Proteolytic Inventory of Thermococcus Kodakaraensis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Structural and Biochemical Characterizations of Three Potential Drug Targets from Pathogens
Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 2020 Structural and Biochemical Characterizations of Three Potential Drug Targets from Pathogens LU LU ACTA UNIVERSITATIS UPSALIENSIS ISSN 1651-6214 ISBN 978-91-513-1148-7 UPPSALA urn:nbn:se:uu:diva-435815 2021 Dissertation presented at Uppsala University to be publicly examined in Room A1:111a, BMC, Husargatan 3, Uppsala, Friday, 16 April 2021 at 13:15 for the degree of Doctor of Philosophy. The examination will be conducted in English. Faculty examiner: Christian Cambillau. Abstract Lu, L. 2021. Structural and Biochemical Characterizations of Three Potential Drug Targets from Pathogens. Digital Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 2020. 91 pp. Uppsala: Acta Universitatis Upsaliensis. ISBN 978-91-513-1148-7. As antibiotic resistance of various pathogens emerged globally, the need for new effective drugs with novel modes of action became urgent. In this thesis, we focus on infectious diseases, e.g. tuberculosis, malaria, and nosocomial infections, and the corresponding causative pathogens, Mycobacterium tuberculosis, Plasmodium falciparum, and the Gram-negative ESKAPE pathogens that underlie so many healthcare-acquired diseases. Following the same- target-other-pathogen (STOP) strategy, we attempted to comprehensively explore the properties of three promising drug targets. Signal peptidase I (SPase I), existing both in Gram-negative and Gram-positive bacteria, as well as in parasites, is vital for cell viability, due to its critical role in signal peptide cleavage, thus, protein maturation, and secreted protein transport. Three factors, comprising essentiality, a unique mode of action, and easy accessibility, make it an attractive drug target. -

Co-Catalytic Metallopeptidases As Pharmaceutical Targets Richard C
Marquette University e-Publications@Marquette Chemistry Faculty Research and Publications Chemistry, Department of 4-1-2003 Co-Catalytic Metallopeptidases as Pharmaceutical Targets Richard C. Holz Marquette University, [email protected] Krzysztof P. Bzymek Utah State University Sabina I. Swierczek Utah State University Accepted version. Current Opinion in Chemical Biology, Vol. 7, No. 2 (April 2003): 197-206. DOI. © 2003 Elsevier Science Ltd. Used with permission. Richard Holz was affiliated with the Utah State University at the time of publication. Marquette University e-Publications@Marquette Chemistry Faculty Research and Publications/College of Arts and Sciences This paper is NOT THE PUBLISHED VERSION; but the author’s final, peer-reviewed manuscript. The published version may be accessed by following the link in the citation below. Current Opinion in Chemical Biology, Vol.7, No. 2 (2003): 197-206. DOI. This article is © Elsevier and permission has been granted for this version to appear in e-Publications@Marquette. Elsevier does not grant permission for this article to be further copied/distributed or hosted elsewhere without the express permission from Elsevier. Co-catalytic metallopeptidases as pharmaceutical targets Richard C Holz Department of Chemistry, Marquette University, Milwaukee, WI Department of Chemistry and Biochemistry, Utah State University, Logan, Utah 84322-0300, USA Krzysztof P Bzymek Department of Chemistry and Biochemistry, Utah State University, Logan, Utah 84322-0300, USA Sabina I Swierczek Department of Chemistry and Biochemistry, Utah State University, Logan, Utah 84322-0300, USA Abstract Understanding the reaction mechanism of co-catalytic metallopeptidases provides a starting point for the design and synthesis of new molecules that can be screened as potential pharmaceuticals. -

Catalytic Properties and Inhibition of Proline-Specific Dipeptidyl Peptidases II, IV and VII
UC Berkeley UC Berkeley Previously Published Works Title Catalytic properties and inhibition of proline-specific dipeptidyl peptidases II, IV and VII Permalink https://escholarship.org/uc/item/4nf146zf Journal Biochemical Journal, 371 ISSN 0264-6021 Authors Leiting, B Pryor, K D Wu, J K et al. Publication Date 2003-04-01 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Biochem. J. (2003) 371, 525–532 (Printed in Great Britain) 525 Catalytic properties and inhibition of proline-specific dipeptidyl peptidases II, IV and VII Barbara LEITING*1, KellyAnn D. PRYOR*, Joseph K. WU*, Frank MARSILIO*, Reshma A. PATEL*, Charles S. CRAIK†, Jonathan A. ELLMAN‡, Richard T. CUMMINGS* and Nancy A. THORNBERRY* *Department of Metabolic Disorders, Merck Research Laboratories, Mail code RY50G-236, P.O. Box 2000, Rahway, NJ 07065, U.S.A., †Department of Pharmaceutical Chemistry, University of California, 513 Parnassus Avenue, San Francisco, CA 94143-0446, U.S.A., and ‡Department of Chemistry, University of California, Berkeley, CA 94720, U.S.A. There is currently intense interest in the emerging group of strates and inhibitors for these enzymes, a complete biochemical proline-specific dipeptidases, and their roles in the regulation profile of these enzymes was obtained. The pH profiles, substrate of biological processes. Dipeptidyl peptidase IV (DPP-IV) is specificities as determined by positional scanning, Michaelis– involved in glucose metabolism by contributing to the regulation Menten constants and inhibition profiles for DPP-VII and DPP- of glucagon family peptides and has emerged as a potential target II were shown to be virtually identical, strongly supporting the for the treatment of metabolic diseases. -

Serine Proteases with Altered Sensitivity to Activity-Modulating
(19) & (11) EP 2 045 321 A2 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 08.04.2009 Bulletin 2009/15 C12N 9/00 (2006.01) C12N 15/00 (2006.01) C12Q 1/37 (2006.01) (21) Application number: 09150549.5 (22) Date of filing: 26.05.2006 (84) Designated Contracting States: • Haupts, Ulrich AT BE BG CH CY CZ DE DK EE ES FI FR GB GR 51519 Odenthal (DE) HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI • Coco, Wayne SK TR 50737 Köln (DE) •Tebbe, Jan (30) Priority: 27.05.2005 EP 05104543 50733 Köln (DE) • Votsmeier, Christian (62) Document number(s) of the earlier application(s) in 50259 Pulheim (DE) accordance with Art. 76 EPC: • Scheidig, Andreas 06763303.2 / 1 883 696 50823 Köln (DE) (71) Applicant: Direvo Biotech AG (74) Representative: von Kreisler Selting Werner 50829 Köln (DE) Patentanwälte P.O. Box 10 22 41 (72) Inventors: 50462 Köln (DE) • Koltermann, André 82057 Icking (DE) Remarks: • Kettling, Ulrich This application was filed on 14-01-2009 as a 81477 München (DE) divisional application to the application mentioned under INID code 62. (54) Serine proteases with altered sensitivity to activity-modulating substances (57) The present invention provides variants of ser- screening of the library in the presence of one or several ine proteases of the S1 class with altered sensitivity to activity-modulating substances, selection of variants with one or more activity-modulating substances. A method altered sensitivity to one or several activity-modulating for the generation of such proteases is disclosed, com- substances and isolation of those polynucleotide se- prising the provision of a protease library encoding poly- quences that encode for the selected variants. -

Proquest Dissertations
urn u Ottawa L'Universitd canadienne Canada's university FACULTE DES ETUDES SUPERIEURES l^^l FACULTY OF GRADUATE AND ET POSTDOCTORALES u Ottawa POSTDOCTORAL STUDIES I.'University emiadienne Canada's university Charles Gyamera-Acheampong AUTEUR DE LA THESE / AUTHOR OF THESIS Ph.D. (Biochemistry) GRADE/DEGREE Biochemistry, Microbiology and Immunology FACULTE, ECOLE, DEPARTEMENT / FACULTY, SCHOOL, DEPARTMENT The Physiology and Biochemistry of the Fertility Enzyme Proprotein Convertase Subtilisin/Kexin Type 4 TITRE DE LA THESE / TITLE OF THESIS M. Mbikay TIRECTWRTDIRICTR^ CO-DIRECTEUR (CO-DIRECTRICE) DE LA THESE / THESIS CO-SUPERVISOR EXAMINATEURS (EXAMINATRICES) DE LA THESE/THESIS EXAMINERS A. Basak G. Cooke F .Kan V. Mezl Gary W. Slater Le Doyen de la Faculte des etudes superieures et postdoctorales / Dean of the Faculty of Graduate and Postdoctoral Studies Library and Archives Bibliotheque et 1*1 Canada Archives Canada Published Heritage Direction du Branch Patrimoine de I'edition 395 Wellington Street 395, rue Wellington OttawaONK1A0N4 Ottawa ON K1A 0N4 Canada Canada Your file Votre reference ISBN: 978-0-494-59504-6 Our file Notre reference ISBN: 978-0-494-59504-6 NOTICE: AVIS: The author has granted a non L'auteur a accorde une licence non exclusive exclusive license allowing Library and permettant a la Bibliotheque et Archives Archives Canada to reproduce, Canada de reproduire, publier, archiver, publish, archive, preserve, conserve, sauvegarder, conserver, transmettre au public communicate to the public by par telecommunication ou par I'lnternet, prefer, telecommunication or on the Internet, distribuer et vendre des theses partout dans le loan, distribute and sell theses monde, a des fins commerciales ou autres, sur worldwide, for commercial or non support microforme, papier, electronique et/ou commercial purposes, in microform, autres formats. -

Genetic Polymorphism of the Angiotensin-Converting Enzyme (ACE) in Asthmatic Patients
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector RESPIRATORY MEDICINE (1998) 92, 1305-1310 Original Articles Genetic polymorphism of the angiotensin-converting enzyme (ACE) in asthmatic patients H. TOMITA, S. SATO, R. MATSUDA, N. OGISU, T. MORI, T. NIIMI AND S. SHIMIZU 2nd Department of Intemal Medicine, Nagoya City University Medical School, Nagoya, Japan Angiotensin-converting enzyme (ACE) inactivates bradykinin, substance P and neurokinin A, which are believed to play important roles in the pathogenesis of asthma, especially in neurogenic inflammation. It has recently been shown that an insertion (1)ldeletion (D) polymorphism in the ACE gene accounts for variation in serum ACE levels. There are thus three genotypes (insertion homozygote, II; deletion homozygote, DD; heterozygotes, DI). The serum ACE level with the DD type is reported to be about double that of the II type and intermediate in the DI case. In the present study, we examined whether asthma is linked with this ACE gene polymorphism. Seventy-one patients with asthma (27 males and 44 females) and 142 sex- and age-matched healthy controls were determined for their genotype by the polymerase chain reaction (PCR) method. Twenty-five asthmatics demonstrated the II type (352%) 37 the DI type (52.1%), and nine the DD type (12.7%). There were no significant differences in the distributions of genotypes and serum ACE levels between healthy controls and patients. No significant differences were evident in serum IgE levels, age at onset, proportion of atopic type patients and severity of asthma among the three genotypes. -

B Number Gene Name Mrna Intensity Mrna
sample) total list predicted B number Gene name assignment mRNA present mRNA intensity Gene description Protein detected - Membrane protein membrane sample detected (total list) Proteins detected - Functional category # of tryptic peptides # of tryptic peptides # of tryptic peptides detected (membrane b0002 thrA 13624 P 39 P 18 P(m) 2 aspartokinase I, homoserine dehydrogenase I Metabolism of small molecules b0003 thrB 6781 P 9 P 3 0 homoserine kinase Metabolism of small molecules b0004 thrC 15039 P 18 P 10 0 threonine synthase Metabolism of small molecules b0008 talB 20561 P 20 P 13 0 transaldolase B Metabolism of small molecules chaperone Hsp70; DNA biosynthesis; autoregulated heat shock b0014 dnaK 13283 P 32 P 23 0 proteins Cell processes b0015 dnaJ 4492 P 13 P 4 P(m) 1 chaperone with DnaK; heat shock protein Cell processes b0029 lytB 1331 P 16 P 2 0 control of stringent response; involved in penicillin tolerance Global functions b0032 carA 9312 P 14 P 8 0 carbamoyl-phosphate synthetase, glutamine (small) subunit Metabolism of small molecules b0033 carB 7656 P 48 P 17 0 carbamoyl-phosphate synthase large subunit Metabolism of small molecules b0048 folA 1588 P 7 P 1 0 dihydrofolate reductase type I; trimethoprim resistance Metabolism of small molecules peptidyl-prolyl cis-trans isomerase (PPIase), involved in maturation of b0053 surA 3825 P 19 P 4 P(m) 1 GenProt outer membrane proteins (1st module) Cell processes b0054 imp 2737 P 42 P 5 P(m) 5 GenProt organic solvent tolerance Cell processes b0071 leuD 4770 P 10 P 9 0 isopropylmalate -

Methionine Aminopeptidase Emerging Role in Angiogenesis
Chapter 2 Methionine Aminopeptidase Emerging role in angiogenesis Joseph A. Vetro1, Benjamin Dummitt2, and Yie-Hwa Chang2 1Department of Pharmaceutical Chemistry, University of Kansas, 2095 Constant Ave., Lawrence, KS 66047, USA. 2Edward A. Doisy Department of Biochemistry and Molecular Biology, St. Louis University Health Sciences Center, 1402 S. Grand Blvd., St. Louis, MO 63104, USA. Abstract: Angiogenesis, the formation of new blood vessels from existing vasculature, is a key factor in a number of vascular-related pathologies such as the metastasis and growth of solid tumors. Thus, the inhibition of angiogenesis has great potential as a therapeutic modality in the treatment of cancer and other vascular-related diseases. Recent evidence suggests that the inhibition of mammalian methionine aminopeptidase type 2 (MetAP2) catalytic activity in vascular endothelial cells plays an essential role in the pharmacological activity of the most potent small molecule angiogenesis inhibitors discovered to date, the fumagillin class. Methionine aminopeptidase (MetAP, EC 3.4.11.18) catalyzes the non-processive, co-translational hydrolysis of initiator N-terminal methionine when the second residue of the nascent polypeptide is small and uncharged. Initiator Met removal is a ubiquitous and essential modification. Indirect evidence suggests that removal of initiator Met by MetAP is important for the normal function of many proteins involved in DNA repair, signal transduction, cell transformation, secretory vesicle trafficking, and viral capsid assembly and infection. Currently, much effort is focused on understanding the essential nature of methionine aminopeptidase activity and elucidating the role of methionine aminopeptidase type 2 catalytic activity in angiogenesis. In this chapter, we give an overview of the MetAP proteins, outline the importance of initiator Met hydrolysis, and discuss the possible mechanism(s) through which MetAP2 inhibition by the fumagillin class of angiogenesis inhibitors leads to cytostatic growth arrest in vascular endothelial cells. -
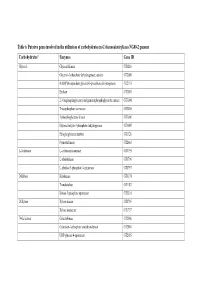
Table 6. Putative Genes Involved in the Utilization of Carbohydrates in G
Table 6. Putative genes involved in the utilization of carbohydrates in G. thermodenitrificans NG80-2 genome Carbohydrates* Enzymes Gene ID Glycerol Glycerol Kinase GT1216 Glycerol-3-phosphate dehydrogenase, aerobic GT2089 NAD(P)H-dependent glycerol-3-phosphate dehydrogenase GT2153 Enolase GT3003 2,3-bisphosphoglycerate-independentphosphoglycerate mutase GT3004 Triosephosphate isomerase GT3005 3-phosphoglycerate kinase GT3006 Glyceraldehyde-3-phosphate dehydrogenase GT3007 Phosphoglycerate mutase GT1326 Pyruvate kinase GT2663 L-Arabinose L-arabinose isomerase GT1795 L-ribulokinase GT1796 L-ribulose 5-phosphate 4-epimerase GT1797 D-Ribose Ribokinase GT3174 Transketolase GT1187 Ribose 5-phosphate epimerase GT3316 D-Xylose Xylose kinase GT1756 Xylose isomerase GT1757 D-Galactose Galactokinase GT2086 Galactose-1-phosphate uridyltransferase GT2084 UDP-glucose 4-epimerase GT2085 Carbohydrates* Enzymes Gene ID D-Fructose 1-phosphofructokinase GT1727 Fructose-1,6-bisphosphate aldolase GT1805 Fructose-1,6-bisphosphate aldolase type II GT3331 Triosephosphate isomerase GT3005 D-Mannose Mannnose-6 phospate isomelase GT3398 6-phospho-1-fructokinase GT2664 D-Mannitol Mannitol-1-phosphate dehydrogenase GT1844 N-Acetylglucosamine N-acetylglucosamine-6-phosphate deacetylase GT2205 N-acetylglucosamine-6-phosphate isomerase GT2204 D-Maltose Alpha-1,4-glucosidase GT0528, GT1643 Sucrose Sucrose phosphorylase GT3215 D-Trehalose Alpha-glucosidase GT1643 Glucose kinase GT2381 Inositol Myo-inositol catabolism protein iolC;5-dehydro-2- GT1807 deoxygluconokinase -

(12) Patent Application Publication (10) Pub. No.: US 2006/0110747 A1 Ramseier Et Al
US 200601 10747A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2006/0110747 A1 Ramseier et al. (43) Pub. Date: May 25, 2006 (54) PROCESS FOR IMPROVED PROTEIN (60) Provisional application No. 60/591489, filed on Jul. EXPRESSION BY STRAIN ENGINEERING 26, 2004. (75) Inventors: Thomas M. Ramseier, Poway, CA Publication Classification (US); Hongfan Jin, San Diego, CA (51) Int. Cl. (US); Charles H. Squires, Poway, CA CI2O I/68 (2006.01) (US) GOIN 33/53 (2006.01) CI2N 15/74 (2006.01) Correspondence Address: (52) U.S. Cl. ................................ 435/6: 435/7.1; 435/471 KING & SPALDING LLP 118O PEACHTREE STREET (57) ABSTRACT ATLANTA, GA 30309 (US) This invention is a process for improving the production levels of recombinant proteins or peptides or improving the (73) Assignee: Dow Global Technologies Inc., Midland, level of active recombinant proteins or peptides expressed in MI (US) host cells. The invention is a process of comparing two genetic profiles of a cell that expresses a recombinant (21) Appl. No.: 11/189,375 protein and modifying the cell to change the expression of a gene product that is upregulated in response to the recom (22) Filed: Jul. 26, 2005 binant protein expression. The process can improve protein production or can improve protein quality, for example, by Related U.S. Application Data increasing solubility of a recombinant protein. Patent Application Publication May 25, 2006 Sheet 1 of 15 US 2006/0110747 A1 Figure 1 09 010909070£020\,0 10°0 Patent Application Publication May 25, 2006 Sheet 2 of 15 US 2006/0110747 A1 Figure 2 Ester sers Custer || || || || || HH-I-H 1 H4 s a cisiers TT closers | | | | | | Ya S T RXFO 1961. -

Structure of the Human Signal Peptidase Complex Reveals the Determinants for Signal Peptide Cleavage
bioRxiv preprint doi: https://doi.org/10.1101/2020.11.11.378711; this version posted November 11, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Structure of the Human Signal Peptidase Complex Reveals the Determinants for Signal Peptide Cleavage A. Manuel Liaci1, Barbara Steigenberger2,3, Sem Tamara2,3, Paulo Cesar Telles de Souza4, Mariska Gröllers-Mulderij1, Patrick Ogrissek1,5, Siewert J. Marrink4, Richard A. Scheltema2,3, Friedrich Förster1* Abstract The signal peptidase complex (SPC) is an essential membrane complex in the endoplasmic reticulum (ER), where it removes signal peptides (SPs) from a large variety of secretory pre-proteins with exquisite specificity. Although the determinants of this process have been established empirically, the molecular details of SP recognition and removal remain elusive. Here, we show that the human SPC exists in two functional paralogs with distinct proteolytic subunits. We determined the atomic structures of both paralogs using electron cryo- microscopy and structural proteomics. The active site is formed by a catalytic triad and abuts the ER membrane, where a transmembrane window collectively formed by all subunits locally thins the bilayer. This unique architecture generates specificity for thousands of SPs based on the length of their hydrophobic segments. Keywords Signal Peptidase Complex, Signal Peptide, Protein Maturation, Membrane Thinning, cryo-EM, Crosslinking Mass Spectrometry, Molecular Dynamics Simulations, Protein Secretion, ER Translocon 1Cryo-Electron Microscopy, Bijvoet Centre for Biomolecular Research, Utrecht University, Padualaan 8, 3584 CH Utrecht, The Netherlands. -

The Microbiota-Produced N-Formyl Peptide Fmlf Promotes Obesity-Induced Glucose
Page 1 of 230 Diabetes Title: The microbiota-produced N-formyl peptide fMLF promotes obesity-induced glucose intolerance Joshua Wollam1, Matthew Riopel1, Yong-Jiang Xu1,2, Andrew M. F. Johnson1, Jachelle M. Ofrecio1, Wei Ying1, Dalila El Ouarrat1, Luisa S. Chan3, Andrew W. Han3, Nadir A. Mahmood3, Caitlin N. Ryan3, Yun Sok Lee1, Jeramie D. Watrous1,2, Mahendra D. Chordia4, Dongfeng Pan4, Mohit Jain1,2, Jerrold M. Olefsky1 * Affiliations: 1 Division of Endocrinology & Metabolism, Department of Medicine, University of California, San Diego, La Jolla, California, USA. 2 Department of Pharmacology, University of California, San Diego, La Jolla, California, USA. 3 Second Genome, Inc., South San Francisco, California, USA. 4 Department of Radiology and Medical Imaging, University of Virginia, Charlottesville, VA, USA. * Correspondence to: 858-534-2230, [email protected] Word Count: 4749 Figures: 6 Supplemental Figures: 11 Supplemental Tables: 5 1 Diabetes Publish Ahead of Print, published online April 22, 2019 Diabetes Page 2 of 230 ABSTRACT The composition of the gastrointestinal (GI) microbiota and associated metabolites changes dramatically with diet and the development of obesity. Although many correlations have been described, specific mechanistic links between these changes and glucose homeostasis remain to be defined. Here we show that blood and intestinal levels of the microbiota-produced N-formyl peptide, formyl-methionyl-leucyl-phenylalanine (fMLF), are elevated in high fat diet (HFD)- induced obese mice. Genetic or pharmacological inhibition of the N-formyl peptide receptor Fpr1 leads to increased insulin levels and improved glucose tolerance, dependent upon glucagon- like peptide-1 (GLP-1). Obese Fpr1-knockout (Fpr1-KO) mice also display an altered microbiome, exemplifying the dynamic relationship between host metabolism and microbiota.