Previous Estradiol Treatment During Midlife Maintains Transcriptional Regulation of Memory
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chr21 Protein-Protein Interactions: Enrichment in Products Involved in Intellectual Disabilities, Autism and Late Onset Alzheimer Disease
bioRxiv preprint doi: https://doi.org/10.1101/2019.12.11.872606; this version posted December 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Chr21 protein-protein interactions: enrichment in products involved in intellectual disabilities, autism and Late Onset Alzheimer Disease Julia Viard1,2*, Yann Loe-Mie1*, Rachel Daudin1, Malik Khelfaoui1, Christine Plancon2, Anne Boland2, Francisco Tejedor3, Richard L. Huganir4, Eunjoon Kim5, Makoto Kinoshita6, Guofa Liu7, Volker Haucke8, Thomas Moncion9, Eugene Yu10, Valérie Hindie9, Henri Bléhaut11, Clotilde Mircher12, Yann Herault13,14,15,16,17, Jean-François Deleuze2, Jean- Christophe Rain9, Michel Simonneau1, 18, 19, 20** and Aude-Marie Lepagnol- Bestel1** 1 Centre Psychiatrie & Neurosciences, INSERM U894, 75014 Paris, France 2 Laboratoire de génomique fonctionnelle, CNG, CEA, Evry 3 Instituto de Neurociencias CSIC-UMH, Universidad Miguel Hernandez-Campus de San Juan 03550 San Juan (Alicante), Spain 4 Department of Neuroscience, The Johns Hopkins University School of Medicine, Baltimore, MD 21205 USA 5 Center for Synaptic Brain Dysfunctions, Institute for Basic Science, Daejeon 34141, Republic of Korea 6 Department of Molecular Biology, Division of Biological Science, Nagoya University Graduate School of Science, Furo, Chikusa, Nagoya, Japan 7 Department of Biological Sciences, University of Toledo, Toledo, OH, 43606, USA 8 Leibniz Forschungsinstitut für Molekulare Pharmakologie -

Neural Stem Cell-Derived Exosomes Revert HFD-Dependent Memory Impairment Via CREB-BDNF Signalling
International Journal of Molecular Sciences Article Neural Stem Cell-Derived Exosomes Revert HFD-Dependent Memory Impairment via CREB-BDNF Signalling Matteo Spinelli 1, Francesca Natale 1,2, Marco Rinaudo 1, Lucia Leone 1,2, Daniele Mezzogori 1, Salvatore Fusco 1,2,* and Claudio Grassi 1,2 1 Department of Neuroscience, Università Cattolica del Sacro Cuore, 00168 Rome, Italy; [email protected] (M.S.); [email protected] (F.N.); [email protected] (M.R.); [email protected] (L.L.); [email protected] (D.M.); [email protected] (C.G.) 2 Fondazione Policlinico Universitario Agostino Gemelli IRCCS, 00168 Rome, Italy * Correspondence: [email protected] Received: 6 October 2020; Accepted: 25 November 2020; Published: 26 November 2020 Abstract: Overnutrition and metabolic disorders impair cognitive functions through molecular mechanisms still poorly understood. In mice fed with a high fat diet (HFD) we analysed the expression of synaptic plasticity-related genes and the activation of cAMP response element-binding protein (CREB)-brain-derived neurotrophic factor (BDNF)-tropomyosin receptor kinase B (TrkB) signalling. We found that a HFD inhibited both CREB phosphorylation and the expression of a set of CREB target genes in the hippocampus. The intranasal administration of neural stem cell (NSC)-derived exosomes (exo-NSC) epigenetically restored the transcription of Bdnf, nNOS, Sirt1, Egr3, and RelA genes by inducing the recruitment of CREB on their regulatory sequences. Finally, exo-NSC administration rescued both BDNF signalling and memory in HFD mice. Collectively, our findings highlight novel mechanisms underlying HFD-related memory impairment and provide evidence of the potential therapeutic effect of exo-NSC against metabolic disease-related cognitive decline. -

BDNF Integrity in Ageing and Stress
MOJ Gerontology & Geriatrics Editorial Open Access BDNF integrity in ageing and stress Editorial Volume 1 Issue 6 - 2017 Alzheimer’s disease (AD) presents a progressive, stage-dependent Trevor Archer age-related neurodegenerative disorder that is characterized by Department of Psychology, University of Gothenburg, Sweden aggregation of toxic forms of amyloid β peptide (Aβ). There is much support for the notion that that brain-derived neurotrophic factor Correspondence: Trevor Archer, Department of Psychology, (BDNF) mediates beneficial effects of exercise on neuroplasticity and University of Gothenburg, Sweden, cellular stress resistance.1‒3 BDNF is associated with neuroplasticity Email [email protected] changes promoting health and well-being whereas these influences Received: July 16, 2017 | Published: August 01, 2017 may be opposed by pro-inflammatory cytokines, key factors in neurodegenerative processes.4 Lower serum levels are linked to greater symptom, cognitive, impairment.5 Thus, interventions, as for example physical exercise, whether acute or chronic, endurance or resistance, promote the mobilization of BDNF and other neurotrophic factors6,7 increase hippocampal and other brain regional resilience8 and therewith progression under conditions of stress when confronted critical life protect against cell death by inducing cell proliferation and maturation episodes or periods, appears to be modulated by the availability and with enhanced neuronal reparation, neurogenesis and the growth and accessibility of BDNF.20‒22 Over a wide range of neurologic and functioning of neurons in neurodegenerative disorders.9,10 Sampaio et psychiatric disease states it is found that the integrity and concentrations al.11 in a review of the therapeutic applications of neurotrophic factors, of brain, serum and plasma BDNF is compromised, particularly under particularly BDNF, postulate that they are essential for survival, stressful conditions. -
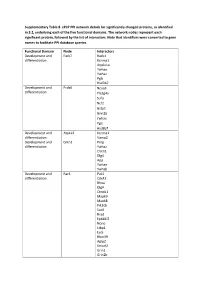
Supplementary Table 8. Cpcp PPI Network Details for Significantly Changed Proteins, As Identified in 3.2, Underlying Each of the Five Functional Domains
Supplementary Table 8. cPCP PPI network details for significantly changed proteins, as identified in 3.2, underlying each of the five functional domains. The network nodes represent each significant protein, followed by the list of interactors. Note that identifiers were converted to gene names to facilitate PPI database queries. Functional Domain Node Interactors Development and Park7 Rack1 differentiation Kcnma1 Atp6v1a Ywhae Ywhaz Pgls Hsd3b7 Development and Prdx6 Ncoa3 differentiation Pla2g4a Sufu Ncf2 Gstp1 Grin2b Ywhae Pgls Hsd3b7 Development and Atp1a2 Kcnma1 differentiation Vamp2 Development and Cntn1 Prnp differentiation Ywhaz Clstn1 Dlg4 App Ywhae Ywhab Development and Rac1 Pak1 differentiation Cdc42 Rhoa Dlg4 Ctnnb1 Mapk9 Mapk8 Pik3cb Sod1 Rrad Epb41l2 Nono Ltbp1 Evi5 Rbm39 Aplp2 Smurf2 Grin1 Grin2b Xiap Chn2 Cav1 Cybb Pgls Ywhae Development and Hbb-b1 Atp5b differentiation Hba Kcnma1 Got1 Aldoa Ywhaz Pgls Hsd3b4 Hsd3b7 Ywhae Development and Myh6 Mybpc3 differentiation Prkce Ywhae Development and Amph Capn2 differentiation Ap2a2 Dnm1 Dnm3 Dnm2 Atp6v1a Ywhab Development and Dnm3 Bin1 differentiation Amph Pacsin1 Grb2 Ywhae Bsn Development and Eef2 Ywhaz differentiation Rpgrip1l Atp6v1a Nphp1 Iqcb1 Ezh2 Ywhae Ywhab Pgls Hsd3b7 Hsd3b4 Development and Gnai1 Dlg4 differentiation Development and Gnao1 Dlg4 differentiation Vamp2 App Ywhae Ywhab Development and Psmd3 Rpgrip1l differentiation Psmd4 Hmga2 Development and Thy1 Syp differentiation Atp6v1a App Ywhae Ywhaz Ywhab Hsd3b7 Hsd3b4 Development and Tubb2a Ywhaz differentiation Nphp4 -

LGI1–ADAM22–MAGUK Configures Transsynaptic Nanoalignment for Synaptic Transmission and Epilepsy Prevention
LGI1–ADAM22–MAGUK configures transsynaptic nanoalignment for synaptic transmission and epilepsy prevention Yuko Fukataa,b,1, Xiumin Chenc,d,1, Satomi Chikenb,e, Yoko Hiranoa,f, Atsushi Yamagatag, Hiroki Inahashia, Makoto Sanboh, Hiromi Sanob,e, Teppei Gotoh, Masumi Hirabayashib,h, Hans-Christian Kornaui,j, Harald Prüssi,k, Atsushi Nambub,e, Shuya Fukail, Roger A. Nicollc,d,2, and Masaki Fukataa,b,2 aDivision of Membrane Physiology, Department of Molecular and Cellular Physiology, National Institute for Physiological Sciences, National Institutes of Natural Sciences, Aichi 444-8787, Japan; bDepartment of Physiological Sciences, School of Life Science, SOKENDAI (The Graduate University for Advanced Studies), Aichi 444-8585, Japan; cDepartment of Cellular and Molecular Pharmacology, University of California, San Francisco, CA 94158; dDepartment of Physiology, University of California, San Francisco, CA 94158; eDivision of System Neurophysiology, Department of System Neuroscience, National Institute for Physiological Sciences, National Institutes of Natural Sciences, Aichi 444-8585, Japan; fDepartment of Pediatrics, Graduate School of Medicine, The University of Tokyo, Tokyo 113-8655, Japan; gLaboratory for Protein Functional and Structural Biology, RIKEN Center for Biosystems Dynamics Research, Kanagawa 230-0045, Japan; hCenter for Genetic Analysis of Behavior, National Institute for Physiological Sciences, National Institutes of Natural Sciences, Okazaki 444-8787, Japan; iGerman Center for Neurodegenerative Diseases (DZNE), 10117 Berlin, Germany; jNeuroscience Research Center, Cluster NeuroCure, Charité-Universitätsmedizin Berlin, 10117 Berlin, Germany; kDepartment of Neurology and Experimental Neurology, Charité-Universitätsmedizin Berlin, 10117 Berlin, Germany; and lDepartment of Chemistry, Graduate School of Science, Kyoto University, 606-8502 Kyoto, Japan Contributed by Roger A. Nicoll, December 1, 2020 (sent for review October 29, 2020; reviewed by David S. -

Quantitative Proteomics Reveals Significant Differences
cells Article Quantitative Proteomics Reveals Significant Differences between Mouse Brain Formations in Expression of Proteins Involved in Neuronal Plasticity during Aging Dominika Drulis-Fajdasz 1,† , Kinga Gostomska-Pampuch 2,3,† , Przemysław Duda 1 , Jacek Roman Wi´sniewski 2 and Dariusz Rakus 1,* 1 Department of Molecular Physiology and Neurobiology, University of Wrocław, Sienkiewicza 21, 50-335 Wrocław, Poland; [email protected] (D.D.-F.); [email protected] (P.D.) 2 Biochemical Proteomics Group, Department of Proteomics and Signal Transduction, Max Planck Institute of Biochemistry, 82152 Martinsried, Germany; [email protected] (K.G.-P.); [email protected] (J.R.W.) 3 Department of Biochemistry and Immunochemistry, Wrocław Medical University, Chałubi´nskiego10, 50-368 Wrocław, Poland * Correspondence: [email protected] † The authors participated equally. Abstract: Aging is associated with a general decline in cognitive functions, which appears to be due to alterations in the amounts of proteins involved in the regulation of synaptic plasticity. Here, we present a quantitative analysis of proteins involved in neurotransmission in three brain regions, namely, the hippocampus, the cerebral cortex and the cerebellum, in mice aged 1 and 22 months, Citation: Drulis-Fajdasz, D.; using the total protein approach technique. We demonstrate that although the titer of some proteins Gostomska-Pampuch, K.; Duda, P.; involved in neurotransmission and synaptic plasticity is affected by aging in a similar manner Wi´sniewski,J.R.; Rakus, D. Quantitative Proteomics Reveals in all the studied brain formations, in fact, each of the formations represents its own mode of Significant Differences between aging. -

Dlg1 Maintains Dendritic Cell Function by Securing Voltage-Gated K+ Channel Integrity
Dlg1 Maintains Dendritic Cell Function by Securing Voltage-Gated K + Channel Integrity This information is current as Xuejiao Dong, Lisi Wei, Xueheng Guo, Zhiyong Yang, of September 24, 2021. Chuan Wu, Peiyu Li, Lu Lu, Hai Qi, Yan Shi, Xiaoyu Hu, Li Wu, Liangyi Chen and Wanli Liu J Immunol published online 26 April 2019 http://www.jimmunol.org/content/early/2019/04/25/jimmun ol.1900089 Downloaded from Supplementary http://www.jimmunol.org/content/suppl/2019/04/25/jimmunol.190008 Material 9.DCSupplemental http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on September 24, 2021 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2019 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. Published April 26, 2019, doi:10.4049/jimmunol.1900089 The Journal of Immunology Dlg1 Maintains Dendritic Cell Function by Securing Voltage-Gated K+ Channel Integrity Xuejiao Dong,*,1 Lisi Wei,†,1 Xueheng Guo,‡,x,1 Zhiyong Yang,{ Chuan Wu,‖ Peiyu Li,#,** Lu Lu,# Hai Qi,‡ Yan Shi,‡ Xiaoyu Hu,‡ Li Wu,‡ Liangyi Chen,† and Wanli Liu* Dendritic cells (DCs) play key roles in Ab responses by presenting Ags to lymphocytes and by producing proinflammatory cytokines. -

Hypoxia Transcriptomic Modifications Induced by Proton Irradiation In
Journal of Personalized Medicine Article Hypoxia Transcriptomic Modifications Induced by Proton Irradiation in U87 Glioblastoma Multiforme Cell Line Valentina Bravatà 1,2,† , Walter Tinganelli 3,†, Francesco P. Cammarata 1,2,* , Luigi Minafra 1,2,* , Marco Calvaruso 1,2 , Olga Sokol 3 , Giada Petringa 2, Giuseppe A.P. Cirrone 2, Emanuele Scifoni 4 , Giusi I. Forte 1,2,‡ and Giorgio Russo 1,2,‡ 1 Institute of Molecular Bioimaging and Physiology–National Research Council (IBFM-CNR), 90015 Cefalù, Italy; [email protected] (V.B.); [email protected] (M.C.); [email protected] (G.I.F.); [email protected] (G.R.) 2 Laboratori Nazionali del SUD, Istituto Nazionale di Fisica Nucleare (LNS-INFN), 95100 Catania, Italy; [email protected] (G.P.); [email protected] (G.A.P.C.) 3 Biophysics Department, GSI Helmholtzzentrum für Schwerionenforschung GmbH, 64291 Darmstadt, Germany; [email protected] (W.T.); [email protected] (O.S.) 4 Trento Institute for Fundamental Physics and Applications (TIFPA), Istituto Nazionale Fisica Nucleare (INFN), 38123 Trento, Italy; [email protected] * Correspondence: [email protected] (F.P.C.); [email protected] (L.M.) † These authors contributed equally to this work. ‡ These authors contributed equally to this work. Abstract: In Glioblastoma Multiforme (GBM), hypoxia is associated with radioresistance and poor prognosis. Since standard GBM treatments are not always effective, new strategies are needed Citation: Bravatà, V.; Tinganelli, W.; to overcome resistance to therapeutic treatments, including radiotherapy (RT). Our study aims Cammarata, F.P.; Minafra, L.; to shed light on the biomarker network involved in a hypoxic (0.2% oxygen) GBM cell line that Calvaruso, M.; Sokol, O.; Petringa, G.; is radioresistant after proton therapy (PT). -

Perkinelmer Genomics to Request the Saliva Swab Collection Kit for Patients That Cannot Provide a Blood Sample As Whole Blood Is the Preferred Sample
Autism and Intellectual Disability TRIO Panel Test Code TR002 Test Summary This test analyzes 2429 genes that have been associated with Autism and Intellectual Disability and/or disorders associated with Autism and Intellectual Disability with the analysis being performed as a TRIO Turn-Around-Time (TAT)* 3 - 5 weeks Acceptable Sample Types Whole Blood (EDTA) (Preferred sample type) DNA, Isolated Dried Blood Spots Saliva Acceptable Billing Types Self (patient) Payment Institutional Billing Commercial Insurance Indications for Testing Comprehensive test for patients with intellectual disability or global developmental delays (Moeschler et al 2014 PMID: 25157020). Comprehensive test for individuals with multiple congenital anomalies (Miller et al. 2010 PMID 20466091). Patients with autism/autism spectrum disorders (ASDs). Suspected autosomal recessive condition due to close familial relations Previously negative karyotyping and/or chromosomal microarray results. Test Description This panel analyzes 2429 genes that have been associated with Autism and ID and/or disorders associated with Autism and ID. Both sequencing and deletion/duplication (CNV) analysis will be performed on the coding regions of all genes included (unless otherwise marked). All analysis is performed utilizing Next Generation Sequencing (NGS) technology. CNV analysis is designed to detect the majority of deletions and duplications of three exons or greater in size. Smaller CNV events may also be detected and reported, but additional follow-up testing is recommended if a smaller CNV is suspected. All variants are classified according to ACMG guidelines. Condition Description Autism Spectrum Disorder (ASD) refers to a group of developmental disabilities that are typically associated with challenges of varying severity in the areas of social interaction, communication, and repetitive/restricted behaviors. -

Reduced Expression of the Psychiatric Risk Gene DLG2 (PSD93) Impairs
bioRxiv preprint doi: https://doi.org/10.1101/2021.08.02.454736; this version posted August 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Reduced expression of the psychiatric risk gene DLG2 (PSD93) impairs hippocampal synaptic integration and plasticity Simonas Griesius1, Cian O’Donnell2, Sophie Waldron3,6, Kerrie L. Thomas3,5, Dominic M. Dwyer3,6, Lawrence S. Wilkinson3,4,6, Jeremy Hall3,4,5, Emma S. J. Robinson1, Jack R. Mellor1* 1 Centre for Synaptic Plasticity, School of Physiology, Pharmacology and Neuroscience, University of Bristol, University Walk, Bristol BS8 1TD, UK 2 Computational Neuroscience Unit, School of Computer Science, Electrical and Electronic Engineering, and Engineering Mathematics, University of Bristol, Bristol BS8 1UB, UK 3 Neuroscience and Mental Health Research Institute, 4 MRC Centre for Neuropsychiatric Genetics and Genomics, Schools of 5 Medicine and 6 Psychology, Cardiff CF24 4HQ, UK * Corresponding author: [email protected], +44 117 331 1944 Running title Low dosage DLG2 impairs synaptic plasticity Keywords Schizophrenia, autism spectrum disorder, dendritic integration, plateau potential, muscarinic M1 receptor, phenotype rescue 1 bioRxiv preprint doi: https://doi.org/10.1101/2021.08.02.454736; this version posted August 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. -

The Pdx1 Bound Swi/Snf Chromatin Remodeling Complex Regulates Pancreatic Progenitor Cell Proliferation and Mature Islet Β Cell
Page 1 of 125 Diabetes The Pdx1 bound Swi/Snf chromatin remodeling complex regulates pancreatic progenitor cell proliferation and mature islet β cell function Jason M. Spaeth1,2, Jin-Hua Liu1, Daniel Peters3, Min Guo1, Anna B. Osipovich1, Fardin Mohammadi3, Nilotpal Roy4, Anil Bhushan4, Mark A. Magnuson1, Matthias Hebrok4, Christopher V. E. Wright3, Roland Stein1,5 1 Department of Molecular Physiology and Biophysics, Vanderbilt University, Nashville, TN 2 Present address: Department of Pediatrics, Indiana University School of Medicine, Indianapolis, IN 3 Department of Cell and Developmental Biology, Vanderbilt University, Nashville, TN 4 Diabetes Center, Department of Medicine, UCSF, San Francisco, California 5 Corresponding author: [email protected]; (615)322-7026 1 Diabetes Publish Ahead of Print, published online June 14, 2019 Diabetes Page 2 of 125 Abstract Transcription factors positively and/or negatively impact gene expression by recruiting coregulatory factors, which interact through protein-protein binding. Here we demonstrate that mouse pancreas size and islet β cell function are controlled by the ATP-dependent Swi/Snf chromatin remodeling coregulatory complex that physically associates with Pdx1, a diabetes- linked transcription factor essential to pancreatic morphogenesis and adult islet-cell function and maintenance. Early embryonic deletion of just the Swi/Snf Brg1 ATPase subunit reduced multipotent pancreatic progenitor cell proliferation and resulted in pancreas hypoplasia. In contrast, removal of both Swi/Snf ATPase subunits, Brg1 and Brm, was necessary to compromise adult islet β cell activity, which included whole animal glucose intolerance, hyperglycemia and impaired insulin secretion. Notably, lineage-tracing analysis revealed Swi/Snf-deficient β cells lost the ability to produce the mRNAs for insulin and other key metabolic genes without effecting the expression of many essential islet-enriched transcription factors. -

DNA Methylation of DLG4 and GJA-1 of Human Hippocampus And
essio epr n D an f d Kaut et al., J Depress Anxiety 2016, S1 o A l a n n x r DOI: 10.4172/2167-1044.S1-022 i e u t y o J Journal of Depression and Anxiety ISSN: 2167-1044 ResearchShort Communication Article OpenOpen Access Access DNA Methylation of DLG4 and GJA-1 of Human Hippocampus and Prefrontal Cortex in Major Depression is Unchanged in Comparison to Healthy Individuals Oliver Kaut1#, Amit Sharma1#, Ina Schmitt1, René Hurlemann2 and Ullrich Wüllner1,3* 1Department of Neurology, University Clinic of Bonn, Bonn, Germany 2Division of Molecular Psychology, University Clinic of Bonn, Bonn, Germany 3German Centre for Neurodegenerative Diseases (DZNE), Bonn, Germany #Equally Contributed Abstract Epigenetic alterations provide a potential mechanism to account for the numerous gene-environment interactions that have been reported in association with neuropsychiatric phenotypes. In context to major depression disorder (MDD), where postmortem and neuroimaging studies provide insights into dysfunctional brain regions, involvement of genetic heterogeneity also revealed the complexity of this disorder. Despite intensive research during the past several decades and information from genome wide studies, pathophysiology of depressive disorders remained elusive. To evaluate the impact of epigenetic pressure on this disease, we took advantage of DNA isolated from different sections of human brain (prefrontal cortex and hippocampus) from clinically well-defined depressed patients and healthy individuals and performed pyrosequencing for DNA methylation analysis. Herein, we focused on two genes DLG4 (PSD-95) and GJA-1 (Connexin43) known to be associated with neuropsychiatric behavior. Comparing MDD with controls we found no differences of DNA methylation.