Berkeley, California ...J V1 \Jj -C
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
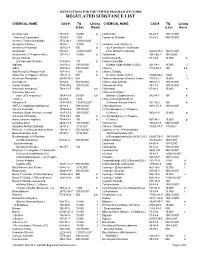
Regulated Substance List
INSTRUCTIONS FOR THE UNIFIED PROGRAM (UP) FORM REGULATED SUBSTANCE LIST CHEMICAL NAME CAS # TQ Listing CHEMICAL NAME CAS # TQ Listing (Lbs) Basis (Lbs) Basis Acetaldehyde 75-07-0 10,000 g Cantharidin 56-25-7 100/10,0001 * Acetone Cyanohydrin 75-86-5 1,000 Carbachol Chloride 51-83-2 500/10,0001 Acetone Thiosemicarbazide 1752-30-3 1,000/10,0001 Acetylene (Ethyne) 74-86-2 10,000 f Carbamic Acid, Methyl-,o- Acrolein (2-Propenal) 107-02-8 500 b (((2,4-Dimethyl-1,3-Dithiolan- Acrylamide 79-06-1 1,000/10,0001 2-YL) Methylene)Amino)- 26419-73-8 100/10,0001 Acrylonitrile (2- Propenenitrile) 107-13-1 10,000 b Carbofuran 1563-66-2 10/10,0001 Acrylyl Chloride Carbon Disulfide 75-15-0 10,000 b (2-Propenoyl Chloride) 814-68-6 100 b Carbon Oxysulfide Aldicarb 116-06-3 100/10,0001 (Carbon Oxide Sulfide (COS)) 463-58-1 10,000 f Aldrin 309-00-2 500/10,0001 Chlorine 7782-50-5 100 a,b Allyl Alcohol (2-Propen-1-ol) 107-18-6 1,000 b Chlorine Dioxide Allylamine (2-Propen-1-Amine) 107-11-9 500 b (Chlorine Oxide (ClO2)) 10049-04-4 1,000 c Aluminum Phosphide 20859-73-8 500 Chlorine Monoxide (Chlorine Oxide) 7791-21-1 10,000 f Aminopterin 54-62-6 500/10,0001 Chlormequat Chloride 999-81-5 100/10,0001 Amiton Oxalate 3734-97-2 100/10,0001 Chloroacetic Acid 79-11-8 100/10,0001 Ammonia, Anhydrous 2 7664-41-7 500 a,b Chloroform 67-66-3 10,000 b Ammonia, Aqueous Chloromethyl Ether (conc 20% or greater) 7664-41-7 20,000 a,b (Methane,Oxybis(chloro-) 542-88-1 100 b * Aniline 62-53-3 1,000 Chloromethyl Methyl Ether Antimycin A 1397-94-0 1,000/10,0001 (Chloromethoxymethane) -

Germane Facts About Germanium Sesquioxide: I. Chemistry and Anticancer Properties
THE JOURNAL OF ALTERNATIVE AND COMPLEMENTARY MEDICINE Volume 10, Number 2, 2004, pp. 337–344 ©Mary Ann Liebert, Inc. Germane Facts About Germanium Sesquioxide: I. Chemistry and Anticancer Properties BONNIEJ. KAPLAN, Ph.D., 1 W. WESLEYPARISH, Ph.D., 2 G. MERRILLANDRUS, Ph.D., 2 J. STEVENA. SIMPSON, Ph.D., M.D., 3 and CATHERINEJ. FIELD, Ph.D., R.D. 4 ABSTRACT This paper reviews the history, chemistry, safety, toxicity, and anticancer effects of the organogermanium compound bis (2-carboxyethylgermanium) sesquioxide (CEGS). A companion review follows, discussing the inaccuracies in the scientific record that have prematurely terminated research on clinical uses of CEGS. CEGS is a unique organogermanium compound first made by Mironov and coworkers in Russia and, shortly there- after, popularized by Asai and his colleagues in Japan. Low concentrations of germanium occur in nearly all soils, plants and animal life; natural occurrence of the CEGS form is postulated but not yet demonstrated. The literature demonstrating its anticancer effect is particularly strong: CEGS induces interferon- g (IFN-g), en- hances natural killer cell activity, and inhibits tumor and metastatic growth—effects often detectable after a single oral dose. In addition, oral consumption of CEGS is readily assimilated and rapidly cleared from the body without evidence of toxicity. Given these findings, the absence of human clinical trials of CEGS is un- expected. Possible explanations of why the convincing findings from animal research have not been used to support clinical trials are discussed. Clinical trials on CEGS are recommended. INTRODUCTION bispropionic acid; 3-oxygermylpropionic acid polymer; poly- trans-(2-carboxyethyl) germasesquioxane); proxigerma- n general, dietary supplements are an underinvestigated nium; repagermanium; and Serocion. -

Safety Data Sheet
SAFETY DATA SHEET Revision Date 27-February-2020 Revision Number 2 1. Identification Product Name Potassium hydride, 30% w/w in mineral oil Cat No. : L13266 Synonyms No information available Recommended Use Laboratory chemicals. Uses advised against Food, drug, pesticide or biocidal product use. Details of the supplier of the safety data sheet Company Importer/Distributor Manufacturer Fisher Scientific Alfa Aesar 112 Colonnade Road, Thermo Fisher Scientific Chemicals, Inc. Ottawa, ON K2E 7L6, 30 Bond Street, Ward Hill, MA 01835-8099 Canada Tel: 800-343-0660 Fax: 800-322-4757 Tel: 1-800-234-7437 Email: [email protected] www.alfa.com Emergency Telephone Number During normal business hours (Monday-Friday, 8am-7pm EST), call (800) 343-0660. After normal business hours, call Carechem 24 at (800) 579-7421. 2. Hazard(s) identification Classification WHMIS 2015 Classification Classified as hazardous under the Hazardous Products Regulations (SOR/2015-17) Substances/mixtures which, in contact with water, emit Category 1 flammable gases Skin Corrosion/Irritation Category 1 B Serious Eye Damage/Eye Irritation Category 1 Specific target organ toxicity (single exposure) Category 3 Target Organs - Respiratory system. Aspiration Toxicity Category 1 Physical Hazards Not Otherwise Classified Category 1 Reacts violently with water Label Elements Signal Word Danger Hazard Statements In contact with water releases flammable gases which may ignite spontaneously ______________________________________________________________________________________________ Page 1 / 7 Potassium hydride, 30% w/w in mineral oil Revision Date 27-February-2020 ______________________________________________________________________________________________ May be fatal if swallowed and enters airways Causes severe skin burns and eye damage May cause respiratory irritation Reacts violently with water Precautionary Statements Manufacturer Alfa Aesar Thermo Fisher Scientific Chemicals, Inc. -

Page 1 of 26 RSC Advances
RSC Advances This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. This Accepted Manuscript will be replaced by the edited, formatted and paginated article as soon as this is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/advances Page 1 of 26 RSC Advances The ternary amide KLi 3(NH 2)4: an important intermediate in the potassium compounds-added Li –N–H systems Bao-Xia Dong, Liang Song, Jun Ge, Yun-Lei Teng*, Shi-Yang Zhang College of Chemistry and Chemical Engineering, Yangzhou University, Yangzhou, 225002, P. R. China. In this paper, the KH-added LiH–NH 3, KH-added LiH–LiNH 2, KH-added LiNH 2, and KNH 2-added LiNH 2 systems were systematically investigated. It was found that the ternary amide KLi 3(NH 2)4 was an important intermediate that was inclined to be formed in the dehydrogenation and hydrogenation processes of the potassium compounds-added Li–N–H system. -

Chemical Chemical Hazard and Compatibility Information
Chemical Chemical Hazard and Compatibility Information Acetic Acid HAZARDS & STORAGE: Corrosive and combustible liquid. Serious health hazard. Reacts with oxidizing and alkali materials. Keep above freezing point (62 degrees F) to avoid rupture of carboys and glass containers.. INCOMPATIBILITIES: 2-amino-ethanol, Acetaldehyde, Acetic anhydride, Acids, Alcohol, Amines, 2-Amino-ethanol, Ammonia, Ammonium nitrate, 5-Azidotetrazole, Bases, Bromine pentafluoride, Caustics (strong), Chlorosulfonic acid, Chromic Acid, Chromium trioxide, Chlorine trifluoride, Ethylene imine, Ethylene glycol, Ethylene diamine, Hydrogen cyanide, Hydrogen peroxide, Hydrogen sulfide, Hydroxyl compounds, Ketones, Nitric Acid, Oleum, Oxidizers (strong), P(OCN)3, Perchloric acid, Permanganates, Peroxides, Phenols, Phosphorus isocyanate, Phosphorus trichloride, Potassium hydroxide, Potassium permanganate, Potassium-tert-butoxide, Sodium hydroxide, Sodium peroxide, Sulfuric acid, n-Xylene. Acetone HAZARDS & STORAGE: Store in a cool, dry, well ventilated place. INCOMPATIBILITIES: Acids, Bromine trifluoride, Bromine, Bromoform, Carbon, Chloroform, Chromium oxide, Chromium trioxide, Chromyl chloride, Dioxygen difluoride, Fluorine oxide, Hydrogen peroxide, 2-Methyl-1,2-butadiene, NaOBr, Nitric acid, Nitrosyl chloride, Nitrosyl perchlorate, Nitryl perchlorate, NOCl, Oxidizing materials, Permonosulfuric acid, Peroxomonosulfuric acid, Potassium-tert-butoxide, Sulfur dichloride, Sulfuric acid, thio-Diglycol, Thiotrithiazyl perchlorate, Trichloromelamine, 2,4,6-Trichloro-1,3,5-triazine -

Germane 99.99+%
GEG5001 - GERMANE 99.99+% GERMANE 99.99+% Safety Data Sheet GEG5001 Date of issue: 01/05/2015 Version: 1.0 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product form : Substance Physical state : Gas Substance name : GERMANE 99.99+% Product code : GEG5001 Formula : GeH4 Synonyms : MONOGERMANE; GERMANIUM HYDRIDE; GERMANIUM TETRAHYDRIDE Chemical family : GERMANE 1.2. Relevant identified uses of the substance or mixture and uses advised against Use of the substance/mixture : Chemical intermediate For research and industrial use only 1.3. Details of the supplier of the safety data sheet GELEST, INC. 11 East Steel Road Morrisville, PA 19067 USA T 215-547-1015 - F 215-547-2484 - (M-F): 8:00 AM - 5:30 PM EST [email protected] - www.gelest.com 1.4. Emergency telephone number Emergency number : CHEMTREC: 1-800-424-9300 (USA); +1 703-527-3887 (International) SECTION 2: Hazards identification 2.1. Classification of the substance or mixture Classification (GHS-US) Flam. Gas 1 H220 Liquefied gas H280 Acute Tox. 2 (Inhalation:gas) H330 Eye Irrit. 2A H319 STOT SE 3 H335 Full text of H-phrases: see section 16 2.2. Label elements GHS-US labeling Hazard pictograms (GHS-US) : GHS02 GHS04 GHS06 GHS07 Signal word (GHS-US) : Danger Hazard statements (GHS-US) : H220 - Extremely flammable gas H280 - Contains gas under pressure; may explode if heated H319 - Causes serious eye irritation H330 - Fatal if inhaled H335 - May cause respiratory irritation Precautionary statements (GHS-US) : P284 - [In case of inadequate ventilation] wear respiratory protection P280 - Wear protective gloves/protective clothing/eye protection/face protection P260 - Do not breathe gas P264 - Wash hands thoroughly after handling P310 - Immediately call a doctor P210 - Keep away from heat, open flames, sparks. -

Solid-State Structures of the Covalent Hydrides Germane and Stannane
Edinburgh Research Explorer Solid-state structures of the covalent hydrides germane and stannane Citation for published version: Maley, IJ, Brown, DH, Ibberson, RM & Pulham, CR 2008, 'Solid-state structures of the covalent hydrides germane and stannane', Acta Crystallographica Section B - Structural Science, vol. 64, no. Pt 3, pp. 312-7. https://doi.org/10.1107/S0108768108010379 Digital Object Identifier (DOI): 10.1107/S0108768108010379 Link: Link to publication record in Edinburgh Research Explorer Document Version: Publisher's PDF, also known as Version of record Published In: Acta Crystallographica Section B - Structural Science Publisher Rights Statement: Copyright © 2008 International Union of Crystallography; all rights reserved. General rights Copyright for the publications made accessible via the Edinburgh Research Explorer is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The University of Edinburgh has made every reasonable effort to ensure that Edinburgh Research Explorer content complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 02. Oct. 2021 electronic reprint Acta Crystallographica Section B Structural Science, Crystal Engineering and Materials ISSN 2052-5192 Solid-state structures of the covalent hydrides germane and stannane Iain J. Maley, Daniel H. Brown, Richard M. Ibberson and Colin R. Pulham Acta Cryst. (2008). B64, 312–317 Copyright c International Union of Crystallography Author(s) of this paper may load this reprint on their own web site or institutional repository provided that this cover page is retained. -

Germane Interim AEGL Document
1 2 3 4 ACUTE EXPOSURE GUIDELINE LEVELS (AEGLs) 5 FOR 6 GERMANE 7 (CAS Reg. No. 7782-65-2) 8 9 GeH4 10 11 12 13 14 15 16 17 INTERIM 18 19 GERMANE Interim: 09-2009/ Page 2 of 31 1 2 ACUTE EXPOSURE GUIDELINE LEVELS (AEGLs) 3 FOR 4 GERMANE 5 (CAS Reg. No. 7782-65-2) 6 7 8 9 10 INTERIM 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 GERMANE Interim: 09-2009/ Page 3 of 31 1 PREFACE 2 3 Under the authority of the Federal Advisory Committee Act (FACA) P. L. 92-463 of 4 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous 5 Substances (NAC/AEGL Committee) has been established to identify, review and interpret 6 relevant toxicologic and other scientific data and develop AEGLs for high priority, acutely toxic 7 chemicals. 8 9 AEGLs represent threshold exposure limits for the general public and are applicable to 10 emergency exposure periods ranging from 10 minutes to 8 hours. Three levels C AEGL-1, 11 AEGL-2 and AEGL-3 C are developed for each of five exposure periods (10 and 30 minutes, 1 12 hour, 4 hours, and 8 hours) and are distinguished by varying degrees of severity of toxic effects. 13 The three AEGLs are defined as follows: 14 15 AEGL-1 is the airborne concentration (expressed as parts per million or milligrams per 16 cubic meter [ppm or mg/m3]) of a substance above which it is predicted that the general 17 population, including susceptible individuals, could experience notable discomfort, irritation, or 18 certain asymptomatic, non-sensory effects. -

Digermane SDS 1
SAFETY DATA SHEET Prepared to U.S. OSHA, CMA, ANSI, Canadian WHMIS Standards, European Union CLP EC 1272/2008, REACH, and the Global Harmonization Standard 1. SECTION 1 – IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING CHEMICAL NAME; CLASS: DIGERMANE SYNONYMS: Digerman; λ2-germanylidenegermanium; Germanium Hexahydride; Germanium Hydride, Germanium (III) Hydride, Germanium Trihydride CHEMICAL FAMILY: Hydride FORMULA: Ge 2H6 Docu ment Numbe r: 80 019 PR ODUCT USE: Various MANU FA CTUR ED/SU PPL IED FOR: SUPPLIER/MANUFACTURER'S NAME: AIR LIQUIDE AMERICA ADDRESS : 2700 Post Oak Drive Hous ton , TX 7705 6-822 9 EM AIL ADD RES S FOR PRODUC T INF ORM ATION: webmas ter.us @airliqu ide.co m EMERGENCY PHONE: CHEMTREC: (U.S., Canada) 1-800-424-9300 (24 hrs) (International) +703-527-3887 (collec t-24 hrs) BUSINESS PHONE: General MSDS Information: 1-713/896-2896 (8 am to 5 pm U.S. Central Time) Fax on Demand: 1-800/231-1366 ALL WHMIS required information is included in appropriate sections based on the ANSI Z400.1-2004 format. This product has been classified in accordance with the hazard criteria of the CPR and the MSDS contains all the information required by the CPR. The product is also classified per all applicable European Union CLP EC 1272/2008 and the Global Harmonization Standard. TSCA Status: This material is not included in the TSCA Inventory. In accordance with the conditions listed in 40 CFR 720.36 and 721.47, this product must be used only for research and development, pharmaceutical manufacture, or export. -

Chapter 9 Hydrogen
Chapter 9 Hydrogen Diborane, B2 H6• is the simplest member of a large class of compounds, the electron-deficient boron hydrid Like all boron hydrides, it has a positive standard free energy of formation, and so cannot be prepared direct from boron and hydrogen. The bridge B-H bonds are longer and weaker than the tenninal B-H bonds (13: vs. 1.19 A). 89.1 Reactions of hydrogen compounds? (a) Ca(s) + H2Cg) ~ CaH2Cs). This is the reaction of an active s-me: with hydrogen, which is the way that saline metal hydrides are prepared. (b) NH3(g) + BF)(g) ~ H)N-BF)(g). This is the reaction of a Lewis base and a Lewis acid. The product I Lewis acid-base complex. (c) LiOH(s) + H2(g) ~ NR. Although dihydrogen can behave as an oxidant (e.g., with Li to form LiH) or reductant (e.g., with O2 to form H20), it does not behave as a Br0nsted or Lewis acid or base. It does not r with strong bases, like LiOH, or with strong acids. 89.2 A procedure for making Et3MeSn? A possible procedure is as follows: ~ 2Et)SnH + 2Na 2Na+Et3Sn- + H2 Ja'Et3Sn- + CH3Br ~ Et3MeSn + NaBr 9.1 Where does Hydrogen fit in the periodic chart? (a) Hydrogen in group 1? Hydrogen has one vale electron like the group 1 metals and is stable as W, especially in aqueous media. The other group 1 m have one valence electron and are quite stable as ~ cations in solution and in the solid state as simple 1 salts. -

HIGH HAZARD GAS Review Date: 09/23/2019
University of Pittsburgh EH&S Guideline Number: 04-021 Safety Manual Subject: Effective Date: 04/19/2017 Page 1 of 9 HIGH HAZARD GAS Review Date: 09/23/2019 STORAGE AND USE OF HIGH HAZARD GAS 1. Definition of High Hazard (HH) Gases For these guidelines, any gas meeting one or more of the following definitions based on International Fire Code (IFC) and National Fire Protection Association (NFPA) standards: 1.1. Flammable gas – a material that is a gas at 68ºF (20ºC) or less at an absolute pressure of 14.7 psi (101.325 kPa) when in a mixture of 13% or less by volume with air, or that has a flammable range at an absolute pressure of 14.7 psi (101.325 kPa) with air of at least 12%, regardless of the lower limit 1.2. Pyrophoric gas – a gas with an autoignition temperature in air at or below 130ºF (54.4ºC) 1.3. Health Hazard 3 (HH3) gas – material that, under emergency conditions and according to the standards, can cause serious or permanent injury 1.4. Health Hazard 4 (HH4) gas – material that, under emergency conditions and according to the standards, can be lethal The storage and usage of a gas or gases meeting any of the above definitions must follow all applicable IFC and NFPA guidelines and the requirements outlined in this document. Consult EH&S for specific guidance on gas mixtures containing corrosive, flammable or poisonous gas components (ex. 1% carbon monoxide/nitrogen, 5% hydrogen sulfide/helium). 2. Notification Requirements Prior to Obtaining High Hazard Gases 2.1. -

Stannane 2629
TRIMETHPLSTANNANE 2629 [CONTRIBUTION FROM THE CHEWCAL LABORATORY OF CLARK UNIVERSITY, I, 161 THE PREPARATION AND PROPERTIES OF TRIMETHYL- STANNANE BY CHARLESA. KRAUSAUD WILLARDTu'. GREER Recci\ed .4ugiist 11 1922 Introduction.-The hydrogen derivatives of the various elements decreasc in stability as we proceed from the elements of the halogen group to the elements of higher negative 1-alence,and as wc proceed from elements of lower to elements of higher atomic weight. A11 the elements of the halogen group give stable hydrides at ordinary temperatures. The hydrides of the higher member4 of the oxygen group have comparatively little stability, while in the case of the nitrogen group only nitrogen and phosphorus form comparatively stable normal hydrides. Of the fourth group of elements, carbon and silicon form stable hydrides, but the higher members form hydrides possessing little stability. It is only recently that the existence of hydrides o€ tin, lend and bismuth has been estab- lished.' ff one or more of the 111 drogens of a hydride is replaced by an alkyl or am1 group, the stability of the resulting compound is greatly increased over that of the hydride. Kearly all the common elements yield stable compounds of this type when all the hydrogens are substituted in this way. Tlie metallo-organic compounds of the elements of the fourth group exhibit unusual stability, corresponding somewhat to the exceptional stability of methane, the first member of the group. The tetra-alkyl tin compounds are, on the whole, very stable substances. It might be expected, therefore, that, in the case of tin if only a portion of the hydrogens mere wbstituted by alkyl groups, the resulting compound would exhibit a con- siclt~abledegree of stability.