Research Priorities for Helminth Infections WHO Technical Report Series
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Human Ascariasis and Trichuriasis in Mazandaran Province, Northern Iran
Environmental Health Engineering and Management Journal 2017, 4(1), 1–6 doi 10.15171/EHEM.2017.01 http://ehemj.com Environmental Health H E M J Engineering and Management Journal Review Article Open Access Publish Free Geohelminthic: human ascariasis and trichuriasis in Mazandaran province, northern Iran Hajar Ziaei1, Fatemeh Sayyahi2, Mahboobeh Hoseiny3, Mohammad Vahedi4, Shirzad Gholami5* 1Associate Professor, Toxoplasmosis Research Center, Mazandaran University of Medical Sciences, Sari, Iran 2Medical Student, Research Committee, Faculty of Medicine, Mazandaran University of Medical Sciences, Sari, Iran 3MSC Statistic, GIS Research Center, Mazandaran University of Medical Sciences, Sari, Iran 4MSC Microbiology, Faculty Member, Department of Microbiology, Mazandaran University of Medical Sciences, Sari, Iran 5Associate Professor, Molecular and Cell Biology Research Center, Department of Parasitology and Mycology, Mazandaran University of Medical Sciences, Sari, Iran Abstract Article History: Background: Ascariasis and trichuriasis are the most common intestinal geohelminthic diseases, and Received: 21 October 2015 as such they are significant in terms of clinical and public health. This study was done to determine Accepted: 8 January 2016 prevalence, status and geographic distribution patterns for Ascariasis and Trichuriasis. The study was ePublished: 5 February 2016 done in the period 1991-2014 in northern Iran using Aregis 9.2 software. Methods: This was a review study, using description and analysis, of geographical distribution of Ascaris and Trichuris relating to townships in Mazandran province, northern Iran, covering a 23-year period. Data were collected from a review of the relevant literature, summarized and classified using Arc GIS, 9.2 to design maps and tables. Results: Based on results presented in tables and maps, means for prevalence of Ascaris and Trichuris were divided into five groups. -

A Literature Survey of Common Parasitic Zoonoses Encountered at Post-Mortem Examination in Slaughter Stocks in Tanzania: Economic and Public Health Implications
Volume 1- Issue 5 : 2017 DOI: 10.26717/BJSTR.2017.01.000419 Erick VG Komba. Biomed J Sci & Tech Res ISSN: 2574-1241 Research Article Open Access A Literature Survey of Common Parasitic Zoonoses Encountered at Post-Mortem Examination in Slaughter Stocks in Tanzania: Economic and Public Health Implications Erick VG Komba* Department of Veterinary Medicine and Public Health, Sokoine University of Agriculture, Tanzania Received: September 21, 2017; Published: October 06, 2017 *Corresponding author: Erick VG Komba, Senior lecturer, Department of Veterinary Medicine and Public Health, College of Veterinary Medicine and Biomedical Sciences, Sokoine University of Agriculture, P.O. Box 3021, Morogoro, Tanzania Abstract Zoonoses caused by parasites constitute a large group of infectious diseases with varying host ranges and patterns of transmission. Their public health impact of such zoonoses warrants appropriate surveillance to obtain enough information that will provide inputs in the design anddistribution, implementation prevalence of control and transmission strategies. Apatterns need therefore are affected arises by to the regularly influence re-evaluate of both human the current and environmental status of zoonotic factors. diseases, The economic particularly and in view of new data available as a result of surveillance activities and the application of new technologies. Consequently this paper summarizes available information in Tanzania on parasitic zoonoses encountered in slaughter stocks during post-mortem examination at slaughter facilities. The occurrence, in slaughter stocks, of fasciola spp, Echinococcus granulosus (hydatid) cysts, Taenia saginata Cysts, Taenia solium Cysts and ascaris spp. have been reported by various researchers. Information on these parasitic diseases is presented in this paper as they are the most important ones encountered in slaughter stocks in the country. -

The Functional Parasitic Worm Secretome: Mapping the Place of Onchocerca Volvulus Excretory Secretory Products
pathogens Review The Functional Parasitic Worm Secretome: Mapping the Place of Onchocerca volvulus Excretory Secretory Products Luc Vanhamme 1,*, Jacob Souopgui 1 , Stephen Ghogomu 2 and Ferdinand Ngale Njume 1,2 1 Department of Molecular Biology, Institute of Biology and Molecular Medicine, IBMM, Université Libre de Bruxelles, Rue des Professeurs Jeener et Brachet 12, 6041 Gosselies, Belgium; [email protected] (J.S.); [email protected] (F.N.N.) 2 Molecular and Cell Biology Laboratory, Biotechnology Unit, University of Buea, Buea P.O Box 63, Cameroon; [email protected] * Correspondence: [email protected] Received: 28 October 2020; Accepted: 18 November 2020; Published: 23 November 2020 Abstract: Nematodes constitute a very successful phylum, especially in terms of parasitism. Inside their mammalian hosts, parasitic nematodes mainly dwell in the digestive tract (geohelminths) or in the vascular system (filariae). One of their main characteristics is their long sojourn inside the body where they are accessible to the immune system. Several strategies are used by parasites in order to counteract the immune attacks. One of them is the expression of molecules interfering with the function of the immune system. Excretory-secretory products (ESPs) pertain to this category. This is, however, not their only biological function, as they seem also involved in other mechanisms such as pathogenicity or parasitic cycle (molting, for example). Wewill mainly focus on filariae ESPs with an emphasis on data available regarding Onchocerca volvulus, but we will also refer to a few relevant/illustrative examples related to other worm categories when necessary (geohelminth nematodes, trematodes or cestodes). -

Epidemiology of Human Fascioliasis
eserh ipidemiology of humn fsiolisisX review nd proposed new lssifition I P P wF F wsEgomD tFqF istenD 8 wFhF frgues he epidemiologil piture of humn fsiolisis hs hnged in reent yersF he numer of reports of humns psiol hepti hs inresed signifintly sine IWVH nd severl geogrphil res hve een infeted with desried s endemi for the disese in humnsD with prevlene nd intensity rnging from low to very highF righ prevlene of fsiolisis in humns does not neessrily our in res where fsiolisis is mjor veterinry prolemF rumn fsiolisis n no longer e onsidered merely s seondry zoonoti disese ut must e onsidered to e n importnt humn prsiti diseseF eordinglyD we present in this rtile proposed new lssifition for the epidemiology of humn fsiolisisF he following situtions re distinguishedX imported sesY utohthonousD isoltedD nononstnt sesY hypoED mesoED hyperED nd holoendemisY epidemis in res where fsiolisis is endemi in nimls ut not humnsY nd epidemis in humn endemi resF oir pge QRR le reÂsume en frnËisF in l p gin QRR figur un resumen en espnÄ olF ± severl rtiles report tht the inidene is sntrodution signifintly ggregted within fmily groups psiolisisD n infetion used y the liver fluke euse the individul memers hve shred the sme ontminted foodY psiol heptiD hs trditionlly een onsidered to e n importnt veterinry disese euse of the ± severl rtiles hve reported outreks not neessrily involving only fmily memersY nd sustntil prodution nd eonomi losses it uses in livestokD prtiulrly sheep nd ttleF sn ontrstD ± few rtiles hve reported epidemiologil surveys -
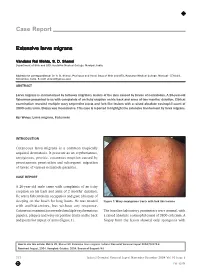
Extensive Larva Migrans
Case Report Extensive larva migrans Vandana Rai Mehta, S. D. Shenoi Department of Skin and STD, Kasturba Medical College, Manipal, India. Address for correspondence: Dr. S. D. Shenoi, Professor and Head, Dept of Skin and STD, Kasturba Medical College, Manipal - 576104, Karnataka, India. E-mail: [email protected] ABSTRACT Larva migrans is characterized by tortuous migratory lesions of the skin caused by larvae of nematodes. A 26-year-old fisherman presented to us with complaints of an itchy eruption on his back and arms of two months’ duration. Clinical examination revealed multiple wavy serpentine tracts and fork like lesions with a raised absolute eosinophil count of 3800 cells/cmm. Biopsy was inconclusive. This case is reported to highlight the extensive involvement by larva migrans. KEY WORDS: Larva migrans, Fisherman INTRODUCTION Cutaneous larva migrans is a common tropically acquired dermatosis. It presents as an erythematous, serpiginous, pruritic, cutaneous eruption caused by percutaneous penetration and subsequent migration of larvae of various nematode parasites. CASE REPORT A 26-year-old male came with complaints of an itchy eruption on his back and arms of 2 months’ duration. He was a fisherman by occupation and gave a history of sleeping on the beach for long hours. He was treated Figure 1: Wavy serpiginous tracts with fork like lesions with antihistamines, but without any response. Cutaneous examination revealed multiple erythematous The baseline laboratory parameters were normal, with papules, plaques and wavy serpentine tracts on the back a raised absolute eosinophil count of 3800 cell/cmm. A and posterior aspect of arms (Figure 1). biopsy from the lesion showed only spongiosis with How to cite this article: Mehta VR, Shenoi SD. -

Insecurities and Dogs: an Obstacle to the Eradication of Dracunculiasis
dicine & Me S l u a r ic g e p r o y r T ISSN: 2329-9088 Tropical Medicine & Surgery Review Article Insecurities and Dogs: An Obstacle to the Eradication of Dracunculiasis Aja Kalu1*,Nwufo Amanda2 Department of Care of the Elderly, Barking, Havering and Redbridge University Hospital, NHS Trust, Romford, Essex, UK; 2 Department of Public Health, University of Chester, Chester, UK. ABSTRACT Dracunculiasis is a parasitic worm infection also known as Guinea Worm Disease (GWD). It is caused by a nematode called Dracunculiasis Medinensis. It belongs to a group of communicable disease named Neglected Tropic Disease (NTD). Dracunculiasis is caused by drinking water contaminated with the vector copepods (water fleas). Although the disease is not fatal, the sores caused by the emerging worm in the lower limb can become secondarily infected and complications such as sepsis, tetanus can ensue. Also, the sores can cause abscess and cellulitis, leaving the individual incapacitated for weeks which extends beyond the emergence of the worm. Over the last three decades, the prevalence of Guinea worm disease has reduced drastically through cost effective intervention provided by The Cater Center, WHO, UNICEF with the disease targeted for eradication. Some African countries like Nigeria, Ghana, South Africa, and Kenya being the most recent, have eliminated the disease. Guinea worm is still present in Chad, Cameroon, Mali, Ethiopia where political instability, social inequalities and infection of dogs by the worm pose an increasing threat and obstacle to the elimination of the disease. Dracunculiasis represents a disease that can be eradicated without a drug or vaccine but with a cost-effective intervention that involves community efforts. -

Pathophysiology and Gastrointestinal Impacts of Parasitic Helminths in Human Being
Research and Reviews on Healthcare: Open Access Journal DOI: 10.32474/RRHOAJ.2020.06.000226 ISSN: 2637-6679 Research Article Pathophysiology and Gastrointestinal Impacts of Parasitic Helminths in Human Being Firew Admasu Hailu1*, Geremew Tafesse1 and Tsion Admasu Hailu2 1Dilla University, College of Natural and Computational Sciences, Department of Biology, Dilla, Ethiopia 2Addis Ababa Medical and Business College, Addis Ababa, Ethiopia *Corresponding author: Firew Admasu Hailu, Dilla University, College of Natural and Computational Sciences, Department of Biology, Dilla, Ethiopia Received: November 05, 2020 Published: November 20, 2020 Abstract Introduction: This study mainly focus on the major pathologic manifestations of human gastrointestinal impacts of parasitic worms. Background: Helminthes and protozoan are human parasites that can infect gastrointestinal tract of humans beings and reside in intestinal wall. Protozoans are one celled microscopic, able to multiply in humans, contributes to their survival, permits serious infections, use one of the four main modes of transmission (direct, fecal-oral, vector-borne, and predator-prey) and also helminthes are necked multicellular organisms, referred as intestinal worms even though not all helminthes reside in intestines. However, in their adult form, helminthes cannot multiply in humans and able to survive in mammalian host for many years due to their ability to manipulate immune response. Objectives: The objectives of this study is to assess the main pathophysiology and gastrointestinal impacts of parasitic worms in human being. Methods: Both primary and secondary data were collected using direct observation, books and articles, and also analyzed quantitativelyResults and and conclusion: qualitatively Parasites following are standard organisms scientific living temporarily methods. in or on other organisms called host like human and other animals. -

Waterborne Zoonotic Helminthiases Suwannee Nithiuthaia,*, Malinee T
Veterinary Parasitology 126 (2004) 167–193 www.elsevier.com/locate/vetpar Review Waterborne zoonotic helminthiases Suwannee Nithiuthaia,*, Malinee T. Anantaphrutib, Jitra Waikagulb, Alvin Gajadharc aDepartment of Pathology, Faculty of Veterinary Science, Chulalongkorn University, Henri Dunant Road, Patumwan, Bangkok 10330, Thailand bDepartment of Helminthology, Faculty of Tropical Medicine, Mahidol University, Ratchawithi Road, Bangkok 10400, Thailand cCentre for Animal Parasitology, Canadian Food Inspection Agency, Saskatoon Laboratory, Saskatoon, Sask., Canada S7N 2R3 Abstract This review deals with waterborne zoonotic helminths, many of which are opportunistic parasites spreading directly from animals to man or man to animals through water that is either ingested or that contains forms capable of skin penetration. Disease severity ranges from being rapidly fatal to low- grade chronic infections that may be asymptomatic for many years. The most significant zoonotic waterborne helminthic diseases are either snail-mediated, copepod-mediated or transmitted by faecal-contaminated water. Snail-mediated helminthiases described here are caused by digenetic trematodes that undergo complex life cycles involving various species of aquatic snails. These diseases include schistosomiasis, cercarial dermatitis, fascioliasis and fasciolopsiasis. The primary copepod-mediated helminthiases are sparganosis, gnathostomiasis and dracunculiasis, and the major faecal-contaminated water helminthiases are cysticercosis, hydatid disease and larva migrans. Generally, only parasites whose infective stages can be transmitted directly by water are discussed in this article. Although many do not require a water environment in which to complete their life cycle, their infective stages can certainly be distributed and acquired directly through water. Transmission via the external environment is necessary for many helminth parasites, with water and faecal contamination being important considerations. -

Public Health Significance of Intestinal Parasitic Infections*
Articles in the Update series Les articles de la rubrique give a concise, authoritative, Le pointfournissent un bilan and up-to-date survey of concis et fiable de la situa- the present position in the tion actuelle dans les do- Update selectedfields, coveringmany maines consideres, couvrant different aspects of the de nombreux aspects des biomedical sciences and sciences biomedicales et de la , po n t , , public health. Most of santepublique. Laplupartde the articles are written by ces articles auront donc ete acknowledged experts on the redigeis par les specialistes subject. les plus autorises. Bulletin of the World Health Organization, 65 (5): 575-588 (1987) © World Health Organization 1987 Public health significance of intestinal parasitic infections* WHO EXPERT COMMITTEE' Intestinal parasitic infections are distributed virtually throughout the world, with high prevalence rates in many regions. Amoebiasis, ascariasis, hookworm infection and trichuriasis are among the ten most common infections in the world. Other parasitic infections such as abdominal angiostrongyliasis, intestinal capil- lariasis, and strongyloidiasis are of local or regional public health concern. The prevention and control of these infections are now more feasible than ever before owing to the discovery of safe and efficacious drugs, the improvement and sim- plification of some diagnostic procedures, and advances in parasite population biology. METHODS OF ASSESSMENT The amount of harm caused by intestinal parasitic infections to the health and welfare of individuals and communities depends on: (a) the parasite species; (b) the intensity and course of the infection; (c) the nature of the interactions between the parasite species and concurrent infections; (d) the nutritional and immunological status of the population; and (e) numerous socioeconomic factors. -

Cytotaxonomy, Morphology and Molecular Systematics of the Bioko Form of Simulium Yahense (Diptera: Simuliidae)
Bulletin of Entomological Research (2003) 93, 145–157 DOI: 10.1079/BER2003228 Cytotaxonomy, morphology and molecular systematics of the Bioko form of Simulium yahense (Diptera: Simuliidae) R.J. Post1*, P.K. Flook2, A.L. Millest3, R.A. Cheke4, P.J. McCall2, M.D. Wilson5, M. Mustapha1, S. Somiari6, J.B. Davies2, R.A. Mank7, P. Geenen7, P. Enyong8, A. Sima9 and J. Mas10 1Department of Entomology, The Natural History Museum, Cromwell Road, London, SW7 5BD, UK: 2Division of Parasite and Vector Biology, Liverpool School of Tropical Medicine, UK: 3Department of Biological Sciences, University of Salford, UK: 4Natural Resources Institute, University of Greenwich, Chatham, UK: 5Noguchi Memorial Institute for Medical Research, University of Ghana, Ghana: 6Windber Research Institute, Windber, USA: 7Animal Taxonomy Section, Wageningen University, The Netherlands: 8Medical Research Station, Kumba, Cameroon: 9Onchocerciasis Control Programme, Ministry of Health, Malabo, Equatorial Guinea: 10Onchocerciasis Control Programme, Spanish International Cooperation Agency, Malabo, Equatorial Guinea Abstract Cytotaxonomic analysis of the polytene chromosomes from larvae of the Simulium damnosum Theobald complex from the island of Bioko in Equatorial Guinea is reported, and a new endemic cytoform is described. Chromosomally this cytoform is close to both S. squamosum (Enderlein) and S. yahense Vajime & Dunbar, but is not identical to either. However, it is morphologically and enzymatically identical to S. yahense. The Bioko form was also found to differ from other cytoforms of the S. damnosum complex in West Africa in the copy number or RFLP pattern of several different repetitive DNA sequences. It is clear that the Bioko form is genetically distinct from other populations of the S. -

Biliary Obstruction Caused by the Liver Fluke, Fasciola Hepatica
CME Practice CMAJ Cases Biliary obstruction caused by the liver fluke, Fasciola hepatica Takuya Ishikawa MD PhD, Vanessa Meier-Stephenson MD PhD, Steven J. Heitman MD MSc Competing interests: None 20-year-old previously healthy man declared. presented to hospital with a two-day This article has been peer A history of right upper quadrant pain reviewed. and vomiting. Nine months earlier, he had The authors have obtained immigrated to Canada from Sudan, but he had patient consent. also lived in Djibouti and Ethiopia. Four Correspondence to: months before he presented to hospital, he Steven Heitman, received a diagnosis of tuberculous lymphade- [email protected] nitis and a four-drug course of tuberculosis CMAJ 2016. DOI:10.1503 treatment was started. However, he was non- /cmaj.150696 adherent after only two months of treatment. In addition, results from screening tests at that time showed evidence of schistosomiasis for Figure 1: A flat, leaf-shaped, brown worm emerg- which he was prescribed praziquantel. ing from the common bile duct of a 20-year-old On examination, he was alert and without man with abdominal pain. jaundice or scleral icterus. He had right upper quadrant tenderness on abdominal examination, ter of 1.1 cm. A computed tomography scan of but there were no palpable masses. The remain- the abdomen also showed prominence of the der of his examination was unremarkable. Labo- common bile duct, but no calcified stone was ratory test results showed elevated liver enzymes identified (Appendix 1). A hepatobiliary imino- (aspartate transaminase 133 [normal < 40] U/L, diacetic acid scan suggested distal obstruction in alanine transaminase 217 [normal < 41] U/L, the common bile duct. -

Mandrillus Leucophaeus Poensis)
Ecology and Behavior of the Bioko Island Drill (Mandrillus leucophaeus poensis) A Thesis Submitted to the Faculty of Drexel University by Jacob Robert Owens in partial fulfillment of the requirements for the degree of Doctor of Philosophy December 2013 i © Copyright 2013 Jacob Robert Owens. All Rights Reserved ii Dedications To my wife, Jen. iii Acknowledgments The research presented herein was made possible by the financial support provided by Primate Conservation Inc., ExxonMobil Foundation, Mobil Equatorial Guinea, Inc., Margo Marsh Biodiversity Fund, and the Los Angeles Zoo. I would also like to express my gratitude to Dr. Teck-Kah Lim and the Drexel University Office of Graduate Studies for the Dissertation Fellowship and the invaluable time it provided me during the writing process. I thank the Government of Equatorial Guinea, the Ministry of Fisheries and the Environment, Ministry of Information, Press, and Radio, and the Ministry of Culture and Tourism for the opportunity to work and live in one of the most beautiful and unique places in the world. I am grateful to the faculty and staff of the National University of Equatorial Guinea who helped me navigate the geographic and bureaucratic landscape of Bioko Island. I would especially like to thank Jose Manuel Esara Echube, Claudio Posa Bohome, Maximilliano Fero Meñe, Eusebio Ondo Nguema, and Mariano Obama Bibang. The journey to my Ph.D. has been considerably more taxing than I expected, and I would not have been able to complete it without the assistance of an expansive list of people. I would like to thank all of you who have helped me through this process, many of whom I lack the space to do so specifically here.