Tramadol - SAFE PRESCRIBING - Consider the Risks! 1
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Tramadol (Ultram)
TRAMADOL (ULTRAM) Tramadol is FDA approved for the treatment of musculoskeletal pain. Studies have shown it is useful in treating the pain associated with diabetic neuropathy and other pain conditions. Tramadol comes in 50 mg tablets. The maximum dose is two tablets four times per day unless your kidney function is below normal or you are over 75 years old, in which case the maximum dose is two tablets three times per day. The main side effects of Tramadol are drowsiness, sedation, and stomach upset, all of which are minimized by slowly raising the dose. About 5% of patients have stomach upset at any dose of Tramadol and cannot take the medicine. Other risks include seizures (occur in less than 1/100,000 and are more likely if you have seizures) and possibly abuse (relevant if you have abused drugs in the past). Tramadol should be started at a low dose and raise the dose slowly toward the maximum dose. Start with one tablet at bedtime. After 3 - 7 days, increase to one tablet twice daily (morning and bedtime). After an additional 3 - 7 days, increase to one tablet three times per day (morning, noon, and bedtime). After an additional 3 - 7 days, increase to one tablet four times per day (1 tablet with each meal and 1 at bedtime). At that point, the dose may be increased or adjusted depending on how you are doing. To increase further, you will: Add a second tablet at bedtime (one tablet three times per day and two tablets at bedtime). After 3 - 7 days, add a second tablet to another dose (one tablet twice per day and two tablets twice per day). -

Effects of Prophylactic Ketamine and Pethidine to Control Postanesthetic Shivering: a Comparative Study
Biomedical Research and Therapy, 5(12):2898-2903 Original Research Effects of prophylactic ketamine and pethidine to control postanesthetic shivering: A comparative study Masoum Khoshfetrat1, Ali Rosom Jalali2, Gholamreza Komeili3, Aliakbar Keykha4;∗ ABSTRACT Background: Shivering is an undesirable complication following general anesthesia and spinal anesthesia, whose early control can reduce postoperative metabolic and respiratory complications. Therefore, this study aims to compare the effects of prophylactic injection of ketamine and pethi- dine on postoperative shivering.Methods: This double-blind clinical trial was performed on 105 patients with short-term orthopedic and ENT surgery. The patients were randomly divided into three groups; 20 minutes before the end of the surgery, 0.4 mg/kg of pethidine was injected to the first group, 0.5 mg/kg of ketamine was injected to the second group, and normal saline was injected to the third group. After the surgery, the tympanic membrane temperature was measured at 0, 10, 20, and 30 minutes. The shivering was also measured by a four-point grading from zero (no shiv- ering) to four (severe shivering). Data were analyzed by one-way ANOVA, Kruskal Wallis, Chi-square 1Doctor of Medicine (MD), Fellow of and Pearson correlation. Results: The mean age of patients was 35.811.45 years in the ketamine Critical Care Medicine (FCCM), group, 34.811.64 years in the normal saline group, and 33.1110.5 years in the pethidine group. Department of Anesthesiology and The one-way ANOVA showed no significant difference in the mean age between the three groups Critical Care, Khatam-Al-Anbiya (P=0.645). -

Current Awareness in Clinical Toxicology Editors: Damian Ballam Msc and Allister Vale MD
Current Awareness in Clinical Toxicology Editors: Damian Ballam MSc and Allister Vale MD February 2016 CONTENTS General Toxicology 9 Metals 38 Management 21 Pesticides 41 Drugs 23 Chemical Warfare 42 Chemical Incidents & 32 Plants 43 Pollution Chemicals 33 Animals 43 CURRENT AWARENESS PAPERS OF THE MONTH How toxic is ibogaine? Litjens RPW, Brunt TM. Clin Toxicol 2016; online early: doi: 10.3109/15563650.2016.1138226: Context Ibogaine is a psychoactive indole alkaloid found in the African rainforest shrub Tabernanthe Iboga. It is unlicensed but used in the treatment of drug and alcohol addiction. However, reports of ibogaine's toxicity are cause for concern. Objectives To review ibogaine's pharmacokinetics and pharmacodynamics, mechanisms of action and reported toxicity. Methods A search of the literature available on PubMed was done, using the keywords "ibogaine" and "noribogaine". The search criteria were "mechanism of action", "pharmacokinetics", "pharmacodynamics", "neurotransmitters", "toxicology", "toxicity", "cardiac", "neurotoxic", "human data", "animal data", "addiction", "anti-addictive", "withdrawal", "death" and "fatalities". The searches identified 382 unique references, of which 156 involved human data. Further research revealed 14 detailed toxicological case reports. Current Awareness in Clinical Toxicology is produced monthly for the American Academy of Clinical Toxicology by the Birmingham Unit of the UK National Poisons Information Service, with contributions from the Cardiff, Edinburgh, and Newcastle Units. The NPIS is commissioned by Public Health England Current Awareness in Clinical Toxicology Editors: Damian Ballam MSc and Allister Vale MD February 2016 Current Awareness in Clinical Toxicology is produced monthly for the American Academy of Clinical Toxicology by the Birmingham Unit of the UK National Poisons Information Service, with contributions from the Cardiff, Edinburgh, and Newcastle Units. -

Quantitative Drug Test Menu Section 2
1 Guthrie Square, Sayre, PA 18840 Bill To: Client GMG Toxicology Laboratory Requisition Toll Free Phone (844) 617-4719 Insurance Request Date: _____/______/______ Medical Director: Hani Hojjati, MD Fax (570) 887-4729 Patient PATIENT INFORMATION (PLEASE PRINT IN BLACK INK) INSURANCE BILLING INFORMATION (PLEASE PRINT IN BLACK INK) Pt Last Name First M I PRIMARY Medicare Medicaid Other Ins. Self Spouse Child __ Subscriber Last Name First M Address Birth Date Sex M F Beneficiary/Member # Group # City Pt. SS# or MRN Claims Name and Address City ST ZIP ST ZIP Home Phone (Attach a copy of the patient's insurance card and information) SECONDARY Medicare Medicaid Other Ins. Self Spouse Child Employer Work Phone Subscriber Last Name First M Work Address City ST ZIP Beneficiary/Member # Group # __ CLIENT INFORMATION - REFERRING PHYSICIAN Claims Name and Address City ST ZIP Client Address: (Atttach a copy of the patient's insurance card and information) COLLECTION / REPORTING INFORMATION Copy to: FAX Results to __ CALL Results to Phone: Fax: Date Collected: Time Collected: AM PM Specimen Type: Urine Saliva Other ___________________ Physician Signature (legible - No Stamp) For Lab Use Only (Required for Medicare & Medicaid patient orders) Signed ABN Obtained Place Lab Label Here Contact Laboratory Medical Director (570-887-4719) with questions concerning medical necessity PHYSICIAN When ordering tests, the physician is required to make an independent medical necessity decision with regard to each test thelaboratory will bill. The physician also understands he or she is required NOTICE to (1) submit ICD-10 diagnosis supported in the patient's medical record as documentation of the medical necessity or (2) explain and have the patient sign an ABN. -

CAN YOU TAKE TRAMADOL with NEFOPAM Can You Take Tramadol with Nefopam
CAN YOU TAKE TRAMADOL WITH NEFOPAM can you take tramadol with nefopam tramadol 37 5 vs percocet 5 325 ultram tramadol pictures tramadol hcl tabs 50 mg tramadol 200 mg recreational drugs and heart can tramadol and percocet be mixed hbs robaxin tramadol interaction generic tramadol 319 immediate release how long tramadol stay in your urine does tramadol make you sleepy or awake tramadol acetaminophen\/codeine 120 12mg sol b tracert ex tramadol dosage for adults meloxicam/tramadol/amitriptyline/lidocaine/prilocaine apo tramadol high feeling on hydrocodone tramadol apteka internetowa olmed order tramadol/paracetamol from mexico tramadol quizlet flashcards microbiology tramadol has mu opioid agonist activity director jobs tramadol met ritalin sr strengths hur ta tramadol withdrawal in dogs tramadol te gebruiken bij tramadol dosis cachorros bulldog 2015 100mg tramadol 10mg hydrocodone images 100 tramadol termasuk jenis obat apa acyclovir side how to get rid of a tramadol high 200 ml tramadol withdrawal timeline drug interactions between percocet and tramadol comparison tramadol e morfina presentacion de tres can tramadol be taken with paracetamol indication and action tramadol review article template with photos tramadol codeine allergy rash best price tramadol online tramadol 93 58 dosage for ibuprofen tramadol v oxycodone pill colors can tramadol make you drowsy doll b tracert ex tramadol addiction withdrawal tramadol instant release oxycontin pictures can you drink wine with tramadol i can function tramadol hydrochloride sleepy tramadol cva -

Parkinson's Disease Fact Sheet
Parkinson’s Disease Fact Sheet About Parkinson’s Disease Parkinson’s disease is a progressive, incurable neurological disorder associated with a loss of dopamine-generating cells in the brain. It is primarily associated with progressive loss of motor control, but it results in a complex array of symptoms, including many non-motor symptoms. Parkinson’s impacts an estimated one million people in the United States. Critical Clinical Care Considerations • To avoid serious side effects, Parkinson’s patients need their medications on time, every time — do not skip or postpone doses. • Write down the exact times of day medications are to be administered so that doses are given on the same schedule the patient follows at home. • Do not substitute Parkinson’s medications or stop levodopa therapy abruptly. • Resume medications immediately following procedures, unless vomiting or severely incapacitated. • If an antipsychotic is necessary, use pimavanserin (Nuplazid), quetiapine (Seroquel) or clozapine (Clozaril). • Be alert for symptoms of dysphagia (trouble swallowing) and risk of pneumonia. • Ambulate as soon as medically safe. Patients may require assistance. Common Symptoms of Parkinson’s Disease Motor Non-Motor • Shaking or tremor at rest • Depression • Bradykinesia or freezing (being stuck • Anxiety in place when attempting to walk) • Constipation • Low voice volume or muffled speech • Cognitive decline and dementia • Lack of facial expression • Impulse control disorders • Stiffness or rigidity of the arms, legs • Orthostatic hypotension or -
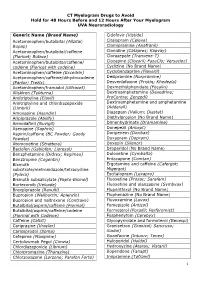
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology Generic Name (Brand Name) Cidofovir (Vistide) Acetaminophen/butalbital (Allzital; Citalopram (Celexa) Bupap) Clomipramine (Anafranil) Acetaminophen/butalbital/caffeine Clonidine (Catapres; Kapvay) (Fioricet; Butace) Clorazepate (Tranxene-T) Acetaminophen/butalbital/caffeine/ Clozapine (Clozaril; FazaClo; Versacloz) codeine (Fioricet with codeine) Cyclizine (No Brand Name) Acetaminophen/caffeine (Excedrin) Cyclobenzaprine (Flexeril) Acetaminophen/caffeine/dihydrocodeine Desipramine (Norpramine) (Panlor; Trezix) Desvenlafaxine (Pristiq; Khedezla) Acetaminophen/tramadol (Ultracet) Dexmethylphenidate (Focalin) Aliskiren (Tekturna) Dextroamphetamine (Dexedrine; Amitriptyline (Elavil) ProCentra; Zenzedi) Amitriptyline and chlordiazepoxide Dextroamphetamine and amphetamine (Limbril) (Adderall) Amoxapine (Asendin) Diazepam (Valium; Diastat) Aripiprazole (Abilify) Diethylpropion (No Brand Name) Armodafinil (Nuvigil) Dimenhydrinate (Dramamine) Asenapine (Saphris) Donepezil (Aricept) Aspirin/caffeine (BC Powder; Goody Doripenem (Doribax) Powder) Doxapram (Dopram) Atomoxetine (Strattera) Doxepin (Silenor) Baclofen (Gablofen; Lioresal) Droperidol (No Brand Name) Benzphetamine (Didrex; Regimex) Duloxetine (Cymbalta) Benztropine (Cogentin) Entacapone (Comtan) Bismuth Ergotamine and caffeine (Cafergot; subcitrate/metronidazole/tetracycline Migergot) (Pylera) Escitalopram (Lexapro) Bismuth subsalicylate (Pepto-Bismol) Fluoxetine (Prozac; Sarafem) -

Tramadol 50 Mg Capsules Tramadol Hydrochloride
Tramadol 50 mg Capsules Tramadol hydrochloride PACKAGE LEAFLET: INFORMATION FOR THE USER Read all of this leaflet carefully before you start taking this medicine. • Keep this leaflet. You may need to read it again. • If you have any further questions, ask your doctor or pharmacist. • This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours. • If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist. In this leaflet 1. What are Tramadol 50 mg Capsules and what are they used for 2. What you need to know before you take Tramadol 50 mg Capsules 3. How to take Tramadol 50 mg Capsules 4. Possible side effects 5. How to store Tramadol 50 mg Capsules 6. Contents of the pack and other information 1. WHAT ARE TRAMADOL 50 mg CAPSULES AND WHAT ARE THEY USED FOR Tramadol 50 mg Capsules are 'analgesics' which act on the central nervous system (the brain and the spinal cord). Analgesics are often called 'pain killers' or 'pain relievers'. Tramadol 50 mg Capsules relieve pain and can also be taken to prevent pain. Pain is a symptom not an illness. There are many types of pain with many different causes, for example back- ache, toothache, pain after an operation or pain from broken bones. Tramadol 50 mg Capsules help your body's system for relieving pain. It does this in two ways: - Acts directly on parts of your brain and spinal cord to reduce the amount of pain you feel - Reduces the size of the pain message passed from one nerve to another. -

Tramadol: a Review of Its Use in Perioperative Pain
Drugs 2000 Jul; 60 (1): 139-176 ADIS DRUG EVA L U AT I O N 0012-6667/00/0007-0139/$25.00/0 © Adis International Limited. All rights reserved. Tramadol A Review of its Use in Perioperative Pain Lesley J. Scott and Caroline M. Perry Adis International Limited, Auckland, New Zealand Various sections of the manuscript reviewed by: S.R. Abel, Indiana University Medical Centre, Indianapolis, Indiana, USA; S.S. Bloomfield, University of Cincinnati, Cincinnati, Ohio, USA; J.E. Caldwell, Department of Anesthesia, University of California, San Francisco, California, USA; K.A. Lehmann, Institute of Anaesthesiology and Intensive Care Medicine, University of Cologne, Cologne, Germany; L. Radbruch, Institute of Anaesthesiology and Intensive Care Medicine, University of Cologne, Cologne, Germany; K. Szymanski, Indiana University Medical Centre, Indianapolis, Indiana, USA; P. Tarkkila, Department of Anaesthesia, Helsinki University Centre Hospital, Helsinki, Finland; C.H. Wilder-Smith, Gastrointestinal Unit and Nociception Research Group, Berne, Switzerland. Data Selection Sources: Medical literature published in any language since 1993 on tramadol, identified using AdisBase (a proprietary database of Adis International, Auckland, New Zealand) and Medline. Additional references were identified from the reference lists of published articles. Bibliographical information, including contributory unpublished data, was also requested from the company developing the drug. Search strategy: AdisBase search terms were ‘tramadol’ or ‘CG-315’ or ‘CG-315E’ or ‘U-26225A’. Medline search terms were ‘tramadol’. Searches were last updated 26 May 2000. Selection: Studies in patients with perioperative moderate to severe pain who received tramadol. Inclusion of studies was based mainly on the methods section of the trials. When available, large, well controlled trials with appropriate statistical methodology were preferred. -

CONSUMER MEDICINE INFORMATION Arrow
CONSUMER MEDICINE INFORMATION Arrow - Tramadol Tramadol 50 mg capsules What is in this leaflet This leaflet answers some common questions about ARROW - TRAMADOL. It does not contain all of the available information. It does not take the place of talking to your doctor or pharmacist. All medicines have benefits and risks. Your doctor has weighed the risks of you taking ARROW – TRAMADOL against the benefits they expect it will have for you. If you have any concerns about taking this medicine, talk to your doctor or pharmacist. Keep this leaflet with your medicine. You may need to read it again. What ARROW - TRAMADOL is used for ARROW - TRAMADOL is used to relieve moderate to severe pain and belongs to a group of medicines called analgesics (pain relievers). Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason. ARROW – TRAMADOL is available only with a doctor’s prescription. Before you take ARROW - TRAMADOL When you must not use it Do not take ARROW – TRAMADOL if • you have a known allergy to ARROW - TRAMADOL or other opioids or any of the ingredients listed at the end of this leaflet. Some of the symptoms of an allergic reaction may include skin rash, itching, difficulty breathing and swelling of the face (including lips, tongue, throat etc) • you have respiratory disease or slow or shallow breathing • you have taken large amounts of alcohol or other substances which can affect your level of consciousness. Some examples of these substances include sleeping pills, pain relievers or other psychotropic medicines (medicines that affect mood and emotions) • you are taking medicine for depression containing a "monoamine oxidase inhibitor" (such as Nardil, Parnate), or have taken one within the past two weeks. -

How to Provide Pain Relief for Laminitis in the Field
HOW-TO SESSION: FIELD ANESTHESIA AND PAIN MANAGEMENT How to Provide Pain Relief for Laminitis in the Field Alonso Guedes, DVM, MS, PhD Author’s address: University of California, Davis, One Shields Avenue, Davis, CA 95616; e-mail: [email protected]. © 2013 AAEP. 1. Introduction characterized by hyperalgesia (ie, exaggerated re- sponse to a painful stimulus) and allodynia (ie, pain Laminitis is an extremely painful condition with 6,13–15 still incompletely understood pathophysiology.1–3 response to a normally nonpainful stimulus). All too often, the uncontainable severe pain associ- The goal of this report is to present techniques tar- ated with the disease is the single most common geting such pathologic pain states that can be used reason for euthanasia of laminitic horses. Treat- to manage pain and suffering in horses with ment is frequently unfinished because owners and laminitis. health providers feel forced to end the extreme pain and suffering by ending the life of the afflicted ani- 2. Recommended Agents and Routes of mal. More positive treatment results could be ob- Administration tained if pain and suffering could be successfully Tramadol tablets can be crushed into powder, mixed modulated.4 Non-steroidal anti-inflammatory with syrup or molasses, and administered orally at a drugs (NSAIDs) such as flunixin meglumine and dose of 5 mg/kg q 12 hours.6 phenylbutazone have long been and still remain the Ketamine can be administered intravenously as a primary pharmacologic agents used to treat pain constant rate infusion at a dose of 0.6 mg/kg per and inflammation in horses with laminitis.5 hour for 6 hours/day for 3 to 5 days (or longer as 6 Tramadol is emerging as an additional option to- needed) through a syringe pump, or it can be di- ward providing multimodal analgesia in equine luted in 0.9% saline and dripped by gravity. -

Medications to Be Avoided Or Used with Caution in Parkinson's Disease
Medications To Be Avoided Or Used With Caution in Parkinson’s Disease This medication list is not intended to be complete and additional brand names may be found for each medication. Every patient is different and you may need to take one of these medications despite caution against it. Please discuss your particular situation with your physician and do not stop any medication that you are currently taking without first seeking advice from your physician. Most medications should be tapered off and not stopped suddenly. Although you may not be taking these medications at home, one of these medications may be introduced while hospitalized. If a hospitalization is planned, please have your neurologist contact your treating physician in the hospital to advise which medications should be avoided. Medications to be avoided or used with caution in combination with Selegiline HCL (Eldepryl®, Deprenyl®, Zelapar®), Rasagiline (Azilect®) and Safinamide (Xadago®) Medication Type Medication Name Brand Name Narcotics/Analgesics Meperidine Demerol® Tramadol Ultram® Methadone Dolophine® Propoxyphene Darvon® Antidepressants St. John’s Wort Several Brands Muscle Relaxants Cyclobenzaprine Flexeril® Cough Suppressants Dextromethorphan Robitussin® products, other brands — found as an ingredient in various cough and cold medications Decongestants/Stimulants Pseudoephedrine Sudafed® products, other Phenylephrine brands — found as an ingredient Ephedrine in various cold and allergy medications Other medications Linezolid (antibiotic) Zyvox® that inhibit Monoamine oxidase Phenelzine Nardil® Tranylcypromine Parnate® Isocarboxazid Marplan® Note: Additional medications are cautioned against in people taking Monoamine oxidase inhibitors (MAOI), including other opioids (beyond what is mentioned in the chart above), most classes of antidepressants and other stimulants (beyond what is mentioned in the chart above).