Reniva Monograph
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Valstybinės Vaistų Kontrolės Tarnybos Prie Lietuvos Respublikos Sveikatos Apsaugos Ministerijos Viršininko Įsakymas
VALSTYBINĖS VAISTŲ KONTROLĖS TARNYBOS PRIE LIETUVOS RESPUBLIKOS SVEIKATOS APSAUGOS MINISTERIJOS VIRŠININKO ĮSAKYMAS DĖL VALSTYBINĖS VAISTŲ KONTROLĖS TARNYBOS PRIE LIETUVOS RESPUBLIKOS SVEIKATOS APSAUGOS MINISTERIJOS VIRŠININKO 2006 M. LAPKRIČIO 2 D. ĮSAKYMO NR. 1A-658 „DĖL TARPTAUTINIŲ PREKĖS ŽENKLU NEREGISTRUOTŲ VAISTINIŲ MEDŽIAGŲ PAVADINIMŲ ATITIKMENŲ LIETUVIŲ KALBA SĄRAŠO PATVIRTINIMO“ PAKEITIMO 2011 m. gegužės 5 d. Nr. 1A-416 Vilnius Atsižvelgdamas į Pasaulinės sveikatos organizacijos 2011 m. paskelbtą 65-ąjį Rekomenduojamų tarptautinių prekės ženklu neregistruotų vaistinių medžiagų pavadinimų (INN) sąrašą, p a k e i č i u Tarptautinių prekės ženklu neregistruotų vaistinių medžiagų pavadinimų atitikmenų lietuvių kalba sąrašą, patvirtintą Valstybinės vaistų kontrolės tarnybos prie Lietuvos Respublikos sveikatos apsaugos ministerijos viršininko 2006 m. lapkričio 2 d. įsakymu Nr. 1A-658 „Dėl Tarptautinių prekės ženklu neregistruotų vaistinių medžiagų pavadinimų atitikmenų lietuvių kalba sąrašo patvirtinimo“ (Žin., 2006, Nr. 119-4557; 2007, Nr. 13-519, Nr. 43-1672; 2008, Nr. 9- 330, Nr. 24-896; 2010, Nr. 152-7773): 1. Papildau nauja eilute, kurią po eilutės „Amtolmetin guacil Amtolmetinas guacilas Amtolmetinum guacilum“ išdėstau taip: „Amuvatinib Amuvatinibas Amuvatinibum“ 2. Papildau nauja eilute, kurią po eilutės „Anagestone Anagestonas Anagestonum“ išdėstau taip: „Anagliptin Anagliptinas Anagliptinum“ 3. Papildau nauja eilute, kurią po eilutės „Atazanavir Atazanaviras Atazanavirum“ išdėstau taip: „Atecegatran Atecegatranas Atecegatranum“ -

Natural Products As Lead Compounds for Sodium Glucose Cotransporter (SGLT) Inhibitors
Reviews Natural Products as Lead Compounds for Sodium Glucose Cotransporter (SGLT) Inhibitors Author ABSTRACT Wolfgang Blaschek Glucose homeostasis is maintained by antagonistic hormones such as insulin and glucagon as well as by regulation of glu- Affiliation cose absorption, gluconeogenesis, biosynthesis and mobiliza- Formerly: Institute of Pharmacy, Department of Pharmaceu- tion of glycogen, glucose consumption in all tissues and glo- tical Biology, Christian-Albrechts-University of Kiel, Kiel, merular filtration, and reabsorption of glucose in the kidneys. Germany Glucose enters or leaves cells mainly with the help of two membrane integrated transporters belonging either to the Key words family of facilitative glucose transporters (GLUTs) or to the Malus domestica, Rosaceae, Phlorizin, flavonoids, family of sodium glucose cotransporters (SGLTs). The intesti- ‑ SGLT inhibitors, gliflozins, diabetes nal glucose absorption by endothelial cells is managed by SGLT1, the transfer from them to the blood by GLUT2. In the received February 9, 2017 kidney SGLT2 and SGLT1 are responsible for reabsorption of revised March 3, 2017 filtered glucose from the primary urine, and GLUT2 and accepted March 6, 2017 GLUT1 enable the transport of glucose from epithelial cells Bibliography back into the blood stream. DOI http://dx.doi.org/10.1055/s-0043-106050 The flavonoid phlorizin was isolated from the bark of apple Published online April 10, 2017 | Planta Med 2017; 83: 985– trees and shown to cause glucosuria. Phlorizin is an inhibitor 993 © Georg Thieme Verlag KG Stuttgart · New York | of SGLT1 and SGLT2. With phlorizin as lead compound, specif- ISSN 0032‑0943 ic inhibitors of SGLT2 were developed in the last decade and some of them have been approved for treatment mainly of Correspondence type 2 diabetes. -

CDR Clinical Review Report for Soliqua
CADTH COMMON DRUG REVIEW Clinical Review Report Insulin glargine and lixisenatide injection (Soliqua) (Sanofi-Aventis) Indication: adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus inadequately controlled on basal insulin (less than 60 units daily) alone or in combination with metformin. Service Line: CADTH Common Drug Review Version: Final (with redactions) Publication Date: January 2019 Report Length: 118 Pages Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services. While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. -

Download Product Insert (PDF)
PRODUCT INFORMATION Remogliflozin A Item No. 14340 CAS Registry No.: 329045-45-6 OH Formal Name: 5-methyl-4-[[4-(1-methylethoxy) N O OH phenyl]methyl]-1-(1-methylethyl)- N 1H-pyrazol-3-yl β-D- O glucopyranoside OH Synonym: GSK189074 OH MF: C23H34N2O7 FW: 450.5 Purity: ≥98% λ: 229 nm UV/Vis.: max O Supplied as: A crystalline solid Storage: -20°C Stability: ≥2 years Information represents the product specifications. Batch specific analytical results are provided on each certificate of analysis. Laboratory Procedures Remogliflozin A is supplied as a crystalline solid. A stock solution may be made by dissolving the remogliflozin A in the solvent of choice. Remogliflozin A is soluble in organic solvents such as ethanol, DMSO, and dimethyl formamide, which should be purged with an inert gas. The solubility of remogliflozin A in these solvents is approximately 30 mg/ml. Remogliflozin A is sparingly soluble in aqueous buffers. For maximum solubility in aqueous buffers, remogliflozin A should first be dissolved in ethanol and then diluted with the aqueous buffer of choice. Remogliflozin A has a solubility of approximately 0.5 mg/ml in a 1:1 solution of ethanol:PBS (pH 7.2) using this method. We do not recommend storing the aqueous solution for more than one day. Description Remogliflozin A is a potent inhibitor of sodium-glucose transporter 2 (SGLT2; Kis = 12.4 and 26 nM 1 for human and rat SGLT2, respectively). It is selective for SGLT2 over SGLT1 (Kis = 4,520 and 997 nM for human and rat SGLT1, respectively). Following administration of a prodrug, remogliflozin etabonate, that is rapidly converted to remogliflozin A in vivo, rat urinary glucose excretion increases and plasma glucose and insulin concentrations decrease. -

Remogliflozin Etabonate, a Selective Inhibitor of the Sodium-Glucose
Clinical Care/Education/Nutrition/Psychosocial Research BRIEF REPORT Remogliflozin Etabonate, a Selective Inhibitor of the Sodium-Glucose Transporter 2, Improves Serum Glucose Profiles in Type 1 Diabetes 1,2 3 SUNDER MUDALIAR, MD JUNE YE, PHD placebo (placebo), 2) mealtime insulin 1 3 DEBRA A. ARMSTRONG, BA, RN, CCRC ELIZABETH K. HUSSEY, PHARMD 2 3 injection + RE placebo (prandial insulin), ANNIE A. MAVIAN, MD DEREK J. NUNEZ, MD 3 3 1,2 ) placebo insulin injection + 50 mg RE (RE ROBIN O’CONNOR-SEMMES, PHD ROBERT R. HENRY, MD 3 3 50 mg), 4) placebo insulin injection + 150 PATRICIA K. MYDLOW, BS ROBERT L. DOBBINS, MD, PHD mg RE (RE 150 mg), and 5) placebo insulin injection + 500 mg RE (RE 500 mg). d fl Each individual received 75-g oral OBJECTIVES Remogli ozin etabonate (RE), an inhibitor of the sodium-glucose transporter glucose and identical meals during all 2, improves glucose profiles in type 2 diabetes. This study assessed safety, tolerability, pharma- cokinetics, and pharmacodynamics of RE in subjects with type 1 diabetes. treatment periods. Frequent samples were obtained for measurement of plasma RESEARCH DESIGN AND METHODSdTen subjects managed with continuous sub- glucose and insulin concentrations. Urine cutaneous insulin infusion were enrolled. In addition to basal insulin, subjects received five samples were collected for 24 h to assess randomized treatments: placebo, prandial insulin, 50 mg RE, 150 mg RE, and mg RE 500. creatinine clearance and glucose excre- d tion. Plasma samples were collected for RESULTS Adverse events and incidence of hypoglycemia with RE did not differ from placebo fl and prandial insulin groups. -
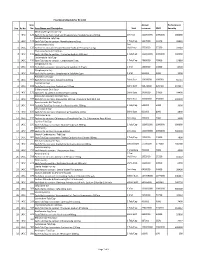
Sno. Rc No Item No. Item Name and Description Unit Annual Turnover
Final Drug Schedule for RC 145C Item Annual Performance Sno. Rc No No. Item Name and Description Unit turnover EMD Security Medroxy Progesterone Inj- 1 145C 126 Each ml to contain: Medroxy Progesterone Acetate Suspn.150mg. 1ml Vial 100000000 1000000 600000 Norethisterone Tab/Cap- 2 145C 128 Each Tab/Cap to contain: Norethisterone 5mg. 1 Tab/Cap 5637000 56370 33822 Dexamethasone Inj- 2ml 3 145C 133 Each ml to contain: Dexamethasone Sodium Phosphate 4 mg. Vial/Amp 5735000 57350 34410 Thyroxine Sodium Tab/Cap- 4 145C 135 Each Tab/Cap to contain: Thyroxine Sodium 100 mcg 1 Tab/Cap 100000000 1000000 600000 Carbimazole Tab/Cap- 5 145C 136 Each Tab/Cap to contain: Carbimazole 5 mg. 1 Tab/Cap 2980000 29800 17880 Streptomycin Inj- 6 145C 151 Each Vial to contain: Streptomycin Sulphate 0.75gm 1 Vial 1400000 14000 8400 Streptomycin Inj- 7 145C 152 Each Vial to contain: Streptomycin Sulphate 1gm 1 Vial 500000 5000 3000 Ampicillin Dry Syp- 8 145C 161 Each 5ml to contain: Ampicillin 125mg 30ml Bott 10959000 109590 65754 Cephalexin Syp- 9 145C 168 Each 5ml to contain: Cephalexin 125mg 30ml Bott 32874000 328740 197244 Erthyromycin Oral Susp- 10 145C 172 Each 5ml to contain: Erythromycin 125mg 30ml Bott 2400000 24000 14400 Amoxy & Clavulanic Acid Dry Syp- 11 145C 183 Each 5ml to contain: Amoxycillin 200 mg, Clavulanic Acid 28.5 mg 30ml Bott 35000000 350000 210000 Pyrazinamide Kid Tab/Cap- 12 145C 191 Each Kid Tab/Cap to contain: Pyrazinamide 300mg 1 Tab/Cap 500000 5000 3000 Chloroquine Syp- 13 145C 196 Each 5ml to contain: Chloroquine Phosphate 50mg. -

An Emerging Protagonist: Sodium Glucose Co-Transporters (Sglts)
abetes & Di M f e o t a l b a o Ahmed et al., J Diabetes Metab 2014, 5:4 n l r i s u m o DOI: 10.4172/2155-6156.1000358 J Journal of Diabetes and Metabolism ISSN: 2155-6156 Review Article Open Access An Emerging Protagonist: Sodium Glucose Co-transporters (SGLTs) as a Burgeoning Target for the Treatment of Diabetes Mellitus Danish Ahmed1*, Manju Sharma2, Vikas Kumar1 and Yadav Pankajkumar Subhashchandra1 1Department of Pharmaceutical Sciences, Sam Higginbottom Institute of Agriculture, Technology and Sciences (SHIATS)-Deemed University, India 2Department of Pharmacology, JamiaHamdard University, Hamdard Nagar, New Delhi, India Abstract Contemporary therapies to rationalize the hyperglycaemia in type 2 diabetes mellitus (T2DM) generally involve insulin-dependent mechanisms and lose their effectiveness as pancreatic b-cell function decreases to a greater extent. Kidney emerges out as a novel and potential target to trim down the complications of T2DM. The filtered glucose is reabsorbed principally through the sodium glucose co-transporter-2 (SGLT2), a low affinity transport system, which are present at the luminal surface cells that covers the first segment of proximal tubules. Competitive inhibition of SGLT2 therefore represents an innovative therapeutic strategy for the treatment of hyperglycaemia and/or obesity in patients with type 1 or type 2 diabetes by enhancing glucose and energy loss through the urine. Selective inhibitors of SGLT2 reduce glucose reabsorption, causing excess glucose to be eliminated in the urine; this decreases plasma glucose. SGLT2 inhibitors are coupled with osmotic dieresis and loss of weight which aid in reducing the blood pressure. The observation that individuals with familial renal glycosuria maintain normal long- term kidney function provides some encouragement that this mode of action will not adversely affect renal function. -

A Review on Evolution in Triglyceride Determination
Available online at www.derpharmachemica.com ISSN 0975-413X Der Pharma Chemica, 2018, 10(5): 84-88 CODEN (USA): PCHHAX (http://www.derpharmachemica.com/archive.html) Sodium-glucose co-transporter 2 (SGLT2) Inhibitors: New Target for Type 2 Diabetes Mellitus (T2DM) Review Swapna Vadlamani* Asst. proffesor, NIPER, Hyderabad, Andhra Pradesh, India ABSTRACT Introduction: Knocking out type2 diabetes by new insulin independent renal glucose transporters as targets, reducing the side effects related to high rise in glucose levels is a more efficient way to manage diabetes. Sodium-glucose co-transporter 2 (SGLT2) inhibitors block reabsorption of glucose back into the blood and stimulate secretion in urine in a way controlling blood glucose levels. Areas discussed: We emphasize in this review an overview of type 2 diabetes. New insulin independent targets, SGLT family inhibitors and their mechanism of action are briefly discussed. Molecular modeling studies carried out for new analogues of SGLT2 were indicated and also about current marketed SGLT drugs their safety issues are briefly outlined. Conclusion: SGLT2 inhibitors are very promising drugs for near future, where insulin sensitization is a problem. A combination of drugs related to insulin dependent pathway and also independent pathway like SGLT2/SGLT1 drugs will be more effective in glycemic control with lesser side effects. Keywords: Type II Diabetes, SGLT2 inhibitors INTRODUCTION Present scenario of food habits and absolutely very less physical activity is becoming the major cause for obesity finally leading to diabetes. Diabetes is said to be a rich man disease and mostly occurs at the later age of 40, but now one in every 5 persons at early age diagnosed turned to be diabetic mainly because of lifestyle changes. -

Exploring Glycosuria As a Mechanism for Weight and Fat Mass Reduction. a Pilot Study with Remogliflozin Etabonate and Sergliflozin Etabonate in Healthy Obese Subjects
Journal of Clinical & Translational Endocrinology 1 (2014) e3ee8 Contents lists available at ScienceDirect Journal of Clinical & Translational Endocrinology journal homepage: www.elsevier.com/locate/jcte Research Paper Exploring glycosuria as a mechanism for weight and fat mass reduction. A pilot study with remogliflozin etabonate and sergliflozin etabonate in healthy obese subjects Antonella Napolitano a,*, Sam Miller a, Peter R. Murgatroyd c, Elizabeth Hussey b, Robert L. Dobbins b, Edward T. Bullmore a, Derek J.R. Nunez b View metadata, citation and similar papers at core.ac.uk brought to you by CORE a Clinical Unit in Cambridge, GlaxoSmithKline, Addenbrookes Hospital, Cambridge, UK b Metabolic Pathways and Cardiovascular Unit, GlaxoSmithKline,provided by NC, Elsevier USA - Publisher Connector c Wellcome Trust Clinical Research Facility, Cambridge University Hospital NHS Trust, UK article info abstract Article history: Inhibitors of sodium-dependent glucose co-transporter 2 (SGLT2) increase glucose excretion in the urine Received 16 October 2013 and improve blood glucose in Type 2 diabetes mellitus. Glycosuria provides an energy and osmotic drain Received in revised form that could alter body composition. We therefore conducted a pilot study comparing the effects on body 26 November 2013 composition of two SGLT2 inhibitors, remogliflozin etabonate (RE) 250 mg TID (n ¼ 9) and sergliflozin Accepted 5 December 2013 etabonate (SE) (1000 mg TID) (n ¼ 9), with placebo (n ¼ 12) in obese non-diabetic subjects. Both drugs Available online 7 February 2014 were well tolerated during 8 weeks of dosing, and the most common adverse event was headache. No urinary tract infections were observed, but there was one case of vaginal candidiasis in the RE group. -

(SGLT2) Inhibitors: a Systematic Review and Meta-Analysis
Open access Research BMJ Open: first published as 10.1136/bmjopen-2018-022577 on 1 February 2019. Downloaded from Comparative safety of the sodium glucose co-transporter 2 (SGLT2) inhibitors: a systematic review and meta-analysis Jennifer R Donnan,1 Catherine A Grandy,1 Eugene Chibrikov,1 Carlo A Marra,1,2 Kris Aubrey-Bassler,3 Karissa Johnston,1 Michelle Swab,3 Jenna Hache,1 Daniel Curnew,1 Hai Nguyen,1 John-Michael Gamble1,4 To cite: Donnan JR, Grandy CA, ABSTRACT Strengths and limitations of this study Chibrikov E, et al. Comparative Objective To estimate the association between the use safety of the sodium glucose of sodium glucose co-transporter-2 (SGLT2) inhibitors ► This study provides a comprehensive systematic co-transporter 2 (SGLT2) and postmarket harms as identified by drug regulatory inhibitors: a systematic review review of potential serious adverse events related agencies. and meta-analysis. BMJ Open to use of sodium glucose co-transporter-2 (SGLT2) Design We conducted a systematic review and meta- 2019;9:e022577. doi:10.1136/ inhibitors identified by drug regulatory agencies. analysis of randomised controlled trials (RCT). Six large bmjopen-2018-022577 ► This study considered select outcomes to provide databases were searched from inception to May 2018. focused attention on the issues concerning regula- ► Prepublication history and Random effects models were used to estimate pooled tors; however, this means that additional knowledge additional material for this relative risks (RRs). paper are available online. To of the clinical benefits and harms needs to be con- Intervention SGLT2 inhibitors, compared with placebo or view these files, please visit sidered before applying the results of this study. -

Efficacy and Safety of Remogliflozin Etabonate, a New Sodium Glucose
Drugs (2020) 80:587–600 https://doi.org/10.1007/s40265-020-01285-0 ORIGINAL RESEARCH ARTICLE Efcacy and Safety of Remoglifozin Etabonate, a New Sodium Glucose Co‑Transporter‑2 Inhibitor, in Patients with Type 2 Diabetes Mellitus: A 24‑Week, Randomized, Double‑Blind, Active‑Controlled Trial Mala Dharmalingam1 · S. R. Aravind2 · Hemant Thacker3 · S. Paramesh4 · Brij Mohan5 · Manoj Chawla6 · Arthur Asirvatham7 · Ramesh Goyal8 · Jayashri Shembalkar9 · R. Balamurugan10 · Pradnya Kadam11 · Hansraj Alva12 · Rahul Kodgule13 · Monika Tandon13 · Sivakumar Vaidyanathan13 · Amol Pendse13 · Rajesh Gaikwad13 · Sagar Katare13 · Sachin Suryawanshi13 · Hanmant Barkate13 Published online: 11 March 2020 © The Author(s) 2020 Abstract Background Metformin is the frst-line treatment for type 2 diabetes mellitus (T2DM), but many patients either cannot tolerate it or cannot achieve glycemic control with metformin alone, so treatment with other glucose-lowering agents in combination with metformin is frequently required. Remoglifozin etabonate, a novel agent, is an orally bioavailable prodrug of remoglifozin, which is a potent and selective sodium-glucose co-transporter-2 inhibitor. Objective Our objective was to evaluate the efcacy and safety of remoglifozin etabonate compared with dapaglifozin in subjects with T2DM in whom a stable dose of metformin as monotherapy was providing inadequate glycemic control. Methods A 24-week randomized, double-blind, double-dummy, active-controlled, three-arm, parallel-group, multi- center, phase III study was conducted in India. Patients aged ≥ 18 and ≤ 65 years diagnosed with T2DM, receiving met- formin ≥ 1500 mg/day, and with glycated hemoglobin (HbA1c) levels ≥ 7 to ≤ 10% at screening were randomized into three groups. Every patient received metformin ≥ 1500 mg and either remoglifozin etabonate 100 mg twice daily (BID) (group 1, n = 225) or remoglifozin etabonate 250 mg BID (group 2, n = 241) or dapaglifozin 10 mg once daily (QD) in the morning and placebo QD in the evening (group 3, n = 146). -

Canagliflozin, Dapagliflozin and Empagliflozin Monotherapy for Treating Type 2 Diabetes: Systematic Review and Economic Evaluation
HEALTH TECHNOLOGY ASSESSMENT VOLUME 21 ISSUE 2 JANUARY 2017 ISSN 1366-5278 Canagliflozin, dapagliflozin and empagliflozin monotherapy for treating type 2 diabetes: systematic review and economic evaluation Rhona Johnston, Olalekan Uthman, Ewen Cummins, Christine Clar, Pamela Royle, Jill Colquitt, Bee Kang Tan, Andrew Clegg, Saran Shantikumar, Rachel Court, J Paul O’Hare, David McGrane, Tim Holt and Norman Waugh DOI 10.3310/hta21020 Canagliflozin, dapagliflozin and empagliflozin monotherapy for treating type 2 diabetes: systematic review and economic evaluation Rhona Johnston,1 Olalekan Uthman,2 Ewen Cummins,1 Christine Clar,3 Pamela Royle,2 Jill Colquitt,4 Bee Kang Tan,2 Andrew Clegg,5 Saran Shantikumar,2 Rachel Court,2 J Paul O’Hare,2 David McGrane,6 Tim Holt7 and Norman Waugh2* 1McMDC, Harrogate, UK 2Warwick Evidence, Division of Health Sciences, University of Warwick, Coventry, UK 3Berlin, Germany 4Effective Evidence, Waterlooville, UK 5University of Central Lancashire, Preston, UK 6Queen Elizabeth University Hospital, Glasgow, UK 7University of Oxford, Oxford, UK *Corresponding author Declared competing interests of authors: David McGrane has spoken at educational meetings sponsored by AstraZeneca, Eli Lilly, Sanofi, MSD, Takeda Pharmaceutical Company, Novo Nordisk, Janssen, and has served on Advisory Boards for Eli Lilly, Sanofi, Novo Nordisk. J Paul O’Hare has received lecture fees, advisory board meeting fees, and grants for research from Novo Nordisk and Sanofi. All fees are paid through University of Warwick to fund access to insulin projects in sub-Saharan Africa. Published January 2017 DOI: 10.3310/hta21020 This report should be referenced as follows: Johnston R, Uthman O, Cummins E, Clar C, Royle P, Colquitt J, et al.