dan Schnell, PhD gives recommendations from a cross-industry regard to MMRM and review published evi- Section Head, Pharmaceutical Statistics, team. We concentrate specifically on continu- dence comparing LOCF and MMRM in terms Procter & Gamble ous response measures analyzed using a linear of validity, bias, power, and type I error. Our Pharmaceuticals, model, when the goal is to estimate and test main conclusion is that the mixed model ap- Mason, Ohio treatment differences at a given time point. proach is more efficient and reliable as a Yahong Peng, PhD Traditionally, the primary analysis of such tri- method of primary analysis, and should be Senior Biometrician, Clinical Biostatistics, Merck als handled missing data by simple imputation preferred to the inherently biased and statisti- Research Lab, Upper using the last, or baseline, observation carried cally invalid simple imputation approaches. -

Pfizer and Mylan Will Own 57% and 43% of the New Company (“Newco”), Respectively
DEAL PROFILE PFIZER | MYLAN VALUES 57% 43% $12.0bn PFIZER SHARE OF NEWCO MYLAN SHARE OF NEWCO NEW DEBT RAISED Pfizer Inc. (NYSE:PFE) Pfizer Inc. (“Pfizer”) is a pharmaceutical company that develops, manufactures, and sells products in internal medicine, vaccines, oncology, inflammation & immunology, and rare diseases. Upjohn, a subsidiary of Pfizer, is composed of Pfizer’s oral solid generics and off-patent drugs franchise, such as Viagra, Lipitor, and Xanax. Upjohn has a strong focus in emerging markets, and is headquartered in Shanghai, China. TEV: $265.4bn LTM EBITDA: $22.5bn LTM Revenue: $55.7bn Mylan N.V. (NASDAQ:MYL) Mylan N.V. (“Mylan”) is a pharmaceutical company that develops, manufactures, and commercializes generic, branded-generic, brand-name, and over-the-counter products. The company focuses in the following therapeutic areas: cardiovascular, CNS and anesthesia, dermatology, diabetes and metabolism, gastroenterology, immunology, infectious disease, oncology, respiratory and allergy, and women’s health. Mylan was founded in 1961 and is headquartered in Canonsburg, PA. TEV: $23.9bn LTM EBITDA: $3.5bn LTM Revenue: $11.3bn Bourne Partners provides strategic and financial advisory services to BOURNE clients throughout the business evolution life cycle. In order to provide PARTNERS the highest level of service, we routinely analyze relevant industry MARKET trends and transactions. These materials are available to our clients and partners and provide detailed insight into the pharma, pharma services, RESEARCH OTC, consumer health, and biotechnology sectors. On July 29, 2019, Pfizer announced the spin-off of its off-patent drug unit, Upjohn, in order to merge the unit with generic drug manufacturer, Mylan. -

Mylan Signs Agreement with Gilead to Enhance Access to Tenofovir Alafenamide (TAF)- Based HIV Treatments in Developing Countries
December 1, 2014 Mylan Signs Agreement with Gilead to Enhance Access to Tenofovir Alafenamide (TAF)- based HIV Treatments in Developing Countries PITTSBURGH and HYDERABAD, India, Dec. 1, 2014 /PRNewswire/ -- Mylan Inc. (Nasdaq: MYL) today announced that its subsidiary Mylan Laboratories Limited has entered into an agreement with Gilead Sciences, Inc. under which Mylan has licensed the non-exclusive rights to manufacture and distribute Tenofovir Alafenamide (TAF) as both a single agent product and in combination with other drugs. Tenofovir Alafenamide (TAF) is an investigational antiretroviral drug for the treatment of HIV-1 infection. The license being granted to Mylan extends to 112 countries, which together account for more than 30 million people living with HIV, representing 84% of those infected globally.1 Mylan CEO Heather Bresch said, "Mylan's mission is to provide the world's 7 billion people access to high quality medicines and set new standards in health care. By working with partners like Gilead to help ensure access to innovative new products such as Tenofovir Alafenamide (TAF) in the countries hardest hit by this disease, we can help stem the tide of HIV/AIDS around the world." As part of the licensing agreement, on U.S. Food and Drug Administration (FDA) approval, Mylan will receive a technology transfer from Gilead, enabling the company to manufacture low-cost versions of Tenofovir Alafenamide (TAF), if approved as a single agent or in approved combinations containing Tenofovir Alafenamide (TAF) for developing markets. Phase III trials by Gilead Sciences met their primary objective supporting the potential for Tenofovir Alafenamide (TAF) to provide a new treatment option for individuals living with HIV. -

Personalized Medicine for Reconstruction of Critical-Size Bone
www.nature.com/npjregenmed ARTICLE OPEN Personalized medicine for reconstruction of critical-size bone defects – a translational approach with customizable vascularized bone tissue ✉ Annika Kengelbach-Weigand 1 , Carolina Thielen 1, Tobias Bäuerle2, Rebekka Götzl 1,5, Thomas Gerber3, Carolin Körner4, Justus P. Beier1,5, Raymund E. Horch 1 and Anja M. Boos1,5 Tissue engineering principles allow the generation of functional tissues for biomedical applications. Reconstruction of large-scale bone defects with tissue-engineered bone has still not entered the clinical routine. In the present study, a bone substitute in combination with mesenchymal stem cells (MSC) and endothelial progenitor cells (EPC) with or without growth factors BMP-2 and VEGF-A was prevascularized by an arteriovenous (AV) loop and transplanted into a critical-size tibia defect in the sheep model. With 3D imaging and immunohistochemistry, we could show that this approach is a feasible and simple alternative to the current clinical therapeutic option. This study serves as proof of concept for using large-scale transplantable, vascularized, and customizable bone, generated in a living organism for the reconstruction of load-bearing bone defects, individually tailored to the patient’s needs. With this approach in personalized medicine for the reconstruction of critical-size bone defects, regeneration of parts of the human body will become possible in the near future. npj Regenerative Medicine (2021) 6:49 ; https://doi.org/10.1038/s41536-021-00158-8 1234567890():,; INTRODUCTION vascular networks consisting of endothelial cells can be created Therapeutic options for bone defects that cannot heal sponta- directly within tissue replacement materials7. Vascularization may neously, the so-called critical-size bone defects, are still limited be further supported by the addition of endothelial cells and and often associated with a great social burden. -

Endo Pharmaceuticals Inc. V. Actavis
United States Court of Appeals for the Federal Circuit ______________________ ENDO PHARMACEUTICALS INC., MALLINCKRODT LLC, Plaintiffs-Appellees v. ACTAVIS LLC, FKA ACTAVIS INC., ACTAVIS SOUTH ATLANTIC LLC, TEVA PHARMACEUTICALS USA, INC., Defendants-Appellants ______________________ 2018-1054 ______________________ Appeal from the United States District Court for the District of Delaware in No. 1:14-cv-01381-RGA, Judge Richard G. Andrews. ______________________ Decided: May 3, 2019 ______________________ MARTIN JAY BLACK, Dechert LLP, Philadelphia, PA, ar- gued for plaintiffs-appellees. Also represented by SHARON K. GAGLIARDI; BLAKE GREENE, Austin, TX; JONATHAN LOEB, Mountain View, CA; ROBERT RHOAD, Princeton, NJ. JEFFREY J. TONEY, Kasowitz, Benson, Torres & Fried- man LLP, Atlanta, GA, for plaintiff-appellee Mallinckrodt LLC. Also represented by RODNEY R. MILLER, PAUL GUNTER WILLIAMS. 2 ENDO PHARM. INC. v. ACTAVIS LLC JOHN C. O'QUINN, Kirkland & Ellis LLP, Washington, DC, argued for defendants-appellants. Also represented by WILLIAM H. BURGESS; CHARLES A. WEISS, ERIC H. YECIES, Holland & Knight, LLP, New York, NY. ______________________ Before WALLACH, CLEVENGER, and STOLL, Circuit Judges. Opinion for the court filed by Circuit Judge WALLACH. Dissenting opinion filed by Circuit Judge STOLL. WALLACH, Circuit Judge. Appellees Endo Pharmaceuticals Inc. (“Endo Pharma- ceuticals”) and Mallinckrodt LLC (“Mallinckrodt”) (collec- tively, “Endo”) sued Appellants Actavis LLC, Actavis South Atlantic LLC, and Teva Pharmaceuticals USA, Inc. (collec- tively, “Actavis”) in the U.S. District Court for the District of Delaware (“District Court”), alleging that two Abbrevi- ated New Drug Applications filed by Actavis infringed claims 1–6 (“the Asserted Claims”) of Mallinckrodt’s U.S. Patent No. 8,871,779 (“the ’779 patent”), which Endo Phar- maceuticals licenses. -
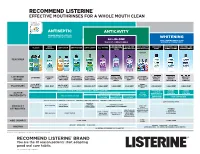
Listerine Floss Product Chart EN V9black
RECOMMEND LISTERINE® EFFECTIVE MOUTHRINSES FOR A WHOLE MOUTH CLEAN® 3 ANTISEPTIC ANTICAVITY 2X MORE HEALTHY SITES VS. JUST BRUSHING & FLOSSING WHITENING ALL-IN-ONE ONLY LEADING BRAND THAT THE MOST COMPLETE RINSE WHITENS & STRENGTHENS ALL-IN-ONE ZERO ALL-IN-ONE ANTI-CAVITY PEROXIDE WHITENS AND WHITENS AND CLASSIC ANTI-STAIN ANTI-TARTAR ANTI-CAVITY ALL-IN-ONE FOR SENSITIVE ALCOHOL TEETH ZERO ALCOHOL ZERO ALCOHOL FREE STRENGTHENS STRENGTHENS METERED DOSING FOR KIDS FEATURES ® LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE LISTERINE TOTAL CARE HEALTHY HEALTHY HEALTHY ULTRACLEAN ULTRACLEAN ULTRACLEAN TOTAL CARE SMART RINSE ZERO TOTAL CARE FOR SENSITIVE WHITE™ WHITE™ WHITE™ BRAND ANTI-STAIN ANTI-TARTAR ANTI-CAVITY TEETH ZERO FOR KIDS GENTLE RESTORING VIBRANT COOL MINT BERRY FRESHBURST ARCTIC MINT BUBBLE GUM FLAVOURS MILD MINT COOL MINT FRESHBURST CLEAN MINT CLEAN MINT MILD MINT CLEAN MINT CLEAN MINT CLEAN MINT ORIGINAL COOL CITRUS MINT EUCALYPTOL 0.091% W/V MENTHOL 0.042% W/V THYMOL 0.063% W/V SODIUM FLUORIDE SODIUM SODIUM 0.02% W/W FLUORIDE FLUORIDE ACTIVE SODIUM FLUORIDE 0.022% W/V SODIUM TETRAPOTASSIUM 0.022% W/V 0.02% W/W FLUORIDE PYROPHOSPHATE INGREDIENTS ZINC CHLORIDE 0.09% W/V ZINC CHLORIDE POTASSIUM ZINC CHLORIDE 0.022% W/V PENTASODIUM HYDROGEN PEROXIDE 0.09% W/V NITRATE 2.4% W/V 0.09% W/V TRIPHOSPHATE KILLS UP TO 99.9% OF GERMS IN YOUR MOUTH PREVENTS & REDUCES GINGIVITIS PREVENTS & REDUCES PLAQUE SAFELY WHITENS TEETH† PREVENTS CAVITIES MILD FLAVOUR HELPS KEEP PREVENTS CAVITIES PRODUCT TEETH WHITE STRENGTHENS STRENGTHENS TOOTH ENAMEL* 2X BETTER TOOTH ENAMEL CAVITY ATTRIBUTES PROTECTION VS. -

July 21, 2021
1 2nd Quarter 2021 Earnings Call July 21, 2021 Cautionary Note on Forward-looking Statements This presentation contains “forward-looking statements” as defined in the Private Securities Litigation Reform Act of 1995 regarding, among other things: future operating and financial performance, product development, market position and business strategy. The reader is cautioned not to rely on these forward-looking statements. These statements are based on current expectations of future events. If underlying assumptions prove inaccurate or known or unknown risks or uncertainties materialize, actual results could vary materially from the expectations and projections of Johnson & Johnson. Risks and uncertainties include, but are not limited to: risks related to the impact of the COVID-19 global pandemic, such as the scope and duration of the outbreak, government actions and restrictive measures implemented in response, material delays and cancellations of medical procedures, supply chain disruptions and other impacts to the business, or on the Company’s ability to execute business continuity plans, as a result of the COVID-19 pandemic; economic factors, such as interest rate and currency exchange rate fluctuations; competition, including technological advances, new products and patents attained by competitors; challenges inherent in new product research and development, including uncertainty of clinical success and obtaining regulatory approvals; uncertainty of commercial success for new and existing products; challenges to patents; the impact -

Amgen Inc. V. Sanofi, No
No. IN THE Supreme Court of the United States ———— AMGEN INC., AMGEN MANUFACTURING LIMITED, AND AMGEN USA, INC., Petitioners, v. SANOFI, AVENTISUB LLC, REGENERON PHARMACEUTICALS INC., AND SANOFI-AVENTIS U.S., LLC, Respondents. ———— On Petition for a Writ of Certiorari to the United States Court of Appeals for the Federal Circuit ———— PETITION FOR A WRIT OF CERTIORARI ———— STUART L. WATT JEFFREY A. LAMKEN WENDY A. WHITEFORD Counsel of Record ERICA S. OLSON MICHAEL G. PATTILLO, JR. EMILY C. JOHNSON SARAH J. NEWMAN AMGEN INC. MOLOLAMKEN LLP One Amgen Center Drive The Watergate, Suite 660 Thousand Oaks, CA 91320 600 New Hampshire Ave., NW (805) 447-1000 Washington, D.C. 20037 (202) 556-2000 [email protected] Counsel for Petitioners :,/621(3(635,17,1*&2,1&± ±:$6+,1*721'& QUESTION PRESENTED The 1952 Patent Act requires patents to “contain a written description of the invention, and of the manner and process of making and using it.” 35 U.S.C. §112(a). The “written description” must be “in such full, clear, concise, and exact terms as to enable any person skilled in the art to which it pertains, or with which it is most nearly connected, to make and use the same.” Ibid. “The object of the statute is to require the patentee to describe his invention so that others may construct and use it after the expiration of the patent.” Schriber-Schroth Co. v. Cleveland Tr. Co., 305 U.S. 47, 57 (1938). The Federal Circuit has construed §112(a) as impos- ing separate “written description” and “enablement” re- quirements subject to different standards. -

Merck & Co., Inc
As filed with the Securities and Exchange Commission on February 25, 2021 UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D. C. 20549 _________________________________ FORM 10-K (MARK ONE) ☒ Annual Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 For the Fiscal Year Ended December 31, 2020 OR ☐ Transition Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 For the transition period from to Commission File No. 1-6571 _________________________________ Merck & Co., Inc. 2000 Galloping Hill Road Kenilworth New Jersey 07033 (908) 740-4000 New Jersey 22-1918501 (State or other jurisdiction of incorporation) (I.R.S Employer Identification No.) Securities Registered pursuant to Section 12(b) of the Act: Title of Each Class Trading Symbol(s) Name of Each Exchange on which Registered Common Stock ($0.50 par value) MRK New York Stock Exchange 1.125% Notes due 2021 MRK/21 New York Stock Exchange 0.500% Notes due 2024 MRK 24 New York Stock Exchange 1.875% Notes due 2026 MRK/26 New York Stock Exchange 2.500% Notes due 2034 MRK/34 New York Stock Exchange 1.375% Notes due 2036 MRK 36A New York Stock Exchange Number of shares of Common Stock ($0.50 par value) outstanding as of January 31, 2021: 2,530,315,668. Aggregate market value of Common Stock ($0.50 par value) held by non-affiliates on June 30, 2020 based on closing price on June 30, 2020: $195,461,000,000. Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.