Biodiversity Defrosted : Unveiling Non-Compliant Fish Trade In
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Updated Checklist of Marine Fishes (Chordata: Craniata) from Portugal and the Proposed Extension of the Portuguese Continental Shelf
European Journal of Taxonomy 73: 1-73 ISSN 2118-9773 http://dx.doi.org/10.5852/ejt.2014.73 www.europeanjournaloftaxonomy.eu 2014 · Carneiro M. et al. This work is licensed under a Creative Commons Attribution 3.0 License. Monograph urn:lsid:zoobank.org:pub:9A5F217D-8E7B-448A-9CAB-2CCC9CC6F857 Updated checklist of marine fishes (Chordata: Craniata) from Portugal and the proposed extension of the Portuguese continental shelf Miguel CARNEIRO1,5, Rogélia MARTINS2,6, Monica LANDI*,3,7 & Filipe O. COSTA4,8 1,2 DIV-RP (Modelling and Management Fishery Resources Division), Instituto Português do Mar e da Atmosfera, Av. Brasilia 1449-006 Lisboa, Portugal. E-mail: [email protected], [email protected] 3,4 CBMA (Centre of Molecular and Environmental Biology), Department of Biology, University of Minho, Campus de Gualtar, 4710-057 Braga, Portugal. E-mail: [email protected], [email protected] * corresponding author: [email protected] 5 urn:lsid:zoobank.org:author:90A98A50-327E-4648-9DCE-75709C7A2472 6 urn:lsid:zoobank.org:author:1EB6DE00-9E91-407C-B7C4-34F31F29FD88 7 urn:lsid:zoobank.org:author:6D3AC760-77F2-4CFA-B5C7-665CB07F4CEB 8 urn:lsid:zoobank.org:author:48E53CF3-71C8-403C-BECD-10B20B3C15B4 Abstract. The study of the Portuguese marine ichthyofauna has a long historical tradition, rooted back in the 18th Century. Here we present an annotated checklist of the marine fishes from Portuguese waters, including the area encompassed by the proposed extension of the Portuguese continental shelf and the Economic Exclusive Zone (EEZ). The list is based on historical literature records and taxon occurrence data obtained from natural history collections, together with new revisions and occurrences. -

Fishmonger Practice Display and Merchandising
Fishmonger Practice DISPLAY AND MERCHANDISING Draft Materials This is a typescript from the 1989 Training Manual developed by Seafish. The manual will be updated later in 2018, and until then this typescript will be made available to potential users. The contents of this file remain the intellectual property of the Sae Fish Industry Authority. General Objective: On completion of this training programme trainees will be able to apply basic display and merchandising principles in order to create effective displays of fish and fish products. Session Outline Session Title Time Indicator 1. Scope and purpose of display 1.0 hour 2. Display communication 1.0 hour 3. Product display properties 2.0 hours 4. Display equipment and accessories 3.5 hours 5. Product arrangement 5.0 hours 6. Display maintenance 1.5 hours Total Time Indicator 14.0 hours Contents Page TRAINER’S GUIDE Benefits of systematic training 1 Guide to the manual 2 How to design a training session 7 Setting objectives 9 Use of questions in training 10 Correction coaching 12 SESSION OUTLINES 1. Scope and purpose of display 14 Information sheets 23 2. Display communication 41 Information sheets 49 3. Product display properties 68 Information sheets 87 4. Display equipment and accessories 117 Information sheets 186 5. Product arrangement 224 Information sheets 255 6. Display maintenance 304 Information sheets 331 VISUAL AIDS ADDITIONAL TRAINING RESOURCES 338 Benefits of systematic training This instructor’s manual has been designed to assist the on-the-job training of staff employed in fish retail establishments. Below are listed some of the benefits which can be obtained by following a programme of systematic training. -

Fish Bulletin 161. California Marine Fish Landings for 1972 and Designated Common Names of Certain Marine Organisms of California
UC San Diego Fish Bulletin Title Fish Bulletin 161. California Marine Fish Landings For 1972 and Designated Common Names of Certain Marine Organisms of California Permalink https://escholarship.org/uc/item/93g734v0 Authors Pinkas, Leo Gates, Doyle E Frey, Herbert W Publication Date 1974 eScholarship.org Powered by the California Digital Library University of California STATE OF CALIFORNIA THE RESOURCES AGENCY OF CALIFORNIA DEPARTMENT OF FISH AND GAME FISH BULLETIN 161 California Marine Fish Landings For 1972 and Designated Common Names of Certain Marine Organisms of California By Leo Pinkas Marine Resources Region and By Doyle E. Gates and Herbert W. Frey > Marine Resources Region 1974 1 Figure 1. Geographical areas used to summarize California Fisheries statistics. 2 3 1. CALIFORNIA MARINE FISH LANDINGS FOR 1972 LEO PINKAS Marine Resources Region 1.1. INTRODUCTION The protection, propagation, and wise utilization of California's living marine resources (established as common property by statute, Section 1600, Fish and Game Code) is dependent upon the welding of biological, environment- al, economic, and sociological factors. Fundamental to each of these factors, as well as the entire management pro- cess, are harvest records. The California Department of Fish and Game began gathering commercial fisheries land- ing data in 1916. Commercial fish catches were first published in 1929 for the years 1926 and 1927. This report, the 32nd in the landing series, is for the calendar year 1972. It summarizes commercial fishing activities in marine as well as fresh waters and includes the catches of the sportfishing partyboat fleet. Preliminary landing data are published annually in the circular series which also enumerates certain fishery products produced from the catch. -

Hierarchical Fish Species Detection in Real-Time Video Using YOLO
Hierarchical Fish Species Detection in Real-Time Video Using YOLO ESPEN STAUSLAND KALHAGEN, ØRJAN LANGØY OLSEN SUPERVISOR Morten Goodwin University of Agder, 2020 Faculty of Engineering and Science Department of Information and Communication Technology Abstract Information gathering of aquatic life is often based on time consuming methods with a foundation in video feeds. It would be beneficial to capture more information in a cost effective manner from video feeds, and video object detection has an opportunity to achieve this. Recent research has shown promising results with the use of YOLO for object detection of fish. As under-water conditions can be difficult and fish species hard to discrimi- nate, we propose the use of a hierarchical structures in both the classification and the dataset to gain valuable information. With the use of hierarchical classification and other techniques we present YOLO Fish. YOLO Fish is a state of the art object detector on nordic fish species, with an mAP of 91.8%. For a more stable video, YOLO Fish can be used with the object tracking algorithm SORT. This results in a complete fish detector for real-time video. 1 Table of Contents 1 Introduction 5 1.1 Problem Statement . .5 1.2 Contributions . .6 2 Background 7 2.1 Convolutional Neural Networks . .7 2.2 You Only Look Once (YOLO) . .8 2.2.1 The Architecture . .9 2.2.2 Activations and Weight Initialization . 10 2.2.3 Prediction and Priors . 11 2.2.4 Loss Function . 14 2.3 Hierarchical Classification in YOLO . 15 2.4 NMS and Soft-NMS . -

First Record of Pagellus Bellottii (Teleostei: Sparidae) in the Bay of Biscay, France Samuel P
Iglésias and Lorance Marine Biodiversity Records (2016) 9:16 DOI 10.1186/s41200-016-0007-8 MARINE RECORD Open Access First record of Pagellus bellottii (Teleostei: Sparidae) in the Bay of Biscay, France Samuel P. Iglésias1* and Pascal Lorance2 Abstract The red pandora, Pagellus bellottii, is a tropical and subtropical demersal porgy (Sparidae) distributed in the eastern Atlantic, mostly along African coast. Several recent records in the Gulf of Cádiz suggests a northward migration of the species. Two specimens of the red pandora were caught in the southeast Bay of Biscay during the 2014 French fishery survey programme. They represent the first records of the species for the Bay of Biscay and for French waters. In Europe the species has been previously recorded only in the Gulf of Cádiz in the Atlantic and in Spanish Mediterranean waters. The hypothesis that global climate change is a factor for this almost 7° northward species range extension, commonly distributed in the tropical and subtropical eastern Atlantic along the African coast, is analysed using temperature data from the World Ocean Atlas 2013. The northern extension of the distribution range of Pagellus bellottii most probably is a consequence of the warming waters of the Bay of Biscay over the past decades. Keywords: Red pandora, Porgies, Range extension, Immigrant, Tropicalisation, Northeast Atlantic Background com. pers.). Some specimens were also recorded in Span- The red pandora, Pagellus bellottii Steindachner, 1882, is ish Mediterranean waters, in the Bay of Almería about 36° a tropical and subtropical demersal Sparidae (Porgies) 47′ N, 2°25′ W (Lucena, et al. 1982), the northernmost re- that lives over hard or sandy bottoms in coastal marine cords for the Mediterranean waters. -

Alien Species in the Mediterranean Sea by 2010
Mediterranean Marine Science Review Article Indexed in WoS (Web of Science, ISI Thomson) The journal is available on line at http://www.medit-mar-sc.net Alien species in the Mediterranean Sea by 2010. A contribution to the application of European Union’s Marine Strategy Framework Directive (MSFD). Part I. Spatial distribution A. ZENETOS 1, S. GOFAS 2, M. VERLAQUE 3, M.E. INAR 4, J.E. GARCI’A RASO 5, C.N. BIANCHI 6, C. MORRI 6, E. AZZURRO 7, M. BILECENOGLU 8, C. FROGLIA 9, I. SIOKOU 10 , D. VIOLANTI 11 , A. SFRISO 12 , G. SAN MART N 13 , A. GIANGRANDE 14 , T. KATA AN 4, E. BALLESTEROS 15 , A. RAMOS-ESPLA ’16 , F. MASTROTOTARO 17 , O. OCA A 18 , A. ZINGONE 19 , M.C. GAMBI 19 and N. STREFTARIS 10 1 Institute of Marine Biological Resources, Hellenic Centre for Marine Research, P.O. Box 712, 19013 Anavissos, Hellas 2 Departamento de Biologia Animal, Facultad de Ciencias, Universidad de Ma ’laga, E-29071 Ma ’laga, Spain 3 UMR 6540, DIMAR, COM, CNRS, Université de la Méditerranée, France 4 Ege University, Faculty of Fisheries, Department of Hydrobiology, 35100 Bornova, Izmir, Turkey 5 Departamento de Biologia Animal, Facultad de Ciencias, Universidad de Ma ’laga, E-29071 Ma ’laga, Spain 6 DipTeRis (Dipartimento per lo studio del Territorio e della sue Risorse), University of Genoa, Corso Europa 26, 16132 Genova, Italy 7 Institut de Ciències del Mar (CSIC) Passeig Mar tim de la Barceloneta, 37-49, E-08003 Barcelona, Spain 8 Adnan Menderes University, Faculty of Arts & Sciences, Department of Biology, 09010 Aydin, Turkey 9 c\o CNR-ISMAR, Sede Ancona, Largo Fiera della Pesca, 60125 Ancona, Italy 10 Institute of Oceanography, Hellenic Centre for Marine Research, P.O. -

A Molecular Phylogeny of the Sparidae (Perciformes: Percoidei)
W&M ScholarWorks Dissertations, Theses, and Masters Projects Theses, Dissertations, & Master Projects 2000 A molecular phylogeny of the Sparidae (Perciformes: Percoidei) Thomas M. Orrell College of William and Mary - Virginia Institute of Marine Science Follow this and additional works at: https://scholarworks.wm.edu/etd Part of the Genetics Commons, and the Zoology Commons Recommended Citation Orrell, Thomas M., "A molecular phylogeny of the Sparidae (Perciformes: Percoidei)" (2000). Dissertations, Theses, and Masters Projects. Paper 1539616799. https://dx.doi.org/doi:10.25773/v5-x8gj-1114 This Dissertation is brought to you for free and open access by the Theses, Dissertations, & Master Projects at W&M ScholarWorks. It has been accepted for inclusion in Dissertations, Theses, and Masters Projects by an authorized administrator of W&M ScholarWorks. For more information, please contact [email protected]. INFORMATION TO USERS This manuscript has been reproduced from the microfilm master. UMI films the text directly from (he original or copy submitted. Thus, some thesis and dissertation copies are in typewriter face, while others may be from any type of computer printer. The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bieedthrough, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send UMI a complete manuscript and there are missing pages, these will be noted. Also, if unauthorized copyright material had to be removed, a note will indicate the deletion. Oversize materials (e.g., maps, drawings, charts) are reproduced by sectioning the original, beginning at the upper left-hand comer and continuing from left to right in equal sections with small overlaps. -
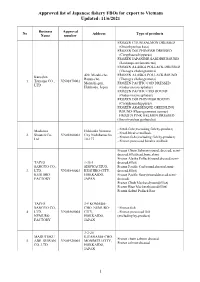
Approved List of Japanese Fishery Fbos for Export to Vietnam Updated: 11/6/2021
Approved list of Japanese fishery FBOs for export to Vietnam Updated: 11/6/2021 Business Approval No Address Type of products Name number FROZEN CHUM SALMON DRESSED (Oncorhynchus keta) FROZEN DOLPHINFISH DRESSED (Coryphaena hippurus) FROZEN JAPANESE SARDINE ROUND (Sardinops melanostictus) FROZEN ALASKA POLLACK DRESSED (Theragra chalcogramma) 420, Misaki-cho, FROZEN ALASKA POLLACK ROUND Kaneshin Rausu-cho, (Theragra chalcogramma) 1. Tsuyama CO., VN01870001 Menashi-gun, FROZEN PACIFIC COD DRESSED LTD Hokkaido, Japan (Gadus macrocephalus) FROZEN PACIFIC COD ROUND (Gadus macrocephalus) FROZEN DOLPHIN FISH ROUND (Coryphaena hippurus) FROZEN ARABESQUE GREENLING ROUND (Pleurogrammus azonus) FROZEN PINK SALMON DRESSED (Oncorhynchus gorbuscha) - Fresh fish (excluding fish by-product) Maekawa Hokkaido Nemuro - Fresh bivalve mollusk. 2. Shouten Co., VN01860002 City Nishihamacho - Frozen fish (excluding fish by-product) Ltd 10-177 - Frozen processed bivalve mollusk Frozen Chum Salmon (round, dressed, semi- dressed,fillet,head,bone,skin) Frozen Alaska Pollack(round,dressed,semi- TAIYO 1-35-1 dressed,fillet) SANGYO CO., SHOWACHUO, Frozen Pacific Cod(round,dressed,semi- 3. LTD. VN01840003 KUSHIRO-CITY, dressed,fillet) KUSHIRO HOKKAIDO, Frozen Pacific Saury(round,dressed,semi- FACTORY JAPAN dressed) Frozen Chub Mackerel(round,fillet) Frozen Blue Mackerel(round,fillet) Frozen Salted Pollack Roe TAIYO 3-9 KOMABA- SANGYO CO., CHO, NEMURO- - Frozen fish 4. LTD. VN01860004 CITY, - Frozen processed fish NEMURO HOKKAIDO, (excluding by-product) FACTORY JAPAN -

Distribution and Habits of Marine Fish and Invertebrates in Katlian Bay
Alaska Fisheries Science Center National Marine Fisheries Service U.S DEPARTMENT OF COMMERCE AFSC PROCESSED REPORT 2006-04 Distribution and Habitats of Marine Fish and Invertebrates in Katlian Bay, Southeastern Alaska, 1967 and 1968 February 2006 This report does not constitute a publication and is for information only. All data herein are to be considered provisional. DISTRIBUTION AND HABITATS OF MARINE FISH AND INVERTEBRATES IN KATLIAN BAY, SOUTHEASTERN ALASKA, 1967 AND 1968. By Richard E. Haight, Gerald M. Reid, and Noele Weemes Auke Bay Laboratory Alaska Fisheries Science Center National Marine Fisheries Service National Oceanic and Atmospheric Administration 11305 Glacier Highway Juneau, Alaska 99801-8626 February 2006 iii ABSTRACT In 1967 and 1968, scientists from the National Marine Fisheries Service’s Auke Bay Laboratory carried out four surveys of marine fauna in Katlian Bay, near Sitka, Alaska as part of an impact study associated with plans to build a wood pulp processing plant in the bay. Here we report the results of our surveys and also provide a broad literature review on several of the species that were captured in the bay. Fifty-nine fish species and more than 44 invertebrate species (32 identified to species level (see page 8) were captured. Habitats examined were intertidal, the steep sides of the bay, and the bay’s deep central basin. Many species occupied a single habitat type but others overlapped into adjacent habitats. Five fish species were collected in the intertidal zone and major invertebrate fauna included the sunflower star (Pycnopodia helianthoides) and numerous members of Gastropoda. Thirty-nine fish and 23 invertebrate species were collected on the bay’s sides below 5 m. -

Electronic Supplementary Material
Electronic supplementary material Trenkel et al. Portfolio composition not width reduces annual variability of fisheries economic returns and energy ratios. Table S1. Functional group membership and energetic content. NA not available. The "scaling method" consists of scaling the energy content E (kilo Calories) of a fillet found in Dorosz (1992) to whole fish live weight (kilo Joules) by multiplying with 6.7*E. English name Latin name Functional group Energy (kJ Data source/Method per kg live weight) Small sandeel Ammodytes tobianus Benthivore 5800 Spitz et al 2010 Imperial scaldfish Arnoglossus Benthivore 5400 Spitz et al 2010 imperialis Red gurnard Aspitrigla cuculus Benthivore 8200 Spitz et al 2010 Triggerfishes, Balistidae Benthivore 6900 Spondyliosoma cantharus durgons nei Spitz et al 2010 Bogue Boops boops Benthivore 8000 Spitz et al 2010 Dragonet Callionymus lyra Benthivore 5200 Spitz et al 2010 Boarfish Capros aper Benthivore 6200 Spitz et al 2010 Red band fish Cepola Benthivore 3900 Spitz et al 2010 macrophthalma Streaked gurnard Chelidonichthys Benthivore 8200 Chelidonichthys cuculus lastoviza Spitz et al 2010 Longfin gurnard Chelidonichthys Benthivore 8200 Chelidonichthys cuculus obscurus Spitz et al 2010 Wedge sole Dicologlossa cuneata Benthivore 6500 Spitz et al 2010 Grey gurnard Eutrigla gurnardus Benthivore 8200 Chelidonichthys cuculus Spitz et al 2010 Silvery pout Gadiculus argenteus Benthivore 5000 Spitz et al 2010 Witch flounder Glyptocephalus Benthivore 5600 Spitz et al 2010 cynoglossus Blackbelly rose fish Helicolenus Benthivore 9200 Spitz et al 2010 dactylopterus Amer. plaice(=Long Hippoglossoides Benthivore 6100 Lepidorhombus whiffiagonis rough dab) platessoides Spitz et al 2010 Ballan wrasse Labrus bergylta Benthivore 5400 Spitz et al 2010 Common dab Limanda limanda Benthivore 5800 Pleuronectes platessa Spitz et al. -

Species Composition, Distribution and Ecology of the Demersal Fish Community Along the Norwegian Coast North of Stad Under Varying Environmental Conditions
Species composition, distribution and ecology of the demersal fish community along the Norwegian coast north of Stad under varying environmental conditions Kristina Dypvik Skants University of Bergen Department of Biological Sciences - Marine Biology The Institute of Marine Research 1 Species composition, distribution and ecology of the demersal fish community along the Norwegian coast north of Stad under varying environmental conditions Kristina Dypvik Skants – Master thesis (M.Sc.) June 2019 Supervisor: |Anne Gro Vea Salvanes University of Bergen, Norway Co-supervisors: Arved Staby Institute of Marine Research, Bergen, Norway Sigbjørn Mehl Institute of Marine Research, Bergen, Norway 2 Acknowledgements First and foremost I want to thank my wonderful supervisors for all their time and commitment in helping me with this thesis. I would like to thank Anne Gro Vea Salvanes for her helpful comments and in making sure I commit to the deadlines set during the last semester of writing. Thank you to Arved Staby for all the help with the data, the (almost) monthly meetings at IMR and for always reviewing all the results I’ve sent (no matter how unfinished they’ve been). Thank you to Sigbjørn Mehl for valuable insight in the survey- design and for detailed feedback on drafts sent to you during this spring. Also, a thank you to Mikko Heino for great comments on the materials and methods and on the last draft. I would also like to thank IMR and their demersal fish research-group for the data provided for this thesis, and to the people on Johan Hjort for allowing me to join in on their annual coastal-survey in October 2018, providing me with great insight in methods used. -

Levantine Basin) Adib Saad1* , Mai Masri1 and Waad Sabour2
Saad et al. Marine Biodiversity Records (2020) 13:1 https://doi.org/10.1186/s41200-020-0185-2 MARINE RECORD Open Access First confirmed record of Sparid Pagellus bogaraveo (Brünnich, 1768) in the Syrian marine waters (Levantine Basin) Adib Saad1* , Mai Masri1 and Waad Sabour2 Abstract This paper aims to present the first record of Pagellus bogaraveo (Brünnich, 1768) from Syria marine waters. One specimen was caught by trawl nets at about 300 m depth in Rass Albassit, north of Lattakia, on 25 February 2019, and 6 additional specimens were caught by gill nets at a depth of 120 m off Jablah coast on 7 April 2019. This record represents the first sighting of this immigrant Atlantic species introduced in the Eastern Mediterranean (Levantine Basin). These specimens were found mixing in the same net haul with populations other Sparidae species such as: Pagellus acarne and Dentex macrophthalmus. Keywords: Sparidae, Pagellus bogaraveo, First record, Syrian marine waters, Levantine Basin Background by three congeneric species: Pagellus acarne (Risso, 1826), The Mediterranean is considered as a semi-enclosed sea; P. erythrinus (Linneans, 1758) and P. bellottii (Steindcner, the Western and Eastern basins are separated by siculo- 1882) (Saad 2005;Ali2018). Tunisian strait. The narrow Gibraltar strait is the only The Blackspot seabream P. bogaraveo (Brünnich, 1768), sill connection with the Atlantic Ocean. Due to strong Sparidae, is a demersal fish, inhabiting the Eastern Atlantic evaporation in the Eastern Basin, there is a constant flow Ocean, extending in the East Atlantic from Mauritania to of near-surface waters that enter through Gibraltar from Norway.