Microbiology Specimen Collection and Transport Guide
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Use of Cell Culture in Virology for Developing Countries in the South-East Asia Region © World Health Organization 2017
USE OF CELL C USE OF CELL U LT U RE IN VIROLOGY FOR DE RE IN VIROLOGY V ELOPING C O U NTRIES IN THE NTRIES IN S O U TH- E AST USE OF CELL CULTURE A SIA IN VIROLOGY FOR R EGION ISBN: 978-92-9022-600-0 DEVELOPING COUNTRIES IN THE SOUTH-EAST ASIA REGION World Health House Indraprastha Estate, Mahatma Gandhi Marg, New Delhi-110002, India Website: www.searo.who.int USE OF CELL CULTURE IN VIROLOGY FOR DEVELOPING COUNTRIES IN THE SOUTH-EAST ASIA REGION © World Health Organization 2017 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial- ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition.” Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. -

Food Packaging (FS 522 / FS 495) Aseptic Processing and Packaging
Food Packaging (FS 522 / FS 495) Aseptic Processing and Packaging Components of an aseptic processing system Aseptic processing is a thermal process in which the product and container are sterilized separately and brought together in a sterile environment. It involves pumping, deaeration, and sterilization of a food product, followed by holding it for a specified period of time (in a holding tube -- required to have a 1/4" rise per foot length of tube), cooling it, and finally packaging it in a sterile container. The use of high temperature for a short period of time (in comparison with conventional canning) in aseptic processing yields a high quality product. Care should be taken to ensure that all process calculations are performed after the deaeration stage and not based on the initial raw product. Deaeration is accomplished in a vessel maintained at a certain degree of vacuum by means of a vacuum pump. The product is fed into the vessel at 55 - 70 /C through a nozzle at the center of the vessel. Vacuum is controlled to obtain a product flash of about 5 /C. An internal spiral condenser condenses vapors and other condensable gases. The deaerated product is discharged through the bottom and pumped to the heating section. Another important part of an aseptic processing system is the back pressure valve which provides sufficient pressure to prevent boiling of the product at processing temperatures which can be as high as 125-130 /C. An aseptic surge tank provides the means for product to be continuously processed even if the packaging system is not operational due to any malfunction. -

Commercial Model of the Future
BioPharma BD Quarterly Newsletter Q2 2018 Syneos Health Consulting Commercial Strategy & Planning | Portfolio Strategy Solution Center Syneos Health BioPharma BD Quarterly Newsletter | Q2 2018 Q2 FDA Approvals | Q3 PDUFA Dates | Q2 Deal Summary | Q2 Mergers & Acquisitions | Q2 Partnerships | Q2 Asset Purchases Q2 FDA Approvals1,2 In order of 2020 sales forecast No sales listed Aimovig (erenumab) | Novartis / Amgen (co- Akynzeo IV (fosnetupitant) | Helsinn Group commercialize) . Prevention of acute and delayed nausea Migraine, first CGRP receptor blocker associated with cancer chemotherapy . 2020E Sales: $470M Bendamustine HCl Injection (bendamustine AndexXa (andexanet alfa) | Portola hydrochloride) | Spectrum Pharmaceuticals / Pharmaceuticals Eagle Pharmaceuticals (co-promotion Neel Patel . Used for uncontrolled bleeding in patients that agreement) used rivaroxaban and apixaban . Chronic lymphocytic leukemia Managing Director . 2020E Sales: $258M [email protected] Consensi (amlodipine besylate) | Kitov / Dexcel Fulphila (pegfilgrastim) | Mylan / Biocon Pharma Technology (cooperation agreement- (partnership) Dexcel will develop drugs final formulation-Kitov . Decrease incidence of infection, as manifested by pays Dexcel $3.5M for company’s facilities) febrile neutropenia . Amlodipine for hypertension and celecoxib for . 2020E Sales: $244M osteoarthritis Lokelma (sodium zirconium cyclosilicate) | Crysvita (burosumab) | Ultragenyx AstraZeneca / Incyte (clinical trial collaboration) Pharmaceutical . Treatment of hyperkalemia in adults -

Gas Generator Bottle Introduction SCIENTIFIC This Gas Generator Setup Provides an Easy Way to Generate and Collect Gas
Gas Generator Bottle Introduction SCIENTIFIC This gas generator setup provides an easy way to generate and collect gas. Specific instructions are provided for the generation of hydrogen gas using zinc and acid. Concepts • Generation of gases • Water displacement Materials Hydrochloric acid solution, HCl, 3 M Glass plates or Sulfuric acid solution, H2SO4, 3 M Glass tubing Mossy zinc, Zn, 6 g Pneumatic trough Water, tap Rubber tubing Bent glass tubing* Silicone grease packet* Gas collecting bottles or tubes, 3 or 4 Thistle tube* Gas generator bottle* Two-hole rubber stopper* *Materials included. Safety Precautions Hydrochloric acid solution is toxic by ingestion and inhalation and is severely corrosive to skin, eyes and other tissues, as is sulfuric acid solu- tion. Hydrogen gas is a highly flammable gas and a severe fire hazard. Exercise extreme caution when testing the gas and keep the gas generator away from flames. Wear chemical splash goggles, chemical-resistant gloves, and a chemical-resistant apron. This activity requires the use of hazardous components and/or has the potential for hazardous reactions. Please review current Material Safety Data Sheets for additional safety, handling, and disposal information. Procedure 1. Set up the apparatus as shown in the figure to the right. Lubricate the glass tubing and thistle tube with silicone grease before inserting into the stopper. Make sure Thistle tube the water level is above the platform. Prepare bottles for collecting gas by water Two-hole rubber stopper displacement. To do this, fill each gas collecting bottle (or tube) over the brim with tap water, and then cover each with a flat glass plate. -

Trace Elements Specimen Collection Guide
500 Chipeta Way Salt Lake City, Utah 84108-1221 Phone: 801-583-2787 | toll free: 800-242-2787 A nonprofit enterprise of the University of Utah and its Department of Pathology Fax: 801-522-2706 | aruplab.com TRACE ELEMENTS SPECIMEN COLLECTION GUIDE Analysis for trace elements at ARUP is performed in a clean laboratory environment that includes a system of positive- pressure and HEPA-filtered air. Since detection limits for many trace elements are calculated in parts per billion, this helps minimize environmental contamination of specimens. Additionally, contamination control during specimen collection provides more accurate and clinically useful results. Note: Refer to the ARUP Laboratory Test Directory for specific instructions when collecting specimens for cadmium exposure testing (http://ltd.aruplab.com/Tests/Pub/0025013). Urine Collection Tissue Although it is nearly impossible to obtain a completely Follow information on individual pages of the laboratory uncontaminated urine specimen, steps can be taken to test directory. minimize environmental contamination. Avoid collecting the specimen in an area where Whole Blood, Serum, and Red Blood Cell Collection Materials comprising the tube and the tube top are a environmental contamination is likely to occur. In an consideration. Rubber stoppers in collection tubes other industrial or construction setting, it is important that than the royal blue tube are known to contain elements clothing worn in the workplace be removed prior to that can contaminate a specimen. Contamination can be specimen collection to prevent dust on the clothing introduced as the needle punctures the stopper in a tube from contaminating the specimen. other than royal blue. Most of this also holds true for Wash and dry hands thoroughly prior to collection. -

Pdfs/ Ommended That Initial Cultures Focus on Common Pathogens, Pscmanual/9Pscssicurrent.Pdf)
Clinical Infectious Diseases IDSA GUIDELINE A Guide to Utilization of the Microbiology Laboratory for Diagnosis of Infectious Diseases: 2018 Update by the Infectious Diseases Society of America and the American Society for Microbiologya J. Michael Miller,1 Matthew J. Binnicker,2 Sheldon Campbell,3 Karen C. Carroll,4 Kimberle C. Chapin,5 Peter H. Gilligan,6 Mark D. Gonzalez,7 Robert C. Jerris,7 Sue C. Kehl,8 Robin Patel,2 Bobbi S. Pritt,2 Sandra S. Richter,9 Barbara Robinson-Dunn,10 Joseph D. Schwartzman,11 James W. Snyder,12 Sam Telford III,13 Elitza S. Theel,2 Richard B. Thomson Jr,14 Melvin P. Weinstein,15 and Joseph D. Yao2 1Microbiology Technical Services, LLC, Dunwoody, Georgia; 2Division of Clinical Microbiology, Department of Laboratory Medicine and Pathology, Mayo Clinic, Rochester, Minnesota; 3Yale University School of Medicine, New Haven, Connecticut; 4Department of Pathology, Johns Hopkins Medical Institutions, Baltimore, Maryland; 5Department of Pathology, Rhode Island Hospital, Providence; 6Department of Pathology and Laboratory Medicine, University of North Carolina, Chapel Hill; 7Department of Pathology, Children’s Healthcare of Atlanta, Georgia; 8Medical College of Wisconsin, Milwaukee; 9Department of Laboratory Medicine, Cleveland Clinic, Ohio; 10Department of Pathology and Laboratory Medicine, Beaumont Health, Royal Oak, Michigan; 11Dartmouth- Hitchcock Medical Center, Lebanon, New Hampshire; 12Department of Pathology and Laboratory Medicine, University of Louisville, Kentucky; 13Department of Infectious Disease and Global Health, Tufts University, North Grafton, Massachusetts; 14Department of Pathology and Laboratory Medicine, NorthShore University HealthSystem, Evanston, Illinois; and 15Departments of Medicine and Pathology & Laboratory Medicine, Rutgers Robert Wood Johnson Medical School, New Brunswick, New Jersey Contents Introduction and Executive Summary I. -

Rigid Intermediate Bulk Containers: United States
Freedonia Focus Reports US Collection Rigid Intermediate Bulk Containers: United States February 2019 CLICK TO ORDER FULL REPORT BROCHURE www.freedoniafocusreports.com CLICK TO ORDER FULL REPORT Table of Contents 1. Highlights 3 2. Market Environment 4 Historical Trends 4 Key Economic Indicators 5 Competition from Reconditioned & Refurbished Packaging 6 Regulatory Factors 8 3. Segmentation & Forecasts 10 Products 10 Plastic Body 11 Metal Body 12 Other Rigid Intermediate Bulk Containers 13 Markets 14 Chemicals & Pharmaceuticals 15 Food & Beverages 15 Petroleum & Lubricants 16 Hazardous Waste 16 Agricultural & Horticultural Products 16 Plastic & Rubber 17 Other Markets 17 4. Industry Structure 19 Industry Characteristics 19 Market Leaders 20 Mauser Packaging Solutions 20 SCHÜTZ 21 Greif 21 5. About This Report 22 Scope 22 Sources 22 Industry Codes 23 Freedonia Methodology 23 Resources 25 Rigid Intermediate Bulk Containers: United States 1 ©2019 The Freedonia Group. All rights reserved. List of Tables & Figures Figure 1 | Key Trends in the US Rigid Intermediate Bulk Container Market, 2018 – 2023 3 Figure 2 | US Rigid Intermediate Bulk Container Demand Trends, 2008 – 2018 4 Table 1 | Key Indicators for US Rigid Intermediate Bulk Container Demand, 2008 – 2023 (US$ bil) 5 Figure 3 | US Rigid Intermediate Bulk Container Demand by Product, 2008 – 2023 (US$ mil) 10 Table 2 | US Rigid Intermediate Bulk Container Demand by Product, 2008 – 2023 (US$ mil) 10 Figure 4 | US Rigid Intermediate Bulk Container Demand by Product, 2008 – 2023 (%) 12 Figure 5 | US Rigid Intermediate Bulk Container Demand by Market, 2008 – 2023 (US$ mil) 14 Table 3 | US Rigid Intermediate Bulk Container Demand by Market, 2008 – 2023 (US$ mil) 14 Figure 6 | US Rigid Intermediate Bulk Container Demand by Market, 2008 – 2023 (%) 18 Table 4 | Leading Suppliers in the US Rigid Intermediate Bulk Container Market by Product 20 Table 5 | NAICS & SIC Codes Related to Rigid Intermediate Bulk Containers 23 Rigid Intermediate Bulk Containers: United States 2 ©2019 The Freedonia Group. -

Product Brand List November, 2015
Product Brand List November, 2015 BD™ BD Autoguard-N Pro™ BD CARV II™ BD 6CESS™ BD Autoguard™ BD Cato™ 6mm™ (circle design) BD AutoMagic™ BD CatoPAN™ BD A-Cath™ BD AutoNutrient™ BD Chemocato™ BD Accu-Glass™ BD AutoPap™ BD CDT™ BD Accuri™ BD AutoSceptor™ BD Cefinase™ BD Accuspray™ BD AutoShield™ BD CellFIT™ (EU only) BD Acidicase™ BD AutoShield Duo™ BD CellFIX™ (EU only) BD Activ 8™ BD Ayre Cervi-Scraper™ BD Cellmatics™ BD Activation Assist™ BD BACTEC™ BD CellQuest™ Pro BD ACTOne™ BD BACTEC FX™ BD CellWASH™ (EU only) BD Adams™ BD BACTEC™ MGIT™ BD CFlow™ BD Adsyte™ BACTEC Plus Prime™ (Japan) BD Champion™ BD Affirm™ BD Bacto™ BD Chek™ BD AICE™ BD Bact-Plate™ BD CHG™ BD A-Line™ BD Bactrol™ BD CLIC™ BD Amplatz™ BD Bactrol™ Plus BD CLyse™ BD Angiocath Plus/Pro™ (Asia only) BD BaculoGold™ BD CS Quadcount™ BD Angiocath-N™ Autoguard™ BD Barricor™ BD CSampler™ BD Angiocath™ BD BBLCrystal™ BD CX Sampler™ BD Angiocath™ Autoguard™ BD BBL™ CHROMagar (under license) BD Angio-set™ BD BBL™ Crystal™ BD Clay Adams™ BD Apem™ BD BeAware™ BD Cliniject™ BD Asepta-Cell™ BD Bio-Bag™ BD CloneCyt™ BD Asepto™ BD Biosate™ BD CloneCyt™ Plus BD Assurity Linc™ BD BiTek™ BD CMVscan™ BD Attofluor™ BD Bonnano™ BD ColorPAC™ BD Atto™ BD Busher™ BD Connecta™ BD Attovision™ BD Calibrite™ BD Cornwall™ BD Attractors™ BD Calibrite™ 3 BD CPT™ BD Autocrit™ BD CampyPak™ BD CrystalSpec™ BD Autocyte™ BD CampyPak™ Plus BD Crystal™ BD Autocyte Quic™ BD CampyPouch™ BD Autoguard Pro™ BD Campyslide™ Product Brand List November, 2015 cont'd BD CSI™ BD Easy™ BD FACSLyric™ BD CTA Medium™ -

Becton Dickinson 2000 Annual Report
Becton, Dickinson and Company 2000 Annual Report Tools for human health 2000 Annual Report Becton, Dickinson and Company 1 Becton Drive Franklin Lakes, NJ 07417-1880 http://www.bd.com Becton, Dickinson and Company Corporate Information Board of Directors Contents Harry N. Beaty, M.D.1,4 Albert J. Costello1,6 Frank A. Olson2,5,6 James E. Perrella2,3,5 1 Letter to Shareholders Emeritus Dean–Northwestern Retired Chairman of the Board, Chairman of the Board Retired Chairman of the Board– 5 Tools for Human Health University Medical School, and President and Chief Executive and Retired Chief Executive Ingersoll-Rand Company 16 At a Glance Chairman of the Board and Officer–W.R. Grace & Co. Officer– The Hertz Corporation Alfred Sommer1,3 President–Northwestern Gerald M. Edelman, James F. Orr1,4 Dean of the Johns Hopkins 18 Nine-Year Summary University Medical Faculty M.D., Ph.D.4,5,6 Chairman, President and School of Hygiene and Public 20 Financial Review Foundation Director–The Neurosciences Chief Executive Officer– Health, and Professor of 28 Financial Statements Henry P. Becton, Jr.2,3,4 Institute, Member–The Scripps Convergys Corporation Ophthalmology, Epidemiology 49 Corporate Information: President and General Manager– Research Institute Willard J. Overlock, Jr.2,5,6 and International Health 5 1,4 Board of Directors, WGBH Educational Foundation Edward J. Ludwig Retired Partner–Goldman, Margaretha af Ugglas Clateo Castellini3,5 President and Chief Sachs & Co. Member of the Board– Corporate Officers and From laboratory Chairman of the Board–BD Executive Officer–BD Stockholm University and General Information Jarl Hjalmarson Foundation Committees Appointed by the 1 – Audit Committee to living room, Board of Directors 2 – Compensation and Benefits Committee Corporate Officers Edward J. -

Nalgene and Nunc Centrifuge Ware Catalog
Nalgene and Nunc Centrifuge Ware Select the right vessel and spin with confidence Spin with confidence at virtually any scale The process of selecting a centrifuge and rotor can feel like the easy part when faced with choosing the tube or bottle that is the right fit for both the rotor and application. There are several factors to consider when selecting the correct vessel for each application: • Chemical compatibility • Volume • Temperature • Relative centrifugal force (RCF) required • Protocols to be used for loading and sample recovery • Cleaning and autoclaving steps Understanding your requirements before selecting a tube or bottle ensures you make the right choice. Whether your application includes the need for separations, large volume pelleting, protein purification or DNA isolation, the comprehensive selection of Thermo Scientific™ Nalgene™ and Thermo Scientific™ Nunc™ centrifuge ware offers a solution for virtually scales and is available in sizes from 10 mL to 2 L. Such a broad offering means a tube or bottle for many spins – from clinical and bioproduction, to processing bacteria, yeast, tissue, and viruses. Contents Centrifuge tubes 4 Centrifuge bottles 20 Closures and adaptors 31 Resources 36 Simplify performance at every turn with a reliable and safe approach to centrifugation Nalgene Conical-Bottom Centrifuge Tubes polypropylene copolymer Thermo Scientific™ Nalgene™ PPCO conical-bottom centrifuge tubes with molded-in graduations have excellent chemical resistance. Designed for low-speed centrifugation in refrigerated and non-refrigerated centrifuges details • Translucent PPCO is compatible with a wide range of lab reagents • Conical bottoms concentrate pellet in a small area for easy isolation and retrieval • Molded-in graduations last the life of the tube • Last longer than polycarbonate tubes under conditions of repeated Note: Centrifuge tubes must be filled at least 80% for proper performance. -
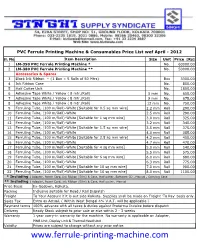
Ferrule Printing Machine & Consumables Price List Wef April - 2012
PVC Ferrule Printing Machine & Consumables Price List wef April - 2012 Sl. No. Item Description Size Unit Price [Rs] 1 LM-390 PVC Ferrule Printing Machine * No. 60000.00 2 LM-380 PVC Ferrule Printing Machine ** No. 50000.00 Accessories & Spares 3 Black Ink Ribbon – {1 Box = 5 Rolls of 50 Mtrs} Box 3300.00 4 Ink Ribbon Case No. 850.00 5 Half Cutter Unit No. 1800.00 6 Adhesive Tape White / Yellow ( 8 mtr./Roll) 5 mm No. 650.00 7 Adhesive Tape White / Yellow ( 8 mtr./Roll) 9 mm No. 675.00 8 Adhesive Tape White / Yellow ( 8 mtr./Roll) 12 mm No. 750.00 9 Ferruling Tube, (100 m/Roll)-White [Suitable for 0.5 sq mm wire] 2.2 mm Roll 290.00 10 Ferruling Tube, (100 m/Roll)-White 2.5 mm Roll 290.00 11 Ferruling Tube, (100 m/Roll)-White [Suitable for 1 sq mm wire] 3.0 mm Roll 325.00 12 Ferruling Tube, (100 m/Roll)-White 3.2 mm Roll 340.00 13 Ferruling Tube, (100 m/Roll)-White [Suitable for 1.5 sq mm wire] 3.5 mm Roll 375.00 14 Ferruling Tube, (100 m/Roll)-White 4.0 mm Roll 385.00 15 Ferruling Tube, (100 m/Roll)-White [Suitable for 2.5 sq mm wire] 4.2 mm Roll 450.00 16 Ferruling Tube, (100 m/Roll)-White 4.7 mm Roll 470.00 17 Ferruling Tube, (100 m/Roll)-White [Suitable for 4 sq mm wire] 5.0 mm Roll 540.00 18 Ferruling Tube, (100 m/Roll)-White 5.5 mm Roll 575.00 19 Ferruling Tube, (100 m/Roll)-White [Suitable for 6 sq mm wire] 6.0 mm Roll 680.00 20 Ferruling Tube, (100 m/Roll)-White 6.3 mm Roll 775.00 21 Ferruling Tube, (100 m/Roll)-White [Suitable for 10 sq mm wire] 7.0 mm Roll 900.00 22 Ferruling Tube, (100 m/Roll)-White [Suitable for 16 sq mm wire] 8.0 mm Roll 1100.00 * Including : Adapter, Power Cord, Ink Ribbon (50m) & Case, Half cutter, Software CD , Manual , Carrying Case ** Including : Adapter, Power Cord, Ink Ribbon (50m) & Case, Half cutter, Manual Price Basis Ex-Godown, Kolkata. -

BD Pyxis™ Inventory Connect Brochure
PIS BD PyxisTM Inventory Connect Safe and efficient inventory management of automated and manual pharmacy stock enabling accurate inventory visibility, compliance and control Medication management faces significant challenges Drug shortages Drug waste 59% 20% of European pharmacists have of hospital inventory is seen care delayed because of discarded2 drug shortages1 Inefficiency Medication errors Over 237 half million of pharmacists’ work time is spent medication errors occur on tasks considered to be in the United Kingdom non-value adding3 each year4 When medication management relies on manual processes, lacks inventory control and involves redundant documentation procedures, it can be time-consuming, inefficient and error-prone3. Some pharmacy information systems (PIS) may have limitations in the way they handle medication inventory in non-automated storage locations adding to these inefficiencies and loss of time. Controlled and efficient inventory management The BD solution for safe, compliant, accurate and integrated inventory management is BD Pyxis™ Inventory Connect. BD Rowa™ Connected and scalable across locations BD Pyxis™ FMD Verify BD Pyxis™ Guided Pick Integrated FMD compliance Improved picking and storing efficiency Actionable analytics - enhanced order management It provides controlled and efficient inventory management, visibility and reporting in both automated and manual stock locations and will integrate to fill the existing gaps of today’s pharmacy information systems. PIS NMVS PIS BD Pyxis™ Inventory Connect BD Pyxis™