Ethics of Vaccine Research
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
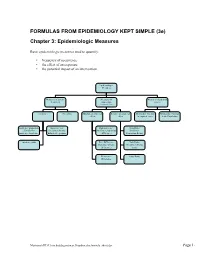
FORMULAS from EPIDEMIOLOGY KEPT SIMPLE (3E) Chapter 3: Epidemiologic Measures
FORMULAS FROM EPIDEMIOLOGY KEPT SIMPLE (3e) Chapter 3: Epidemiologic Measures Basic epidemiologic measures used to quantify: • frequency of occurrence • the effect of an exposure • the potential impact of an intervention. Epidemiologic Measures Measures of disease Measures of Measures of potential frequency association impact (“Measures of Effect”) Incidence Prevalence Absolute measures of Relative measures of Attributable Fraction Attributable Fraction effect effect in exposed cases in the Population Incidence proportion Incidence rate Risk difference Risk Ratio (Cumulative (incidence density, (Incidence proportion (Incidence Incidence, Incidence hazard rate, person- difference) Proportion Ratio) Risk) time rate) Incidence odds Rate Difference Rate Ratio (Incidence density (Incidence density difference) ratio) Prevalence Odds Ratio Difference Macintosh HD:Users:buddygerstman:Dropbox:eks:formula_sheet.doc Page 1 of 7 3.1 Measures of Disease Frequency No. of onsets Incidence Proportion = No. at risk at beginning of follow-up • Also called risk, average risk, and cumulative incidence. • Can be measured in cohorts (closed populations) only. • Requires follow-up of individuals. No. of onsets Incidence Rate = ∑person-time • Also called incidence density and average hazard. • When disease is rare (incidence proportion < 5%), incidence rate ≈ incidence proportion. • In cohorts (closed populations), it is best to sum individual person-time longitudinally. It can also be estimated as Σperson-time ≈ (average population size) × (duration of follow-up). Actuarial adjustments may be needed when the disease outcome is not rare. • In an open populations, Σperson-time ≈ (average population size) × (duration of follow-up). Examples of incidence rates in open populations include: births Crude birth rate (per m) = × m mid-year population size deaths Crude mortality rate (per m) = × m mid-year population size deaths < 1 year of age Infant mortality rate (per m) = × m live births No. -

Statistical Guidance on Reporting Results from Studies Evaluating Diagnostic Tests Document Issued On: March 13, 2007
Guidance for Industry and FDA Staff Statistical Guidance on Reporting Results from Studies Evaluating Diagnostic Tests Document issued on: March 13, 2007 The draft of this document was issued on March 12, 2003. For questions regarding this document, contact Kristen Meier at 240-276-3060, or send an e-mail to [email protected]. U.S. Department of Health and Human Services Food and Drug Administration Center for Devices and Radiological Health Diagnostic Devices Branch Division of Biostatistics Office of Surveillance and Biometrics Contains Nonbinding Recommendations Preface Public Comment Written comments and suggestions may be submitted at any time for Agency consideration to the Division of Dockets Management, Food and Drug Administration, 5630 Fishers Lane, Room 1061, (HFA-305), Rockville, MD, 20852. Alternatively, electronic comments may be submitted to http://www.fda.gov/dockets/ecomments. When submitting comments, please refer to Docket No. 2003D-0044. Comments may not be acted upon by the Agency until the document is next revised or updated. Additional Copies Additional copies are available from the Internet at: http://www.fda.gov/cdrh/osb/guidance/1620.pdf. You may also send an e-mail request to [email protected] to receive an electronic copy of the guidance or send a fax request to 240-276-3151 to receive a hard copy. Please use the document number 1620 to identify the guidance you are requesting. Contains Nonbinding Recommendations Table of Contents 1. Background....................................................................................................4 -

Estimated HIV Incidence and Prevalence in the United States
Volume 26, Number 1 Estimated HIV Incidence and Prevalence in the United States, 2015–2019 This issue of the HIV Surveillance Supplemental Report is published by the Division of HIV/AIDS Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, Centers for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services, Atlanta, Georgia. Estimates are presented for the incidence and prevalence of HIV infection among adults and adolescents (aged 13 years and older) based on data reported to CDC through December 2020. The HIV Surveillance Supplemental Report is not copyrighted and may be used and reproduced without permission. Citation of the source is, however, appreciated. Suggested citation Centers for Disease Control and Prevention. Estimated HIV incidence and prevalence in the United States, 2015–2019. HIV Surveillance Supplemental Report 2021;26(No. 1). http://www.cdc.gov/ hiv/library/reports/hiv-surveillance.html. Published May 2021. Accessed [date]. On the Web: http://www.cdc.gov/hiv/library/reports/hiv-surveillance.html Confidential information, referrals, and educational material on HIV infection CDC-INFO 1-800-232-4636 (in English, en Español) 1-888-232-6348 (TTY) http://wwwn.cdc.gov/dcs/ContactUs/Form Acknowledgments Publication of this report was made possible by the contributions of the state and territorial health departments and the HIV surveillance programs that provided surveillance data to CDC. This report was prepared by the following staff and contractors of the Division of HIV/AIDS Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, CDC: Laurie Linley, Anna Satcher Johnson, Ruiguang Song, Sherry Hu, Baohua Wu, H. -

Understanding Relative Risk, Odds Ratio, and Related Terms: As Simple As It Can Get Chittaranjan Andrade, MD
Understanding Relative Risk, Odds Ratio, and Related Terms: As Simple as It Can Get Chittaranjan Andrade, MD Each month in his online Introduction column, Dr Andrade Many research papers present findings as odds ratios (ORs) and considers theoretical and relative risks (RRs) as measures of effect size for categorical outcomes. practical ideas in clinical Whereas these and related terms have been well explained in many psychopharmacology articles,1–5 this article presents a version, with examples, that is meant with a view to update the knowledge and skills to be both simple and practical. Readers may note that the explanations of medical practitioners and examples provided apply mostly to randomized controlled trials who treat patients with (RCTs), cohort studies, and case-control studies. Nevertheless, similar psychiatric conditions. principles operate when these concepts are applied in epidemiologic Department of Psychopharmacology, National Institute research. Whereas the terms may be applied slightly differently in of Mental Health and Neurosciences, Bangalore, India different explanatory texts, the general principles are the same. ([email protected]). ABSTRACT Clinical Situation Risk, and related measures of effect size (for Consider a hypothetical RCT in which 76 depressed patients were categorical outcomes) such as relative risks and randomly assigned to receive either venlafaxine (n = 40) or placebo odds ratios, are frequently presented in research (n = 36) for 8 weeks. During the trial, new-onset sexual dysfunction articles. Not all readers know how these statistics was identified in 8 patients treated with venlafaxine and in 3 patients are derived and interpreted, nor are all readers treated with placebo. These results are presented in Table 1. -

Outcome Reporting Bias in COVID-19 Mrna Vaccine Clinical Trials
medicina Perspective Outcome Reporting Bias in COVID-19 mRNA Vaccine Clinical Trials Ronald B. Brown School of Public Health and Health Systems, University of Waterloo, Waterloo, ON N2L3G1, Canada; [email protected] Abstract: Relative risk reduction and absolute risk reduction measures in the evaluation of clinical trial data are poorly understood by health professionals and the public. The absence of reported absolute risk reduction in COVID-19 vaccine clinical trials can lead to outcome reporting bias that affects the interpretation of vaccine efficacy. The present article uses clinical epidemiologic tools to critically appraise reports of efficacy in Pfzier/BioNTech and Moderna COVID-19 mRNA vaccine clinical trials. Based on data reported by the manufacturer for Pfzier/BioNTech vaccine BNT162b2, this critical appraisal shows: relative risk reduction, 95.1%; 95% CI, 90.0% to 97.6%; p = 0.016; absolute risk reduction, 0.7%; 95% CI, 0.59% to 0.83%; p < 0.000. For the Moderna vaccine mRNA-1273, the appraisal shows: relative risk reduction, 94.1%; 95% CI, 89.1% to 96.8%; p = 0.004; absolute risk reduction, 1.1%; 95% CI, 0.97% to 1.32%; p < 0.000. Unreported absolute risk reduction measures of 0.7% and 1.1% for the Pfzier/BioNTech and Moderna vaccines, respectively, are very much lower than the reported relative risk reduction measures. Reporting absolute risk reduction measures is essential to prevent outcome reporting bias in evaluation of COVID-19 vaccine efficacy. Keywords: mRNA vaccine; COVID-19 vaccine; vaccine efficacy; relative risk reduction; absolute risk reduction; number needed to vaccinate; outcome reporting bias; clinical epidemiology; critical appraisal; evidence-based medicine Citation: Brown, R.B. -

Disease Incidence, Prevalence and Disability
Part 3 Disease incidence, prevalence and disability 9. How many people become sick each year? 28 10. Cancer incidence by site and region 29 11. How many people are sick at any given time? 31 12. Prevalence of moderate and severe disability 31 13. Leading causes of years lost due to disability in 2004 36 World Health Organization 9. How many people become sick each such as diarrhoeal disease or malaria, it is common year? for individuals to be infected repeatedly and have several episodes. For such conditions, the number The “incidence” of a condition is the number of new given in the table is the number of disease episodes, cases in a period of time – usually one year (Table 5). rather than the number of individuals affected. For most conditions in this table, the figure given is It is important to remember that the incidence of the number of individuals who developed the illness a disease or condition measures how many people or problem in 2004. However, for some conditions, are affected by it for the first time over a period of Table 5: Incidence (millions) of selected conditions by WHO region, 2004 Eastern The Mediter- South- Western World Africa Americas ranean Europe East Asia Pacific Tuberculosisa 7.8 1.4 0.4 0.6 0.6 2.8 2.1 HIV infectiona 2.8 1.9 0.2 0.1 0.2 0.2 0.1 Diarrhoeal diseaseb 4 620.4 912.9 543.1 424.9 207.1 1 276.5 1 255.9 Pertussisb 18.4 5.2 1.2 1.6 0.7 7.5 2.1 Measlesa 27.1 5.3 0.0e 1.0 0.2 17.4 3.3 Tetanusa 0.3 0.1 0.0 0.1 0.0 0.1 0.0 Meningitisb 0.7 0.3 0.1 0.1 0.0 0.2 0.1 Malariab 241.3 203.9 2.9 8.6 0.0 23.3 2.7 -

COVID-19 Vaccine Trials
COVID-19 VACCINE TRIALS SUMMARY OF PFIZER AND MODERNA COVID-19 VACCINE RESULTS Some of the most common concerns voiced involve the A review of unblinded reactogenicity data from the speed with which these vaccines were developed and final analysis which consisted of a randomized subset whether they are safe or not. How were the companies of at least 8,000 participants 18 years and older in able to get these vaccines developed and ready for the phase 2/3 study demonstrates that the vaccine distribution so fast? Were they tested in persons who was well tolerated, with most solicited adverse events are most vulnerable? Are the vaccines safe? resolving shortly after vaccination. Even though COVID-19 vaccines were developed at While the vaccine was well tolerated overall, and side an extraordinary speed, companies were required effects lasted for only a day or two, persons taking to take all of the regulatory and operational steps the vaccine should be aware that the side effects normally required for all vaccine trials, so none of are more than is seen in general for the flu vaccine these steps were skipped. What did occur that does and be prepared for them. Most side effects were not normally occur was some upfront financial mild to moderate and included injection-site pain, assistance from the federal government (Moderna redness and swelling at the injection site, fatigue/ accepted, Pfizer declined) and federal regulatory tiredness, headache, chills, muscle pain, and joint agencies working closely with the companies, pain. “Severe” side effects, defined as those that providing near real-time data with companies prevent persons from continuing daily activities were receiving review and advice more quickly. -

Prevalence and Determinants of Vaccine Hesitancy and Vaccines Recommendation Discrepancies Among General Practitioners in French-Speaking Parts of Belgium
Article Prevalence and Determinants of Vaccine Hesitancy and Vaccines Recommendation Discrepancies among General Practitioners in French-Speaking Parts of Belgium Cathy Gobert 1, Pascal Semaille 2, Thierry Van der Schueren 3 , Pierre Verger 4 and Nicolas Dauby 1,5,6,* 1 Department of Infectious Diseases, CHU Saint-Pierre, Université Libre de Bruxelles (ULB), 1000 Bruxelles, Belgium; [email protected] 2 Department of General Medicine, Université Libre de Bruxelles (ULB), 1070 Bruxelles, Belgium; [email protected] 3 Scientific Society of General Practice, 1060 Bruxelles, Belgium; [email protected] 4 Southeastern Health Regional Observatory (ORS PACA), 13005 Marseille, France; [email protected] 5 School of Public Health, Université Libre de Bruxelles (ULB), 1070 Bruxelles, Belgium 6 Institute for Medical Immunology, Université Libre de Bruxelles (ULB), 1070 Bruxelles, Belgium * Correspondence: [email protected] Abstract: General practitioners (GPs) play a critical role in patient acceptance of vaccination. Vaccine hesitancy (VH) is a growing phenomenon in the general population but also affects GPs. Few data exist on VH among GPs. The objectives of this analysis of a population of GPs in the Belgian Wallonia-Brussels Federation (WBF) were to: (1) determine the prevalence and the features of VH, (2) identify the correlates, and (3) estimate the discrepancy in vaccination’s behaviors between the GPs’ children and the recommendations made to their patients. An online survey was carried out Citation: Gobert, C.; Semaille, P.; Van among the population of general practitioners practicing in the WBF between 7 January and 18 der Schueren, T.; Verger, P.; Dauby, N. March 2020. A hierarchical cluster analysis was carried out based on various dimensions of vaccine Prevalence and Determinants of hesitancy: perception of the risks and the usefulness of vaccines as well as vaccine recommendations Vaccine Hesitancy and Vaccines for their patients. -

Plant-Based COVID-19 Vaccines: Current Status, Design, and Development Strategies of Candidate Vaccines
Review Plant-Based COVID-19 Vaccines: Current Status, Design, and Development Strategies of Candidate Vaccines Puna Maya Maharjan 1 and Sunghwa Choe 2,3,* 1 G+FLAS Life Sciences, 123 Uiryodanji-gil, Osong-eup, Heungdeok-gu, Cheongju-si 28161, Korea; punamaya.maharjan@gflas.com 2 G+FLAS Life Sciences, 38 Nakseongdae-ro, Gwanak-gu, Seoul 08790, Korea 3 School of Biological Sciences, College of Natural Sciences, Seoul National University, Gwanak-gu, Seoul 08826, Korea * Correspondence: [email protected] Abstract: The prevalence of the coronavirus disease 2019 (COVID-19) pandemic in its second year has led to massive global human and economic losses. The high transmission rate and the emergence of diverse SARS-CoV-2 variants demand rapid and effective approaches to preventing the spread, diagnosing on time, and treating affected people. Several COVID-19 vaccines are being developed using different production systems, including plants, which promises the production of cheap, safe, stable, and effective vaccines. The potential of a plant-based system for rapid production at a commercial scale and for a quick response to an infectious disease outbreak has been demonstrated by the marketing of carrot-cell-produced taliglucerase alfa (Elelyso) for Gaucher disease and tobacco- produced monoclonal antibodies (ZMapp) for the 2014 Ebola outbreak. Currently, two plant-based COVID-19 vaccine candidates, coronavirus virus-like particle (CoVLP) and Kentucky Bioprocessing (KBP)-201, are in clinical trials, and many more are in the preclinical stage. Interim phase 2 clinical Citation: Maharjan, P.M.; Choe, S. trial results have revealed the high safety and efficacy of the CoVLP vaccine, with 10 times more Plant-Based COVID-19 Vaccines: neutralizing antibody responses compared to those present in a convalescent patient’s plasma. -

EPI Case Study 1 Incidence, Prevalence, and Disease
EPIDEMIOLOGY CASE STUDY 1: Incidence, Prevalence, and Disease Surveillance; Historical Trends in the Epidemiology of M. tuberculosis STUDENT VERSION 1.0 EPI Case Study 1: Incidence, Prevalence, and Disease Surveillance; Historical Trends in the Epidemiology of M. tuberculosis Estimated Time to Complete Exercise: 30 minutes LEARNING OBJECTIVES At the completion of this Case Study, participants should be able to: ¾ Explain why denominators are necessary when comparing changes in morbidity and mortality over time ¾ Distinguish between incidence rates and prevalence ratios ¾ Calculate and interpret cause-specific morbidity and mortality rates ¾ Describe how changes in mortality or morbidity could be due to an artifact rather than a real change ASPH EPIDEMIOLOGY COMPETENCIES ADDRESSED C. 3. Describe a public health problem in terms of magnitude, person, place, and time C. 6. Apply the basic terminology and definitions of epidemiology C. 7. Calculate basic epidemiology measures C. 9. Draw appropriate inference from epidemiologic data C. 10. Evaluate the strengths and limitations of epidemiologic reports ASPH INTERDISCIPLINARY/CROSS-CUTTING COMPETENCIES ADDRESSED F.1. [Communication and Informatics] Describe how the public health information infrastructure is used to collect, process, maintain, and disseminate data J.1. [Professionalism] Discuss sentinel events in the history and development of the public health profession and their relevance for practice in the field L.2. [Systems Thinking] Identify unintended consequences produced by changes made to a public health system This material was developed by the staff at the Global Tuberculosis Institute (GTBI), one of four Regional Training and Medical Consultation Centers funded by the Centers for Disease Control and Prevention. It is published for learning purposes only. -

Likelihood Ratios, Predictive Values, and Post-Test Probabilities The
Likelihood ratios, predictive values, and post-test probabilities The purpose of this quick tutorial is to help you better understand how to use likelihood ratios (LRs), predictive values (PVs), and post-test probabilities in medical writing. The key point is that LRs are calculated differently depending on whether a test has only two possible results (dichotomous), or whether it has more than two possible outcomes (multichotomous). Examples of the latter include a clinical decision rule (CDR) that identifies low, moderate and high-risk groups, or reporting serum ferritin as < 20, 20 – 60, 61 – 100, and > 100 mg/dl. Dichotomous tests A likelihood ratio (LR) for a dichotomous test is defined as the likelihood of a test result in patients with the disease divided by the likelihood of the test result in patients without the disease. For example, the likelihood of abnormal fecal immunochemical test (FIT) in patients with colorectal cancer, divided by the likelihood of an abnormal FIT in those without cancer. A LR close to 1 means that the test result does not change the likelihood of disease or the outcome of interest appreciably. The more the likelihood ratio for a positive test (LR+) is greater than 1, the more likely the disease or outcome. The more a likelihood ratio for a negative test is less than 1, the less likely the disease or outcome. Thus, LRs correspond nicely to the clinical concepts of ruling in and ruling out disease. One suggested way to interpret LR is as follows (Ebell, 2005 Information Mastery AAFP Home Study): > 10 Large -

Considerations for the Design of Vaccine Efficacy Trials During Public Health Emergencies
bioRxiv preprint doi: https://doi.org/10.1101/261875; this version posted February 13, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-ND 4.0 International license. Considerations for the design of vaccine efficacy trials during public health emergencies Natalie E. Dean1*, Pierre-Stéphane Gsell2, Ron Brookmeyer3, Victor De Gruttola4, Christl A. Donnelly5, M. Elizabeth Halloran6,7, Momodou Jasseh8, Martha Nason9, Ximena Riveros2, Conall Watson10, Ana Maria Henao-Restrepo2, Ira M. Longini, Jr.1* 1Department of Biostatistics, University of Florida, Gainesville, FL, USA. 2World Health Organization, Geneva, Switzerland. 3Department of Biostatistics, University of California, Los Angeles, CA, USA. 4Department of Biostatistics, Harvard University, Boston, MA, USA. 5MRC Centre for Outbreak Analysis and Modelling, Department of Infectious Disease Epidemiology, School of Public Health, Imperial College London, London, UK. 6Vaccine and Infectious Disease Division, Fred Hutchinson Cancer Research Center, Seattle, WA, USA. 7Department of Biostatistics, University of Washington, Seattle, WA, USA. 8Medical Research Council, Banjul, The Gambia. 9Biostatistics Research Branch, National Institute of Allergy and Infectious Diseases, Rockville, MD, USA. 10Department of Infectious Disease Epidemiology, London School of Hygiene & Tropical Medicine, London, UK. *To whom correspondence should be addressed: [email protected] or [email protected], Dauer Hall, PO Box 117450, Gainesville, FL 32611, USA. +1 (352) 294-1945 One Sentence Summary: As part of the WHO research and development Blueprint for action to prevent epidemics, we describe key considerations for the design and analysis of trials and studies to evaluate experimental vaccines during public health emergencies.