Xt GENE PANEL
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Structure and Function of the Human Recq DNA Helicases
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2005 Structure and function of the human RecQ DNA helicases Garcia, P L Posted at the Zurich Open Repository and Archive, University of Zurich ZORA URL: https://doi.org/10.5167/uzh-34420 Dissertation Published Version Originally published at: Garcia, P L. Structure and function of the human RecQ DNA helicases. 2005, University of Zurich, Faculty of Science. Structure and Function of the Human RecQ DNA Helicases Dissertation zur Erlangung der naturwissenschaftlichen Doktorw¨urde (Dr. sc. nat.) vorgelegt der Mathematisch-naturwissenschaftlichen Fakultat¨ der Universitat¨ Z ¨urich von Patrick L. Garcia aus Unterseen BE Promotionskomitee Prof. Dr. Josef Jiricny (Vorsitz) Prof. Dr. Ulrich H ¨ubscher Dr. Pavel Janscak (Leitung der Dissertation) Z ¨urich, 2005 For my parents ii Summary The RecQ DNA helicases are highly conserved from bacteria to man and are required for the maintenance of genomic stability. All unicellular organisms contain a single RecQ helicase, whereas the number of RecQ homologues in higher organisms can vary. Mu- tations in the genes encoding three of the five human members of the RecQ family give rise to autosomal recessive disorders called Bloom syndrome, Werner syndrome and Rothmund-Thomson syndrome. These diseases manifest commonly with genomic in- stability and a high predisposition to cancer. However, the genetic alterations vary as well as the types of tumours in these syndromes. Furthermore, distinct clinical features are observed, like short stature and immunodeficiency in Bloom syndrome patients or premature ageing in Werner Syndrome patients. Also, the biochemical features of the human RecQ-like DNA helicases are diverse, pointing to different roles in the mainte- nance of genomic stability. -

Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse
Welcome to More Choice CD Marker Handbook For more information, please visit: Human bdbiosciences.com/eu/go/humancdmarkers Mouse bdbiosciences.com/eu/go/mousecdmarkers Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse CD3 CD3 CD (cluster of differentiation) molecules are cell surface markers T Cell CD4 CD4 useful for the identification and characterization of leukocytes. The CD CD8 CD8 nomenclature was developed and is maintained through the HLDA (Human Leukocyte Differentiation Antigens) workshop started in 1982. CD45R/B220 CD19 CD19 The goal is to provide standardization of monoclonal antibodies to B Cell CD20 CD22 (B cell activation marker) human antigens across laboratories. To characterize or “workshop” the antibodies, multiple laboratories carry out blind analyses of antibodies. These results independently validate antibody specificity. CD11c CD11c Dendritic Cell CD123 CD123 While the CD nomenclature has been developed for use with human antigens, it is applied to corresponding mouse antigens as well as antigens from other species. However, the mouse and other species NK Cell CD56 CD335 (NKp46) antibodies are not tested by HLDA. Human CD markers were reviewed by the HLDA. New CD markers Stem Cell/ CD34 CD34 were established at the HLDA9 meeting held in Barcelona in 2010. For Precursor hematopoetic stem cell only hematopoetic stem cell only additional information and CD markers please visit www.hcdm.org. Macrophage/ CD14 CD11b/ Mac-1 Monocyte CD33 Ly-71 (F4/80) CD66b Granulocyte CD66b Gr-1/Ly6G Ly6C CD41 CD41 CD61 (Integrin b3) CD61 Platelet CD9 CD62 CD62P (activated platelets) CD235a CD235a Erythrocyte Ter-119 CD146 MECA-32 CD106 CD146 Endothelial Cell CD31 CD62E (activated endothelial cells) Epithelial Cell CD236 CD326 (EPCAM1) For Research Use Only. -

Hypoxia and Oxygen-Sensing Signaling in Gene Regulation and Cancer Progression
International Journal of Molecular Sciences Review Hypoxia and Oxygen-Sensing Signaling in Gene Regulation and Cancer Progression Guang Yang, Rachel Shi and Qing Zhang * Department of Pathology, University of Texas Southwestern Medical Center, Dallas, TX 75390, USA; [email protected] (G.Y.); [email protected] (R.S.) * Correspondence: [email protected]; Tel.: +1-214-645-4671 Received: 6 October 2020; Accepted: 29 October 2020; Published: 31 October 2020 Abstract: Oxygen homeostasis regulation is the most fundamental cellular process for adjusting physiological oxygen variations, and its irregularity leads to various human diseases, including cancer. Hypoxia is closely associated with cancer development, and hypoxia/oxygen-sensing signaling plays critical roles in the modulation of cancer progression. The key molecules of the hypoxia/oxygen-sensing signaling include the transcriptional regulator hypoxia-inducible factor (HIF) which widely controls oxygen responsive genes, the central members of the 2-oxoglutarate (2-OG)-dependent dioxygenases, such as prolyl hydroxylase (PHD or EglN), and an E3 ubiquitin ligase component for HIF degeneration called von Hippel–Lindau (encoding protein pVHL). In this review, we summarize the current knowledge about the canonical hypoxia signaling, HIF transcription factors, and pVHL. In addition, the role of 2-OG-dependent enzymes, such as DNA/RNA-modifying enzymes, JmjC domain-containing enzymes, and prolyl hydroxylases, in gene regulation of cancer progression, is specifically reviewed. We also discuss the therapeutic advancement of targeting hypoxia and oxygen sensing pathways in cancer. Keywords: hypoxia; PHDs; TETs; JmjCs; HIFs 1. Introduction Molecular oxygen serves as a co-factor in many biochemical processes and is fundamental for aerobic organisms to maintain intracellular ATP levels [1,2]. -

A CD22-Shp1 Phosphatase Axis Controls Integrin Β7 Display and B Cell Function in Mucosal Immunity
UCSF UC San Francisco Previously Published Works Title A CD22-Shp1 phosphatase axis controls integrin β7 display and B cell function in mucosal immunity. Permalink https://escholarship.org/uc/item/27j4g9rr Journal Nature immunology, 22(3) ISSN 1529-2908 Authors Ballet, Romain Brennan, Martin Brandl, Carolin et al. Publication Date 2021-03-01 DOI 10.1038/s41590-021-00862-z Peer reviewed eScholarship.org Powered by the California Digital Library University of California Europe PMC Funders Group Author Manuscript Nat Immunol. Author manuscript; available in PMC 2021 August 15. Published in final edited form as: Nat Immunol. 2021 March 01; 22(3): 381–390. doi:10.1038/s41590-021-00862-z. Europe PMC Funders Author Manuscripts A CD22-Shp1 phosphatase axis controls integrin β7 display and B cell function in mucosal immunity Romain Ballet1,2,#, Martin Brennan1,2,10, Carolin Brandl3,10, Ningguo Feng1,4, Jeremy Berri1,2, Julian Cheng1,2, Borja Ocón1,2, Amin Alborzian Deh Sheikh5, Alex Marki6, Yuhan Bi1,2, Clare L. Abram7, Clifford A. Lowell7, Takeshi Tsubata5, Harry B. Greenberg1,4, Matthew S. Macauley8,9, Klaus Ley6, Lars Nitschke3, Eugene C. Butcher1,2,# 1The Center for Molecular Biology and Medicine, Veterans Affairs Palo Alto Health Care System and The Palo Alto Veterans Institute for Research, Palo Alto, CA, United States 2Laboratory of Immunology and Vascular Biology, Department of Pathology, School of Medicine, Stanford University, Stanford, CA, United States 3Division of Genetics, Department of Biology, University of Erlangen-Nürnberg, Erlangen, -
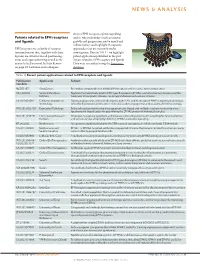
Patents Related to EPH Receptors and Ligands
NEWS & ANALYSIS discuss EPH receptor–ephrin signalling Patents related to EPH receptors and its role in disorders such as tumour and ligands growth and progression, nerve injury and inflammation, and highlight therapeutic EPH receptors are a family of receptor approaches that are currently under tyrosine kinases that, together with their investigation. Here in TABLE 1 we highlight ligands, are involved in cell positioning, patent applications published in the past tissue and organ patterning as well as the 3 years related to EPH receptors and ligands. control of cell survival. In their Review Data were researched using the Espacenet on page 39, Lackman and colleagues database. Table 1 | Recent patent applications related to EPH receptors and ligands Nature Reviews | Drug Discovery Publication Applicants Subject numbers NZ 581397 AstraZeneca Pyrimidine compounds that inhibit EPH receptors and are useful for treating cancer HK 1108702 Sanford-Burnham Peptides that selectively bind to EPH type-B receptors (EPHBs); useful for tumour imaging and the Institute treatment of neoplastic disease, neurological disease and vascular disease US 2013091591 California Institute of During angiogenesis, arterial cells express ephrin B2, and its receptor EPHB4 is expressed on venous Technology cells; this distinction can be used in methods to alter angiogenesis and to assess the effect of drugs WO 2013052710 Expression Pathology Selected reaction monitoring mass spectrometry-based and multiple reaction monitoring mass spectrometry-based assays for quantifying -

Open Full Page
CCR PEDIATRIC ONCOLOGY SERIES CCR Pediatric Oncology Series Recommendations for Childhood Cancer Screening and Surveillance in DNA Repair Disorders Michael F. Walsh1, Vivian Y. Chang2, Wendy K. Kohlmann3, Hamish S. Scott4, Christopher Cunniff5, Franck Bourdeaut6, Jan J. Molenaar7, Christopher C. Porter8, John T. Sandlund9, Sharon E. Plon10, Lisa L. Wang10, and Sharon A. Savage11 Abstract DNA repair syndromes are heterogeneous disorders caused by around the world to discuss and develop cancer surveillance pathogenic variants in genes encoding proteins key in DNA guidelines for children with cancer-prone disorders. Herein, replication and/or the cellular response to DNA damage. The we focus on the more common of the rare DNA repair dis- majority of these syndromes are inherited in an autosomal- orders: ataxia telangiectasia, Bloom syndrome, Fanconi ane- recessive manner, but autosomal-dominant and X-linked reces- mia, dyskeratosis congenita, Nijmegen breakage syndrome, sive disorders also exist. The clinical features of patients with DNA Rothmund–Thomson syndrome, and Xeroderma pigmento- repair syndromes are highly varied and dependent on the under- sum. Dedicated syndrome registries and a combination of lying genetic cause. Notably, all patients have elevated risks of basic science and clinical research have led to important in- syndrome-associated cancers, and many of these cancers present sights into the underlying biology of these disorders. Given the in childhood. Although it is clear that the risk of cancer is rarity of these disorders, it is recommended that centralized increased, there are limited data defining the true incidence of centers of excellence be involved directly or through consulta- cancer and almost no evidence-based approaches to cancer tion in caring for patients with heritable DNA repair syn- surveillance in patients with DNA repair disorders. -

Mutant IDH, (R)-2-Hydroxyglutarate, and Cancer
Downloaded from genesdev.cshlp.org on October 1, 2021 - Published by Cold Spring Harbor Laboratory Press REVIEW What a difference a hydroxyl makes: mutant IDH, (R)-2-hydroxyglutarate, and cancer Julie-Aurore Losman1 and William G. Kaelin Jr.1,2,3 1Department of Medical Oncology, Dana-Farber Cancer Institute, Brigham and Women’s Hospital, Harvard Medical School, Boston, Massachusetts 02215, USA; 2Howard Hughes Medical Institute, Chevy Chase, Maryland 20815, USA Mutations in metabolic enzymes, including isocitrate whether altered cellular metabolism is a cause of cancer dehydrogenase 1 (IDH1) and IDH2, in cancer strongly or merely an adaptive response of cancer cells in the face implicate altered metabolism in tumorigenesis. IDH1 of accelerated cell proliferation is still a topic of some and IDH2 catalyze the interconversion of isocitrate and debate. 2-oxoglutarate (2OG). 2OG is a TCA cycle intermediate The recent identification of cancer-associated muta- and an essential cofactor for many enzymes, including tions in three metabolic enzymes suggests that altered JmjC domain-containing histone demethylases, TET cellular metabolism can indeed be a cause of some 5-methylcytosine hydroxylases, and EglN prolyl-4-hydrox- cancers (Pollard et al. 2003; King et al. 2006; Raimundo ylases. Cancer-associated IDH mutations alter the enzymes et al. 2011). Two of these enzymes, fumarate hydratase such that they reduce 2OG to the structurally similar (FH) and succinate dehydrogenase (SDH), are bone fide metabolite (R)-2-hydroxyglutarate [(R)-2HG]. Here we tumor suppressors, and loss-of-function mutations in FH review what is known about the molecular mechanisms and SDH have been identified in various cancers, in- of transformation by mutant IDH and discuss their im- cluding renal cell carcinomas and paragangliomas. -

A Review of VEGF/VEGFR-Targeted Therapeutics for Recurrent Glioblastoma
414 Original Article A Review of VEGF/VEGFR-Targeted Therapeutics for Recurrent Glioblastoma David A. Reardon, MDa,b; Scott Turner, MDc; Katherine B. Peters, MD, PhDc; Annick Desjardins, MDc; Sridharan Gururangan, MDa,b; John H. Sampson, MD, PhDa; Roger E. McLendon, MDd; James E. Herndon II, PhDe; Lee W. Jones, PhDf; John P. Kirkpatrick, MD, PhDf; Allan H. Friedman, MDa; James J. Vredenburgh, MDc; Darell D. Bigner, MD, PhDd; and Henry S. Friedman, MDa,b; Durham, North Carolina Key Words ability effect of VEGF inhibitors, the Radiologic Assessment in Neu- Glioblastoma, angiogenesis, vascular endothelial growth factor, ro-Oncology (RANO) criteria were recently implemented to bet- malignant glioma ter assess response among patients with glioblastoma. Although bevacizumab improves survival and quality of life, eventual tumor progression is the norm. Better understanding of resistance mech- Abstract anisms to VEGF inhibitors and identification of effective therapy Glioblastoma, the most common primary malignant brain tumor after bevacizumab progression are currently a critical need for pa- among adults, is a highly angiogenic and deadly tumor. Angiogen- tients with glioblastoma. (JNCCN 2011;9:414–427) esis in glioblastoma, driven by hypoxia-dependent and indepen- dent mechanisms, is primarily mediated by vascular endothelial growth factor (VEGF), and generates blood vessels with distinctive features. The outcome for patients with recurrent glioblastoma is poor because of ineffective therapies. However, recent encourag- ing rates of radiographic response and progression-free survival, Malignant gliomas, including the most common sub- and adequate safety, led the FDA to grant accelerated approval of type of glioblastoma, are rapidly growing destructive tu- bevacizumab, a humanized monoclonal antibody against VEGF, for mors that extensively invade locally but rarely metasta- the treatment of recurrent glioblastoma in May 2009. -

FANCL Sirna (H): Sc-45661
SAN TA C RUZ BI OTEC HNOL OG Y, INC . FANCL siRNA (h): sc-45661 BACKGROUND STORAGE AND RESUSPENSION Defects in FANCL are a cause of Fanconi anemia. Fanconi anemia (FA) is an Store lyophilized siRNA duplex at -20° C with desiccant. Stable for at least autosomal recessive disorder characterized by bone marrow failure, birth one year from the date of shipment. Once resuspended, store at -20° C, defects and chromosomal instability. At the cellular level, FA is characterized avoid contact with RNAses and repeated freeze thaw cycles. by spontaneous chromosomal breakage and a unique hypersensitivity to DNA Resuspend lyophilized siRNA duplex in 330 µl of the RNAse-free water cross-linking agents. At least 8 complementation groups have been identified pro vided. Resuspension of the siRNA duplex in 330 µl of RNAse-free water and 6 FA genes (for subtypes A, C, D2, E, F and G) have been cloned. Phospho- makes a 10 µM solution in a 10 µM Tris-HCl, pH 8.0, 20 mM NaCl, 1 mM rylation of FANC (Fanconi anemia complementation group) proteins is thought EDTA buffered solution. to be important for the function of the FA pathway. FA proteins cooperate in a common pathway, culminating in the monoubiquitination of FANCD2 protein APPLICATIONS and colocalization of FANCD2 and BRCA1 proteins in nuclear foci. FANCL is a ligase protein mediating the ubiquitination of FANCD2, a key step in the FANCL shRNA (h) Lentiviral Particles is recommended for the inhibition of DNA damage pathway. FANCL may be required for proper primordial germ cell FANCL expression in human cells. -

Further Insights Into the Regulation of the Fanconi Anemia FANCD2 Protein
University of Rhode Island DigitalCommons@URI Open Access Dissertations 2015 Further Insights Into the Regulation of the Fanconi Anemia FANCD2 Protein Rebecca Anne Boisvert University of Rhode Island, [email protected] Follow this and additional works at: https://digitalcommons.uri.edu/oa_diss Recommended Citation Boisvert, Rebecca Anne, "Further Insights Into the Regulation of the Fanconi Anemia FANCD2 Protein" (2015). Open Access Dissertations. Paper 397. https://digitalcommons.uri.edu/oa_diss/397 This Dissertation is brought to you for free and open access by DigitalCommons@URI. It has been accepted for inclusion in Open Access Dissertations by an authorized administrator of DigitalCommons@URI. For more information, please contact [email protected]. FURTHER INSIGHTS INTO THE REGULATION OF THE FANCONI ANEMIA FANCD2 PROTEIN BY REBECCA ANNE BOISVERT A DISSERTATION SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY IN CELL AND MOLECULAR BIOLOGY UNIVERSITY OF RHODE ISLAND 2015 DOCTOR OF PHILOSOPHY DISSERTATION OF REBECCA ANNE BOISVERT APPROVED: Dissertation Committee: Major Professor Niall Howlett Paul Cohen Becky Sartini Nasser H. Zawia DEAN OF THE GRADUATE SCHOOL UNIVERSITY OF RHODE ISLAND 2015 ABSTRACT Fanconi anemia (FA) is a rare autosomal and X-linked recessive disorder, characterized by congenital abnormalities, pediatric bone marrow failure and cancer susceptibility. FA is caused by biallelic mutations in any one of 16 genes. The FA proteins function cooperatively in the FA-BRCA pathway to repair DNA interstrand crosslinks (ICLs). The monoubiquitination of FANCD2 and FANCI is a central step in the activation of the FA-BRCA pathway and is required for targeting these proteins to chromatin. -

Wnt Signalling in Melanoma and Ageing
MINIREVIEW British Journal of Cancer (2016) 115, 1273–1279 | doi: 10.1038/bjc.2016.332 Keywords: Wnt; metastasis; melanoma; ageing; sFRP2; Wnt5a In the Wnt-er of life: Wnt signalling in melanoma and ageing Amanpreet Kaur1,2, Marie R Webster1 and Ashani T Weeraratna*,1 1Tumor Microenvironment and Metastasis Program, The Wistar Institute, Philadelphia, PA, USA and 2University of the Sciences, Philadelphia, PA, USA Although the clinical landscape of melanoma is improving rapidly, metastatic melanoma remains a deadly disease. Age remains one of the greatest risk factors for melanoma, and patients older than 55 have a much poorer prognosis than younger individuals, even when the data are controlled for grade and stage. The reasons for this disparity have not been fully uncovered, but there is some recent evidence that Wnt signalling may have a role. Wnt signalling is known to have roles both in cancer progression as well as in organismal ageing. In melanoma, the interplay of Wnt signalling pathways is complex, with different members of the Wnt family guiding different aspects of invasion and proliferation. Here, we will briefly review the current literature addressing the roles of different Wnt pathways in melanoma pathogenesis, provide an overview of Wnt signalling during ageing, and discuss the intersection between melanoma and ageing in terms of Wnt signalling. AGE IS A PROGNOSTIC FACTOR FOR MELANOMA also contribute to the age-induced progression of cancer. For example, it has long been proposed that the ageing stroma As human lifespan increases, there is a growing concern over the contributes to cancer progression, based on the studies using availability of treatments to manage the increasing incidence of senescence as an artificial model of ageing (Campisi, 2013; Campisi cancer in aged individuals. -

FANCJ Regulates the Stability of FANCD2/FANCI Proteins and Protects Them from Proteasome and Caspase-3 Dependent Degradation
FANCJ regulates the stability of FANCD2/FANCI proteins and protects them from proteasome and caspase-3 dependent degradation Komaraiah Palle, Ph.D. (Kumar) Assistant Professor of Oncologic Sciences Abraham Mitchell Cancer Research Scholar Mitchell Cancer Institute University of South Alabama Outline • Fanconi anemia (FA) pathway • Role of FA pathway in Genome maintenance • FANCJ and FANCD2 functional relationship • FANCJ-mediated DDR in response to Fork-stalling Fanconi Anemia • Rare, inherited blood disorder. • 1:130,000 births Guido Fanconi 1892-1979 • Affects men and women equally. • Affects all racial and ethnic groups – higher incidence in Ashkenazi Jews and Afrikaners Birth Defects Fanconi anemia pathway • FA is a rare chromosome instability syndrome • Autosomal recessive disorder (or X-linked) • Developmental abnormalities • 17 complementation groups identified to date • FA pathway is involved in DNA repair • Increased cancer susceptibility - many patients develop AML - in adults solid tumors Fanconi Anemia is an aplastic anemia FA patients are prone to multiple types of solid tumors • Increased incidence and earlier onset cancers: oral cavity, GI and genital and reproductive tract head and neck breast esophagus skin liver brain Why? FA is a DNA repair disorder • FA caused by mutations in 17 genes: FANCA FANCF FANCM FANCB FANCG/XRCC9 FANCN/PALB2 FANCC FANCI RAD51C/FANCO FANCD1/BRCA2 FANCJ SLX4/FANCP FANCD2 FANCL ERCC2/XPF/FANCQ FANCE BRCA1/FANCS • FA genes function in DNA repair processes • FA patient cells are highly sensitive