Daiichi Sankyo Group Value Report 2017
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Daiichi Sankyo Company, Limited
[Translation] CONVOCATION NOTICE OF THE 14TH ORDINARY GENERAL MEETING OF SHAREHOLDERS For the Fiscal Year Ended March 31, 2019 Daiichi Sankyo Company, Limited *Note: This translation does not include pictures, charts etc. originally issued in the Japanese version. - 1 - [Translation] To Our Shareholders At the Daiichi Sankyo Group (“the Group”), we are proceeding with the initiatives of 4th mid- term business plan with the aim of becoming a “Global Pharma Innovator with Competitive Advantage in Oncology” as set forth in our 2025 Vision. In fiscal 2018, we made significant progress in developing new drugs in oncology area, including DS-8201, an antibody drug conjugates utilizing our proprietary technologies. In addition, we entered into a strategic collaboration agreement with AstraZeneca, which has strengths in the oncology business, for the global development and commercialization of DS-8201 in order to maximize its value. Furthermore, sales of mainstay products such as edoxaban, an anticoagulant which supports current earnings of the Group, were firm in Japan and overseas. The achievement of initial target of mid-term business plan for fiscal 2020 is expected to be delayed by two years due to failure of achievement of the plan for pain franchise business and additional investments in research and development. However, we are gaining confidence of achieving our 2025 Vision and accelerating growth in the future due to the significant improvement in the value of oncology area pipelines. We will continue to make every effort to achieve the goals of the Medium-Term Management Plan and 2025 Vision. I greatly appreciate your continued support in the future. -

Daiichi Sankyo Group Value Report 2019
External Evaluations (as of June 30,2019) ™ Daiichi Sankyo Group Value Report 2019 Value Daiichi Sankyo Group MSCI Japan Empowering Women Select Index THE INCLUSION OF DAIICHI SANKYO CO.,LTD. IN ANY MSCI INDEX, AND THE USE OF MSCI LOGOS, TRADEMARKS, SERVICE MARKS OR INDEX NAMES HEREIN, DO NOT CONSTITUTE A SPONSORSHIP, ENDORSEMENT OR PROMOTION OF DAIICHI SANKYO CO.,LTD. BY MSCI OR ANY OF ITS AFFILIATES. THE MSCI INDEXES ARE THE EXCLUSIVE PROPERTY OF MSCI. MSCI AND THE MSCI INDEX NAMES AND LOGOS ARE TRADE- MARKS OR SERVICE MARKS OF MSCI OR ITS AFFILIATES. “Eruboshi” Certification Mark “Kurumin” Certification Mark Logo given to Certified Health and Productivity Management Organization (White500) This report uses FSC® certified paper, which indicates that the paper used to print this Paper report was produced from properly managed forests. 3-5-1, Nihonbashi-honcho, Chuo-ku, Tokyo 103-8426, Japan This report was printed using 100% Inks biodegradable printing inks from vegetable Corporate Communications Department oil. Daiichi Sankyo Group Tel: +81-3-6225-1126 CSR Department The waterless printing method used for this Value Report 2019 Tel: +81-3-6225-1067 Printing report minimized the use and release of harmful liquid wastes. https://www.daiichisankyo.com/ Printed in Japan 005_7045687911909.indd 1 2019/09/27 18:22:19 Introduction Our Mission The Core Values and Commitments serve as the criteria for business activities and In addition, we have established the DAIICHI SANKYO Group Corporate Conduct Charter . decision-making used by executive officers and employees in working to fulfill Our Mission . This charter calls on us to fulfill our social responsibilities by acting with the highest ethical Our Corporate Slogan succinctly explains the spirit of Our Mission, Core Values and standards and a good social conscience appropriate for a company engaged in business Commitments. -

Hyperaldosteronism: How Current Concepts Are Transforming the Diagnostic and Therapeutic Paradigm
Kidney360 Publish Ahead of Print, published on July 23, 2020 as doi:10.34067/KID.0000922020 Hyperaldosteronism: How Current Concepts Are Transforming the Diagnostic and Therapeutic Paradigm Michael R Lattanzio(1), Matthew R Weir(2) (1) Division of Nephrology, Department of Medicine, The Chester County Hospital/University of Pennsylvania Health System (2) Division of Nephrology, Department of Medicine, University of Maryland School of Medicine, Baltimore, MD Correspondence: Matthew R Weir University of Maryland Medical Center Division of Nephrology 22 S. Greene St. Room N3W143 Baltimore, Maryland 21201 [email protected] 1 Copyright 2020 by American Society of Nephrology. Abbreviations PA=Primary Hyperaldosteronism CVD=Cardiovascular Disease PAPY=Primary Aldosteronism Prevalence in Hypertension APA=Aldosterone-Producing Adenoma BAH=Bilateral Adrenal Hyperplasia ARR=Aldosterone Renin Ratio AF=Atrial Fibrillation OSA=Obstructive Sleep Apnea OR=Odds Ratio AHI=Apnea Hypopnea Index ABP=Ambulatory Blood Pressure AVS=Adrenal Vein Sampling CT=Computerized Tomography MRI=Magnetic Resonance Imaging SIT=Sodium Infusion Test FST=Fludrocortisone Suppression Test CCT=Captopril Challenge Test PAC=Plasma Aldosterone Concentration PRA=Plasma Renin Activity MRA=Mineralocorticoid Receptor Antagonist MR=Mineralocorticoid Receptor 2 Abstract Nearly seven decades have elapsed since the clinical and biochemical features of Primary Hyperaldosteronism (PA) were described by Conn. PA is now widely recognized as the most common form of secondary hypertension. PA has a strong correlation with cardiovascular disease and failure to recognize and/or properly diagnose this condition has profound health consequences. With proper identification and management, PA has the potential to be surgically cured in a proportion of affected individuals. The diagnostic pursuit for PA is not a simplistic endeavor, particularly since an enhanced understanding of the disease process is continually redefining the diagnostic and treatment algorithm. -

Convocation Notice of the 16Th Ordinary General Meeting of Shareholders
[Translation] CONVOCATION NOTICE OF THE 16TH ORDINARY GENERAL MEETING OF SHAREHOLDERS For the Fiscal Year Ended March 31, 2021 Daiichi Sankyo Company, Limited *Note: This translation does not include pictures, charts etc. originally issued in the Japanese version. [Translation] To Our Shareholders We sincerely appreciate the continuous kindness of our shareholders. In addition, we would like to express our deepest sympathies to those who passed away due to COVID-19, and thank the medical personnel who are close to those who are fighting illness and are making efforts in treatment. We will continue to devote ourselves to the research and development of vaccines and therapeutic agents. Our “Purpose” is to “contribute to the enrichment of quality of life around the world.” As a pharmaceutical company with strengths in science and technology, we continuously create innovative pharmaceuticals and provide pharmaceuticals that meet diverse medical needs to provide sustainable value to society. We were able to launch the anti-cancer drug “Enhertu”, which is an antibody-drug conjugate (ADC) that utilizes our unique technology, in Japan and Europe in fiscal 2020, following the launch in the U.S. in fiscal 2019. Subsequent ADCs such as Dato-DXd and HER3-DXd are also steadily developing. Now, we have newly established our 2030 Vision of being an “Innovative Global Healthcare Company Contributing to the Sustainable Development of Society,” and have established 5-Year Business Plan (fiscal 2021 to fiscal 2025) as a plan to realize our 2025 Vision, “Global Pharma Innovator with Competitive Advantage in Oncology.” By working together as a Daiichi Sankyo Group on 5-Year Business Plan toward the 2030 Vision, we aim to solve the social issues expected of our company and increase shareholder value. -

Daiichi Sankyo Company, Limited
[Translation] CONVOCATION NOTICE OF THE 6TH ORDINARY GENERAL MEETING OF SHAREHOLDERS For the Fiscal Period Ended March 31, 2011 Daiichi Sankyo Company, Limited - 1 - [Translation] (Securities Identification Code 4568) May 31, 2011 To Shareholders, Daiichi Sankyo Company, Limited Joji Nakayama, Representative Director and President & CEO 5-1, Nihonbashi Honcho 3-chome, Chuo-ku, Tokyo, Japan CONVOCATION NOTICE OF THE 6TH ORDINARY GENERAL MEETING OF SHAREHOLDERS We wish to extend our deepest sympathy to all those who have suffered hardship from the Great East Japan Earthquake that occurred in March 2011. Daiichi Sankyo Company, Limited (“the Company”) respectfully requests your attendance at the 6th Ordinary General Meeting of Shareholders (“the Meeting”), which will be held as detailed below. If you will not be able to attend the Meeting, you may exercise your voting rights through either of the methods described below, in which case we ask that you please exercise your voting rights by 17:30 (within our business hours), Friday, June 24, 2011 (Japan Time), after examining the attached reference documents. [Exercise of Voting Rights by Mail] Please indicate your approval or disapproval for the proposals on the enclosed voting form and return the form to the Company. Please note that the form must be received by the Company no later than the above-mentioned deadline. [Exercise of Voting Rights on the Internet etc.] After examining “Information on Exercise of Voting Rights, etc.” on pages 57 and 58, please vote on the Internet at the dedicated voting website (http://www.evote.jp/) no later than the above-mentioned deadline. The Company is participating in the platform for electronic exercise of voting rights for institutional investors operated by ICJ Inc. -

Ovid MEDLINE(R)
Supplementary material BMJ Open Ovid MEDLINE(R) and Epub Ahead of Print, In-Process & Other Non-Indexed Citations and Daily <1946 to September 16, 2019> # Searches Results 1 exp Hypertension/ 247434 2 hypertens*.tw,kf. 420857 3 ((high* or elevat* or greater* or control*) adj4 (blood or systolic or diastolic) adj4 68657 pressure*).tw,kf. 4 1 or 2 or 3 501365 5 Sex Characteristics/ 52287 6 Sex/ 7632 7 Sex ratio/ 9049 8 Sex Factors/ 254781 9 ((sex* or gender* or man or men or male* or woman or women or female*) adj3 336361 (difference* or different or characteristic* or ratio* or factor* or imbalanc* or issue* or specific* or disparit* or dependen* or dimorphism* or gap or gaps or influenc* or discrepan* or distribut* or composition*)).tw,kf. 10 or/5-9 559186 11 4 and 10 24653 12 exp Antihypertensive Agents/ 254343 13 (antihypertensiv* or anti-hypertensiv* or ((anti?hyperten* or anti-hyperten*) adj5 52111 (therap* or treat* or effective*))).tw,kf. 14 Calcium Channel Blockers/ 36287 15 (calcium adj2 (channel* or exogenous*) adj2 (block* or inhibitor* or 20534 antagonist*)).tw,kf. 16 (agatoxin or amlodipine or anipamil or aranidipine or atagabalin or azelnidipine or 86627 azidodiltiazem or azidopamil or azidopine or belfosdil or benidipine or bepridil or brinazarone or calciseptine or caroverine or cilnidipine or clentiazem or clevidipine or columbianadin or conotoxin or cronidipine or darodipine or deacetyl n nordiltiazem or deacetyl n o dinordiltiazem or deacetyl o nordiltiazem or deacetyldiltiazem or dealkylnorverapamil or dealkylverapamil -

Press Release
Press Release Company name: DAIICHI SANKYO COMPANY, LIMITED Representative: Sunao Manabe, Representative Director, President and CEO (Code no.: 4568, First Section, Tokyo Stock Exchange) Please address inquiries to Junichi Onuma, Vice President, Corporate Communications Department Telephone: +81-3-6225-1126 https://www.daiichisankyo.com Daiichi Sankyo Announces Transfer from Astellas Pharma of Three Products in Asia Tokyo, Japan (October 15, 2019) – Daiichi Sankyo Company, Limited (hereafter, Daiichi Sankyo) today announced that it agreed with Astellas Pharma Inc. (hereafter, Astellas Pharma) that Astellas Pharma local subsidiaries companies in six Asian countries will transfer three products to Daiichi Sankyo. The products to be transferred and the countries where they are sold are as follows. Product Korea China Taiwan Thailand Philippines Indonesia [generic name (brand name)] Ramosetron Antiemetic ○ ○ ○ ○ (Nasea) Nicardipine Anti- ○ ○ ○ (Perdipine) hypertensive Barnidipine Anti- ○ (Oldeca) hypertensive ○ indicate the countries where the products are sold The antiemetics are expected to have synergistic effects with mirogabalin and the cancer drugs that Daiichi Sankyo is currently developing in Asia, and the two antihypertensives are expected to effectively utilize Daiichi Sankyo’s current infrastructures in combination with its cardiovascular products, such as olmesartan and edoxaban. The total net sales of Astellas Pharma's three products in fiscal year 2018 were approximately 5.0 billion yen. 1 Daiichi Sankyo will take over the rights -
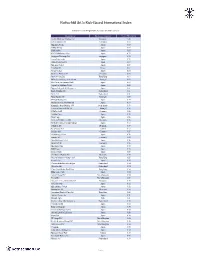
R&Co Risk-Based International Index – Weighting
Rothschild & Co Risk-Based International Index Indicative Index Weight Data as of June 30, 2021 on close Constituent Exchange Country Index Weight(%) Jardine Matheson Holdings Ltd Singapore 1.46 LEG Immobilien SE Germany 0.98 Ajinomoto Co Inc Japan 0.95 SoftBank Corp Japan 0.89 Shimano Inc Japan 0.85 FUJIFILM Holdings Corp Japan 0.73 Singapore Exchange Ltd Singapore 0.72 Japan Tobacco Inc Japan 0.72 Cellnex Telecom SA Spain 0.69 Nintendo Co Ltd Japan 0.69 Carrefour SA France 0.67 Nexon Co Ltd Japan 0.66 Deutsche Wohnen SE Germany 0.65 Bank of China Ltd Hong Kong 0.64 REN - Redes Energeticas Nacion Portugal 0.63 Pan Pacific International Hold Japan 0.63 Japan Post Holdings Co Ltd Japan 0.62 Nippon Telegraph & Telephone C Japan 0.61 Roche Holding AG Switzerland 0.61 Nestle SA Switzerland 0.61 Novo Nordisk A/S Denmark 0.59 ENEOS Holdings Inc Japan 0.59 Nomura Research Institute Ltd Japan 0.59 Koninklijke Ahold Delhaize NV Netherlands 0.59 Jeronimo Martins SGPS SA Portugal 0.58 HelloFresh SE Germany 0.58 Toshiba Corp Japan 0.58 Hoya Corp Japan 0.58 Siemens Healthineers AG Germany 0.58 MS&AD Insurance Group Holdings Japan 0.57 Coloplast A/S Denmark 0.57 Kerry Group PLC Ireland 0.57 Scout24 AG Germany 0.57 SG Holdings Co Ltd Japan 0.56 Symrise AG Germany 0.56 Nitori Holdings Co Ltd Japan 0.56 Beiersdorf AG Germany 0.55 Mitsubishi Corp Japan 0.55 KDDI Corp Japan 0.55 Sysmex Corp Japan 0.55 Chr Hansen Holding A/S Denmark 0.55 Ping An Insurance Group Co of Hong Kong 0.55 Eisai Co Ltd Japan 0.54 Chocoladefabriken Lindt & Spru Switzerland 0.54 Givaudan -

Daiichi Sankyo Group Value Report 2016
Daiichi Sankyo Group Value Report 2016 Daiichi Sankyo Group Value Daiichi Sankyo Group Value Report 2016 Our Mission Core Values and Commitments(Criteria of the Value Judgment to Fulfill Our Mission) Core Values Notes To contribute to the enrichment of quality of life around the world Innovation : the introduction of new ideas, methods, or invention through the creation of innovative pharmaceuticals, and through the Integrity : the quality of being honest and of always having high moral principles provision of pharmaceuticals addressing diverse medical needs. Accountability : being responsible for the effects of your actions, and being willing to explain or be criticized for them Commitments We have established Core Values and Commitments as the criteria for our business activities 1. To create innovative medicines changing SOC * and decision making. Our global brand is a pledge to our stakeholders of what the * SOC (Standard of Care): Universally applied best treatment practice in today’s medical science Company is capable of delivering, now and in the future. Our corporate slogan succinctly 2. To take a global perspective, and respect regional values states how we make efforts for what and for whom. 3. To foster intellectual curiosity and strategic insight In addition, we have established the DAIICHI SANKYO Group Corporate Conduct Charter * to act with the highest ethical standards and a good social conscience appropriate 4. To provide the highest quality medical information for a company engaged in a business that affects human lives. 5. To provide a stable supply of top-quality pharmaceutical products * The full text of the DAIICHI SANKYO Group Corporate Conduct Charter can be found on page 28. -

Mineralocorticoid Receptor Blockers for Moderate Kidney Dysfunction: More Merit Than Ever?
Hypertension Research (2021) 44:1352–1354 https://doi.org/10.1038/s41440-021-00690-6 COMMENT Mineralocorticoid receptor blockers for moderate kidney dysfunction: more merit than ever? 1 1 1 Masashi Mukoyama ● Takashige Kuwabara ● Masataka Adachi Received: 28 May 2021 / Accepted: 31 May 2021 / Published online: 15 July 2021 © The Japanese Society of Hypertension 2021 Mineralocorticoid receptor (MR) antagonists, such as spir- MR activity with high specificity. To date, several clinical onolactone and eplerenone, are currently used to treat studies have been performed to evaluate its effects [4–6], hypertension and chronic heart failure. MR antagonists act which revealed effective blood pressure reduction with rela- on MRs expressed in the distal tubules of the kidney, tively few adverse events, including hyperkalemia (Table 1). including distal convoluted tubules, connecting tubules and In addition, esaxerenone can further reduce urinary albumin the cortical collecting duct, thereby promoting sodium excretion in addition to RAS inhibitors without significantly excretion without the loss of potassium into the urine [1]. affecting renal function [6]. Finerenone, a second nonsteroidal MR antagonists may be effective for the treatment of low- MR blocker for which large-scale clinical trials have been 1234567890();,: 1234567890();,: renin hypertension and are preferentially used for the completed [7, 8], showed a lower risk of CKD progression treatment of primary aldosteronism; furthermore, in patients and cardiovascular events than placebo in patients with CKD with resistant hypertension, additional administration at a and type 2 diabetes (Table 1). Based on these results, much low to moderate dose (spironolactone, 25–50 mg/day) may attention has been given to novel MR blockers as promising further decrease blood pressure [2]. -

Summary Conflict of Interest Statements BCC 2021
Summary conflict of interest statements BCC 2021 Last name First name Type of affiliation/ financial interest Name of commercial company Aapro Matti Receipt of grants/research supports: Amgen, Eisai, Genomic Health, Helsinn, Hospira, Novartis, Merck, Mundipharma, Pfizer, Rache, Sandoz, Tesaro, Teva, Vifor Receipt of honoraria or consultation fees: Accord Pharmaceuticals, Amgen, Astellas, Bayer HealthCare Pharmaceuticals (Schering), Biocon, Boehringer Ingelheim, BMS, Celgene, Cephalon, Chugai Pharmaceutical Co. Ltd., Clinigen Group, Dr.Reddy's Laboratories, Eisai Co. Ltd., Eli Lilly, Genomic Health (Exact Sciences), GlaxoSmithKline (GSK), Glenmark Pharmaceuticals Limited, Gl Therapeutics, lnc., Helsinn Healthcare SA, Hospira (Pfizer), lpsen, Janssen Biotech, Johnson & Johnson, Kyowa Kirin Group, Merck, Merck Serono (Merck KGaA), Mundipharma International Limited, Novartis, Pfizer, Pierre Fabre, Rache, Sandoz, Sanofi, Taiho Pharmaceutical, Tesaro (GSK), Teva Pharmaceutical lndustries Ltd., Vifor Pharma Other support: European Cancer Organisation, SPCC, Cancer Center Genolier Aebi Stephan Receipt of honoraria or consultation fees: Novartis, Roche, Pfizer Other support: Support for CME lectures of the Lucerne Cancer Center: Amgen, Astellas, Bayer, Bristol-Myers Squibb, Debiopharm International SA, Eisai, Ipsen Pharma, Janssen, Merck, MSD, Pfizer, Roche, Sanofi Genzyme, Servier, Takeda André Fabrice Receipt of grants/research supports: Comepensated to the hospital: Roche, AstraZeneca, Daiichi Sankyo, Pfizer, Novartis, Lilly Barrios Carlos -

Senior Executives
Senior Executives Mizuho Financial Group Masayuki Hoshi Tsuyoshi Hatsuzawa Managing Executive Officer Managing Executive Officer Directors and Executive Officers, Head of Financial Institutions & Public Sector Deputy Head of Risk Management Group Business Unit / Head of Transaction Banking Unit as defined in the Companies Act Kazuhisa Sawa Yasuhiro Sato Tomoshige Jingu Managing Executive Officer Director, President & Group CEO Managing Executive Officer Deputy Head of Strategic Planning Group / Deputy (Representative Executive Officer) Head of Human Resources Group (Group CHRO) Head of Financial Control & Accounting Group / Deputy Head of IT & Systems Group / Deputy Head Yasunori Tsujita of Operations Group Executive Officers, as defined in Director, Deputy President & Executive Officer Takashi Kamada (Representative Executive Officer) our internal regulations Managing Executive Officer Head of Internal Control (Risk Management, Human Deputy Head of Personal Banking Unit / Deputy Resources and Compliance) and Head of Compliance Masaaki Kono Head of Retail Banking Unit Group (Group CCO) Deputy President & Executive Officer Deputy President (Western Japan (Kinki, Chugoku Ryusuke Aya and Shikoku regions)) Yoshihito Bando Managing Executive Officer Director, Managing Executive Officer Deputy Head of Asset Management Unit Head of Risk Management Group (Group CRO) Katsunobu Motohashi Managing Executive Officer Junichi Shinbo Head of Asset Management Unit Kenichi Minamitani Managing Executive Officer Director, Managing Executive Officer Deputy