De Novo Mutations in Epileptic Encephalopathies
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Congenital Disorders of Glycosylation from a Neurological Perspective
brain sciences Review Congenital Disorders of Glycosylation from a Neurological Perspective Justyna Paprocka 1,* , Aleksandra Jezela-Stanek 2 , Anna Tylki-Szyma´nska 3 and Stephanie Grunewald 4 1 Department of Pediatric Neurology, Faculty of Medical Science in Katowice, Medical University of Silesia, 40-752 Katowice, Poland 2 Department of Genetics and Clinical Immunology, National Institute of Tuberculosis and Lung Diseases, 01-138 Warsaw, Poland; [email protected] 3 Department of Pediatrics, Nutrition and Metabolic Diseases, The Children’s Memorial Health Institute, W 04-730 Warsaw, Poland; [email protected] 4 NIHR Biomedical Research Center (BRC), Metabolic Unit, Great Ormond Street Hospital and Institute of Child Health, University College London, London SE1 9RT, UK; [email protected] * Correspondence: [email protected]; Tel.: +48-606-415-888 Abstract: Most plasma proteins, cell membrane proteins and other proteins are glycoproteins with sugar chains attached to the polypeptide-glycans. Glycosylation is the main element of the post- translational transformation of most human proteins. Since glycosylation processes are necessary for many different biological processes, patients present a diverse spectrum of phenotypes and severity of symptoms. The most frequently observed neurological symptoms in congenital disorders of glycosylation (CDG) are: epilepsy, intellectual disability, myopathies, neuropathies and stroke-like episodes. Epilepsy is seen in many CDG subtypes and particularly present in the case of mutations -

Associated with Low Dehydrodolichol Diphosphate Synthase (DHDDS) Activity S
Sabry et al. Orphanet Journal of Rare Diseases (2016) 11:84 DOI 10.1186/s13023-016-0468-1 RESEARCH Open Access A case of fatal Type I congenital disorders of glycosylation (CDG I) associated with low dehydrodolichol diphosphate synthase (DHDDS) activity S. Sabry1,2,3,4, S. Vuillaumier-Barrot1,2,5, E. Mintet1,2, M. Fasseu1,2, V. Valayannopoulos6, D. Héron7,8, N. Dorison8, C. Mignot7,8,9, N. Seta5,10, I. Chantret1,2, T. Dupré1,2,5 and S. E. H. Moore1,2* Abstract Background: Type I congenital disorders of glycosylation (CDG-I) are mostly complex multisystemic diseases associated with hypoglycosylated serum glycoproteins. A subgroup harbour mutations in genes necessary for the biosynthesis of the dolichol-linked oligosaccharide (DLO) precursor that is essential for protein N-glycosylation. Here, our objective was to identify the molecular origins of disease in such a CDG-Ix patient presenting with axial hypotonia, peripheral hypertonia, enlarged liver, micropenis, cryptorchidism and sensorineural deafness associated with hypo glycosylated serum glycoproteins. Results: Targeted sequencing of DNA revealed a splice site mutation in intron 5 and a non-sense mutation in exon 4 of the dehydrodolichol diphosphate synthase gene (DHDDS). Skin biopsy fibroblasts derived from the patient revealed ~20 % residual DHDDS mRNA, ~35 % residual DHDDS activity, reduced dolichol-phosphate, truncated DLO and N-glycans, and an increased ratio of [2-3H]mannose labeled glycoprotein to [2-3H]mannose labeled DLO. Predicted truncated DHDDS transcripts did not complement rer2-deficient yeast. SiRNA-mediated down-regulation of DHDDS in human hepatocellular carcinoma HepG2 cells largely mirrored the biochemical phenotype of cells from the patient. -
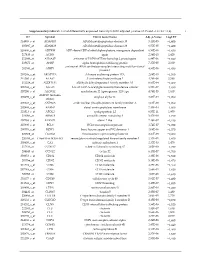
Supplementary Table S1. List of Differentially Expressed
Supplementary table S1. List of differentially expressed transcripts (FDR adjusted p‐value < 0.05 and −1.4 ≤ FC ≥1.4). 1 ID Symbol Entrez Gene Name Adj. p‐Value Log2 FC 214895_s_at ADAM10 ADAM metallopeptidase domain 10 3,11E‐05 −1,400 205997_at ADAM28 ADAM metallopeptidase domain 28 6,57E‐05 −1,400 220606_s_at ADPRM ADP‐ribose/CDP‐alcohol diphosphatase, manganese dependent 6,50E‐06 −1,430 217410_at AGRN agrin 2,34E‐10 1,420 212980_at AHSA2P activator of HSP90 ATPase homolog 2, pseudogene 6,44E‐06 −1,920 219672_at AHSP alpha hemoglobin stabilizing protein 7,27E‐05 2,330 aminoacyl tRNA synthetase complex interacting multifunctional 202541_at AIMP1 4,91E‐06 −1,830 protein 1 210269_s_at AKAP17A A‐kinase anchoring protein 17A 2,64E‐10 −1,560 211560_s_at ALAS2 5ʹ‐aminolevulinate synthase 2 4,28E‐06 3,560 212224_at ALDH1A1 aldehyde dehydrogenase 1 family member A1 8,93E‐04 −1,400 205583_s_at ALG13 ALG13 UDP‐N‐acetylglucosaminyltransferase subunit 9,50E‐07 −1,430 207206_s_at ALOX12 arachidonate 12‐lipoxygenase, 12S type 4,76E‐05 1,630 AMY1C (includes 208498_s_at amylase alpha 1C 3,83E‐05 −1,700 others) 201043_s_at ANP32A acidic nuclear phosphoprotein 32 family member A 5,61E‐09 −1,760 202888_s_at ANPEP alanyl aminopeptidase, membrane 7,40E‐04 −1,600 221013_s_at APOL2 apolipoprotein L2 6,57E‐11 1,600 219094_at ARMC8 armadillo repeat containing 8 3,47E‐08 −1,710 207798_s_at ATXN2L ataxin 2 like 2,16E‐07 −1,410 215990_s_at BCL6 BCL6 transcription repressor 1,74E‐07 −1,700 200776_s_at BZW1 basic leucine zipper and W2 domains 1 1,09E‐06 −1,570 222309_at -

Characterization of X-Linked SNP Genotypic Variation in Globally Distributed Human Populations Genome Biology 2010, 11:R10
Casto et al. Genome Biology 2010, 11:R10 http://genomebiology.com/2010/11/1/R10 RESEARCH Open Access CharacterizationResearch of X-Linked SNP genotypic variation in globally distributed human populations Amanda M Casto*1, Jun Z Li2, Devin Absher3, Richard Myers3, Sohini Ramachandran4 and Marcus W Feldman5 HumanAnhumanulation analysis structurepopulationsX-linked of X-linked variationand provides de geneticmographic insights variation patterns. into in pop- Abstract Background: The transmission pattern of the human X chromosome reduces its population size relative to the autosomes, subjects it to disproportionate influence by female demography, and leaves X-linked mutations exposed to selection in males. As a result, the analysis of X-linked genomic variation can provide insights into the influence of demography and selection on the human genome. Here we characterize the genomic variation represented by 16,297 X-linked SNPs genotyped in the CEPH human genome diversity project samples. Results: We found that X chromosomes tend to be more differentiated between human populations than autosomes, with several notable exceptions. Comparisons between genetically distant populations also showed an excess of X- linked SNPs with large allele frequency differences. Combining information about these SNPs with results from tests designed to detect selective sweeps, we identified two regions that were clear outliers from the rest of the X chromosome for haplotype structure and allele frequency distribution. We were also able to more precisely define the geographical extent of some previously described X-linked selective sweeps. Conclusions: The relationship between male and female demographic histories is likely to be complex as evidence supporting different conclusions can be found in the same dataset. -

Targeted Next-Generation Sequencing in Patients with Suggestive X-Linked Intellectual Disability
G C A T T A C G G C A T genes Article Targeted Next-Generation Sequencing in Patients with Suggestive X-Linked Intellectual Disability Nekane Ibarluzea 1,2 , Ana Belén de la Hoz 1,2, Olatz Villate 1,2,3, Isabel Llano 1,2,3 , Intzane Ocio 4, Itxaso Martí 5, Miriam Guitart 6, Elisabeth Gabau 6, Fernando Andrade 1,2, Blanca Gener 1,2,3 and María-Isabel Tejada 1,2,3,* 1 Biocruces Bizkaia Health Research Institute, 48903 Barakaldo, Spain; [email protected] (N.I.); [email protected] (A.B.d.l.H.); [email protected] (O.V.); [email protected] (I.L.); [email protected] (F.A.); [email protected] (B.G.) 2 Spanish Consortium for Research on Rare Diseases (CIBERER), 28029 Madrid, Spain 3 Genetics Service, Cruces University Hospital, Osakidetza Basque Health Service, 48903 Barakaldo, Spain 4 Department of Paediatric Neurology, Araba University Hospital, Osakidetza Basque Health Service, 01009 Gasteiz, Spain; [email protected] 5 Department of Paediatric Neurology, Donostia University Hospital, Osakidetza Basque Health Service, 20014 Donostia, Spain; [email protected] 6 Genetics Laboratory, Paediatric Unit, Parc Taulí Hospital Universitari, Institut d’Investigació i Innovació Parc Taulí I3PT, Universitat Autònoma de Barcelona, 08208 Sabadell, Spain; [email protected] (M.G.); [email protected] (E.G.) * Correspondence: [email protected] Received: 3 December 2019; Accepted: 30 December 2019; Published: 2 January 2020 Abstract: X-linked intellectual disability (XLID) is known to contribute up to 10% of intellectual disability (ID) in males and could explain the increased ratio of affected males observed in patients with ID. -
Alg13/Alg14 UDP-Glcnac Transferase
Heterodimeric Alg13/Alg14 UDP-GlcNAc transferase Introduction As one of the most frequent and common posttranslational modifications in eukaryotic cell, protein N-glycosylation begins with the multistep biosynthesis of an evolutionarily conserved lipid-linked oligosaccharide (LLO), which is extended by a series of membrane-associated Alg (asparagine-linked glycosylation) glycosyltransferases to produce Glc3Man9GlcNAc2-PP-dolichol (dol) in the endoplasmic reticulum (ER). As shown in figure 1, assembly of the first seven sugars in the LLO synthetic pathway takes place on the cytoplasmic face of the ER membrane, and is catalyzed by cytosolic glycosyltransferases that produce Man5GlcNAc2-PP-dol from the soluble nucleotide sugar substrates UDP-GlcNAc and GDP-man. This dol-linked intermediate then flips into the lumen of the ER where the next seven sugars are added from dol-linked sugar substrates to complete the assembly of the tetradecasaccharide on dolichol diphosphate. An UDP-N-acetylglucosamine transferase (accepted name: N-acetylglucosaminyldiphosphodolichol N-acetylglucosaminyltransferase) catalyzes the second step of LLO synthesis that adds N-acetylglucosamine (GlcNAc) to GlcNAc-pp-dol to produce GlcNAc2-PP-dol. Unlike any other Alg glycosyltransferases, this UDP-GlcNAc transferase is unique because it comprised of two subunits, Alg13 and Alg14 proteins (Figure 1). Alg13 and Alg14 homologous can be found in virtually all eukaryotic cells including man, mammals, insects, worms, plants and fungi. In yeast, both Alg13 and Alg14 subunits -

Landscape of X Chromosome Inactivation Across Human Tissues Taru Tukiainen1,2, Alexandra-Chloé Villani2,3, Angela Yen2,4, Manuel A
OPEN LETTER doi:10.1038/nature24265 Landscape of X chromosome inactivation across human tissues Taru Tukiainen1,2, Alexandra-Chloé Villani2,3, Angela Yen2,4, Manuel A. Rivas1,2,5, Jamie L. Marshall1,2, Rahul Satija2,6,7, Matt Aguirre1,2, Laura Gauthier1,2, Mark Fleharty2, Andrew Kirby1,2, Beryl B. Cummings1,2, Stephane E. Castel6,8, Konrad J. Karczewski1,2, François Aguet2, Andrea Byrnes1,2, GTEx Consortium†, Tuuli Lappalainen6,8, Aviv Regev2,9, Kristin G. Ardlie2, Nir Hacohen2,3 & Daniel G. MacArthur1,2 X chromosome inactivation (XCI) silences transcription from Given the limited accessibility of most human tissues, particularly one of the two X chromosomes in female mammalian cells to in large sample sizes, no global investigation into the impact of incom- balance expression dosage between XX females and XY males. plete XCI on X-chromosomal expression has been conducted in data- XCI is, however, incomplete in humans: up to one-third of sets spanning multiple tissue types. We used the Genotype-Tissue X-chromosomal genes are expressed from both the active and Expression (GTEx) project12,13 dataset (v6p release), which includes inactive X chromosomes (Xa and Xi, respectively) in female cells, high-coverage RNA-seq data from diverse human tissues, to investi- with the degree of ‘escape’ from inactivation varying between genes gate male–female differences in the expression of 681 X-chromosomal and individuals1,2. The extent to which XCI is shared between cells genes that encode proteins or long non-coding RNA in 29 adult tissues and tissues remains poorly characterized3,4, as does the degree to (Extended Data Table 1), hypothesizing that escape from XCI should which incomplete XCI manifests as detectable sex differences in gene typically result in higher female expression of these genes. -

Predominant and Novel De Novo Variants in 29 Individuals with ALG13 Deficiency: Clinical
Predominant and novel de novo variants in 29 individuals with ALG13 deficiency: Clinical description, biomarker status, biochemical analysis and treatment suggestions. Bobby G. Ng1, Erik A. Eklund1,2, Sergey A. Shiryaev1, Yin Y. Dong3, Mary-Alice Abbott4, Carla Asteggiano5, Michael J. Bamshad6,7, Eileen Barr8, Jonathan A. Bernstein9,10, Shabeed Chelakkadan11, John Christodoulou12, 13, Wendy K. Chung14, 15, Michael A. Ciliberto16, Janice Cousin17, Fiona Gardiner18, Suman Ghosh19, William D. Graf20, Stephanie Grunewald21, Katherine Hammond22, Natalie S. Hauser23, George E. Hoganson24, Kimberly M. Houck25, Jennefer N. Kohler9, 26, Eva Morava27, Austin A. Larson28, Pengfei Liu29, 30, Sujana Madathil16, Colleen McCormack9, 26, Naomi J.L. Meeks28, Rebecca Miller23, Kristin G. Monaghan31, Deborah A. Nickerson7, Timothy Blake Palculict31, Gabriela Magali Papazoglu32 , Beth A. Pletcher33, Ingrid E. Scheffer18,,34, Andrea Beatriz Schenone35, Rhonda E. Schnur31, Yue Si31, Leah J. Rowe28, Alvaro H. Serrano Russi36, 37, Rossana Sanchez Russo8, Farouq Thabet38, Allysa Tuite33, María Mercedes Villanueva35, Raymond Y. Wang39, 40, Richard I. Webster41, 42, Dorcas Wilson43, 44, Alice Zalan24, Undiagnosed Diseases Network, University of Washington Center for Mendelian Genomics (UW-CMG), Lynne A. Wolfe45, Jill A. Rosenfeld29, 30, Lindsay Rhodes31, Hudson H. Freeze1 This article has been accepted for publication and undergone full peer review but has not been through the copyediting, typesetting, pagination and proofreading process which may lead to differences between this version and the Version of Record. Please cite this article as doi: 10.1002/jimd.12290 This article is protected by copyright. All rights reserved. (1) Human Genetics Program, Sanford Burnham Prebys Medical Discovery Institute, La Jolla, CA 92037, USA. (2) Lund University, Department of Clinical Sciences, Lund, Pediatrics, Lund, Sweden. -
The Phenotypic Spectrum of X‐Linked, Infantile Onset ALG13‐Related
Received: 11 June 2020 | Revised: 27 September 2020 | Accepted: 23 October 2020 DOI: 10.1111/epi.16761 FULL-LENGTH ORIGINAL RESEARCH The phenotypic spectrum of X-linked, infantile onset ALG13- related developmental and epileptic encephalopathy Alexandre N. Datta1 | Nadia Bahi-Buisson2 | Thierry Bienvenu3 | Sarah E. Buerki4 | Fiona Gardiner5 | J. Helen Cross6 | Bénédicte Heron7 | Anna Kaminska8 | Christian M. Korff9 | Anne Lepine10 | Gaetan Lesca11 | Amy McTague6 | Heather C. Mefford12 | Cyrill Mignot13 | Matthieu Milh9 | Amélie Piton14 | Ronit M. Pressler6,15 | Susanne Ruf16 | Lynette G. Sadleir17 | Anne de Saint Martin18 | Koen Van Gassen19 | Nienke E. Verbeek19 | Dorothée Ville20 | Nathalie Villeneuve10 | Pia Zacher21 | Ingrid E. Scheffer5,22 | Johannes R. Lemke23 1Pediatric Neurology and Developmental Medicine Department, University Children's Hospital, University of Basel, Basel, Switzerland 2Pediatric Neurology, Necker–Enfants Malades Children's Hospital, Paris and Institute IMAGINE, INSERM U1163, University of Paris, Paris, France 3Paris Institute of Psychiatry and Neuroscience, University of Paris, Paris, France 4Pediatric Neurology Department, University Children's Hospital Zürich, Switzerland 5Austin Health, University of Melbourne, Melbourne, Victoria, Australia 6Clinical Neuroscience, University College London–Great Ormond Street Institute of Child Health, London, UK 7Pediatric Neurology Department, Armand Trousseau–La Roche Guyon University Hospital, APHP and GRC No. 19, Sorbonne Universities, Paris, France 8Department of Clinical -

The Transcriptomic Architecture of Mouse Sertoli Cell Clone Embryos Reveals Temporal–Spatial-Specific Reprogramming
REPRODUCTIONRESEARCH The transcriptomic architecture of mouse Sertoli cell clone embryos reveals temporal–spatial-specific reprogramming Feng Cao, Atsushi Fukuda, Hiroshi Watanabe and Tomohiro Kono Department of Bioscience, Tokyo University of Agriculture, Setagaya-ku, Tokyo 156-8502, Japan Correspondence should be addressed to T Kono; Email: [email protected] Abstract Somatic cell nuclear transfer, a technique used to generate clone embryos by transferring the nucleus of a somatic cell into an enucleated oocyte, is an excellent approach to study the reprogramming of the nuclei of differentiated cells. Here, we conducted a transcriptomic study by performing microarray analysis on single Sertoli cell nuclear transfer (SeCNT) embryos throughout preimplantation development. The extensive data collected from the oocyte to the blastocyst stage helped to identify specific genes that were incorrectly reprogrammed at each stage, thereby providing a novel perspective for understanding reprogramming progression in SeCNT embryos. This attempt provided an opportunity to discuss the possibility that ectopic gene expression could be involved in the developmental failure of SeCNT embryos. Network analysis at each stage suggested that in total, 127 networks were involved in developmental and functional disorders in SeCNT embryos. Furthermore, chromosome mapping using our time-lapse expression data highlighted temporal–spatial changes of the abnormal expression, showing the characteristic distribution of the genes on each chromosome. Thus, the present study revealed that the preimplantation development of SeCNT embryos appears normal; however, the progression of incorrect reprogramming is concealed throughout development. Reproduction (2013) 145 277–288 Introduction into an enucleated oocyte (Gurdon et al. 1976), and Q-PCR studies have revealed that many genes were With progression of mammalian development, which abnormally expressed in SCNT embryos (Bortvin et al. -

Genetic Characteristics of Non-Familial Epilepsy
Genetic characteristics of non-familial epilepsy Kyung Wook Kang1, Wonkuk Kim2, Yong Won Cho3, Sang Kun Lee4, Ki-Young Jung4, Wonchul Shin5, Dong Wook Kim6, Won-Joo Kim7, Hyang Woon Lee8, Woojun Kim9, Keuntae Kim3, So-Hyun Lee10, Seok-Yong Choi10 and Myeong-Kyu Kim1 1 Department of Neurology, Chonnam National University Medical School, Gwangju, South Korea 2 Department of Applied Statistics, Chung-Ang University, Seoul, South Korea 3 Department of Neurology, Keimyung University Dongsan Medical Center, Daegu, South Korea 4 Department of Neurology, Seoul National University Hospital, Seoul, South Korea 5 Department of Neurology, Kyung Hee University Hospital at Gangdong, Seoul, South Korea 6 Department of Neurology, Konkuk University School of Medicine, Seoul, South Korea 7 Department of Neurology, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul, South Korea 8 Department of Neurology, Ewha Womans University School of Medicine and Ewha Medical Research Institute, Seoul, South Korea 9 Department of Neurology, Seoul St. Mary’s Hospital, College of Medicine, The Catholic University of Korea, Seoul, South Korea 10 Department of Biomedical Science, Chonnam National University Medical School, Gwangju, South Korea ABSTRACT Background: Knowledge of the genetic etiology of epilepsy can provide essential prognostic information and influence decisions regarding treatment and management, leading us into the era of precision medicine. However, the genetic basis underlying epileptogenesis or epilepsy pharmacoresistance is not well-understood, particularly in non-familial epilepsies with heterogeneous phenotypes that last until or start in adulthood. Methods: We sought to determine the contribution of known epilepsy-associated Submitted 25 June 2019 genes (EAGs) to the causation of non-familial epilepsies with heterogeneous Accepted 22 November 2019 phenotypes and to the genetic basis underlying epilepsy pharmacoresistance. -

1 Supplementary Material Figure S1. Volcano Plot of Differentially
Supplementary material Figure S1. Volcano Plot of differentially expressed genes between preterm infants fed own mother’s milk (OMM) or pasteurized donated human milk (DHM). Table S1. The 10 most representative biological processes filtered for enrichment p- value in preterm infants. Biological Processes p-value Quantity of DEG* Transcription, DNA-templated 3.62x10-24 189 Regulation of transcription, DNA-templated 5.34x10-22 188 Transport 3.75x10-17 140 Cell cycle 1.03x10-13 65 Gene expression 3.38x10-10 60 Multicellular organismal development 6.97x10-10 86 1 Protein transport 1.73x10-09 56 Cell division 2.75x10-09 39 Blood coagulation 3.38x10-09 46 DNA repair 8.34x10-09 39 Table S2. Differential genes in transcriptomic analysis of exfoliated epithelial intestinal cells between preterm infants fed own mother’s milk (OMM) and pasteurized donated human milk (DHM). Gene name Gene Symbol p-value Fold-Change (OMM vs. DHM) (OMM vs. DHM) Lactalbumin, alpha LALBA 0.0024 2.92 Casein kappa CSN3 0.0024 2.59 Casein beta CSN2 0.0093 2.13 Cytochrome c oxidase subunit I COX1 0.0263 2.07 Casein alpha s1 CSN1S1 0.0084 1.71 Espin ESPN 0.0008 1.58 MTND2 ND2 0.0138 1.57 Small ubiquitin-like modifier 3 SUMO3 0.0037 1.54 Eukaryotic translation elongation EEF1A1 0.0365 1.53 factor 1 alpha 1 Ribosomal protein L10 RPL10 0.0195 1.52 Keratin associated protein 2-4 KRTAP2-4 0.0019 1.46 Serine peptidase inhibitor, Kunitz SPINT1 0.0007 1.44 type 1 Zinc finger family member 788 ZNF788 0.0000 1.43 Mitochondrial ribosomal protein MRPL38 0.0020 1.41 L38 Diacylglycerol