LY2875358), As Monotherapy and in Combination with Erlotinib in Advanced Cancer Lee S
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Lung Cancer Drugs in the Pipeline
HemOnc today | JANUARY 10, 2016 | Healio.com/HemOnc 5 Lung Cancer Drugs in the Pipeline HEMONC TODAY presents this guide to drugs in phase 2 or phase 3 development for lung cancer-related indications. Clinicians can use this chart as a quick reference to learn about the status of those drugs that may be clinically significant to their practice. Generic name (Brand name, Manufacturer) Indication(s) Development status abemaciclib (Eli Lilly) non–small cell lung cancer phase 3 ABP 215 (Allergan/Amgen) non–small cell lung cancer (advanced disease) phase 3 ACP-196 (Acerta Pharma) non–small cell lung cancer (advanced disease) phase 2 ado-trastuzumab emtansine (Kadcyla, Genentech) non–small cell lung cancer (HER-2–positive disease) phase 2 afatinib (Gilotrif, Boehringer Ingelheim) lung cancer (squamous cell carcinoma) phase 3 aldoxorubicin (CytRx) small cell lung cancer phase 2 alectinib (Alecensa, Genentech) non–small cell lung cancer (second-line treatment of ALK-positive disease) phase 2 non–small cell lung cancer (first-line treatment of ALK-positive disease); phase 3 alisertib (Takeda) malignant mesothelioma, small cell lung cancer phase 2 avelumab (EMD Serono/Pfizer) non–small cell lung cancer phase 3 AZD9291 (AstraZeneca) non–small cell lung cancer (first-line treatment of advancedEGFR -positive disease; phase 3 second-line treatment of advanced EGFR-positive, T790M-positive disease) bavituximab (Peregrine Pharmaceuticals) non–small cell lung cancer (previously treated advanced/metastatic disease) phase 3 belinostat (Beleodaq, Spectrum -

Predictive QSAR Tools to Aid in Early Process Development of Monoclonal Antibodies
Predictive QSAR tools to aid in early process development of monoclonal antibodies John Micael Andreas Karlberg Published work submitted to Newcastle University for the degree of Doctor of Philosophy in the School of Engineering November 2019 Abstract Monoclonal antibodies (mAbs) have become one of the fastest growing markets for diagnostic and therapeutic treatments over the last 30 years with a global sales revenue around $89 billion reported in 2017. A popular framework widely used in pharmaceutical industries for designing manufacturing processes for mAbs is Quality by Design (QbD) due to providing a structured and systematic approach in investigation and screening process parameters that might influence the product quality. However, due to the large number of product quality attributes (CQAs) and process parameters that exist in an mAb process platform, extensive investigation is needed to characterise their impact on the product quality which makes the process development costly and time consuming. There is thus an urgent need for methods and tools that can be used for early risk-based selection of critical product properties and process factors to reduce the number of potential factors that have to be investigated, thereby aiding in speeding up the process development and reduce costs. In this study, a framework for predictive model development based on Quantitative Structure- Activity Relationship (QSAR) modelling was developed to link structural features and properties of mAbs to Hydrophobic Interaction Chromatography (HIC) retention times and expressed mAb yield from HEK cells. Model development was based on a structured approach for incremental model refinement and evaluation that aided in increasing model performance until becoming acceptable in accordance to the OECD guidelines for QSAR models. -

2017 Immuno-Oncology Medicines in Development
2017 Immuno-Oncology Medicines in Development Adoptive Cell Therapies Drug Name Organization Indication Development Phase ACTR087 + rituximab Unum Therapeutics B-cell lymphoma Phase I (antibody-coupled T-cell receptor Cambridge, MA www.unumrx.com immunotherapy + rituximab) AFP TCR Adaptimmune liver Phase I (T-cell receptor cell therapy) Philadelphia, PA www.adaptimmune.com anti-BCMA CAR-T cell therapy Juno Therapeutics multiple myeloma Phase I Seattle, WA www.junotherapeutics.com Memorial Sloan Kettering New York, NY anti-CD19 "armored" CAR-T Juno Therapeutics recurrent/relapsed chronic Phase I cell therapy Seattle, WA lymphocytic leukemia (CLL) www.junotherapeutics.com Memorial Sloan Kettering New York, NY anti-CD19 CAR-T cell therapy Intrexon B-cell malignancies Phase I Germantown, MD www.dna.com ZIOPHARM Oncology www.ziopharm.com Boston, MA anti-CD19 CAR-T cell therapy Kite Pharma hematological malignancies Phase I (second generation) Santa Monica, CA www.kitepharma.com National Cancer Institute Bethesda, MD Medicines in Development: Immuno-Oncology 1 Adoptive Cell Therapies Drug Name Organization Indication Development Phase anti-CEA CAR-T therapy Sorrento Therapeutics liver metastases Phase I San Diego, CA www.sorrentotherapeutics.com TNK Therapeutics San Diego, CA anti-PSMA CAR-T cell therapy TNK Therapeutics cancer Phase I San Diego, CA www.sorrentotherapeutics.com Sorrento Therapeutics San Diego, CA ATA520 Atara Biotherapeutics multiple myeloma, Phase I (WT1-specific T lymphocyte South San Francisco, CA plasma cell leukemia www.atarabio.com -

The Role of MET Inhibitor Therapies in the Treatment of Advanced Non-Small Cell Lung Cancer
Journal of Clinical Medicine Review The Role of MET Inhibitor Therapies in the Treatment of Advanced Non-Small Cell Lung Cancer Ramon Andrade De Mello 1,2,3,* , Nathália Moisés Neves 2 , Giovanna Araújo Amaral 2, Estela Gudin Lippo 4, Pedro Castelo-Branco 1, Daniel Humberto Pozza 5 , Carla Chizuru Tajima 6 and Georgios Antoniou 7 1 Algarve Biomedical Centre, Department of Biomedical Sciences and Medicine University of Algarve (DCBM UALG), 8005-139 Faro, Portugal; [email protected] 2 Division of Medical Oncology, Escola Paulista de Medicina, Federal University of São Paulo (UNIFESP), São Paulo 04037-004, Brazil; [email protected] (N.M.N.); [email protected] (G.A.A.) 3 Precision Oncology and Health Economics Group (ONCOPRECH), Post-Graduation Program in Medicine, Nine of July University (UNINOVE), São Paulo 01525-000, Brazil 4 School of Biomedical Sciences, Santo Amaro University, São Paulo 01525-000, Brazil; [email protected] 5 Department of Biomedicine & I3S, Faculty of Medicine, University of Porto (FMUP), 4200-317 Porto, Portugal; [email protected] 6 Hospital São José & Hospital São Joaquim, A Beneficência Portuguesa de São Paulo, São Paulo 01323-001, Brazil; [email protected] 7 Division of Medical Oncology, Mount Vernon Cancer Center, London HA6 2RN, UK; [email protected] * Correspondence: [email protected] Received: 15 May 2020; Accepted: 10 June 2020; Published: 19 June 2020 Abstract: Introduction: Non-small cell lung cancer (NSCLC) is the second most common cancer globally. The mesenchymal-epithelial transition (MET) proto-oncogene can be targeted in NSCLC patients. Methods: We performed a literature search on PubMed in December 2019 for studies on MET inhibitors and NSCLC. -

Atezolizumab Plus Nab-Paclitaxel As First-Line Treatment for Unresectable, Locally Advanced Or Metastatic Triple-Negative Breast
Articles Atezolizumab plus nab-paclitaxel as first-line treatment for unresectable, locally advanced or metastatic triple-negative breast cancer (IMpassion130): updated efficacy results from a randomised, double-blind, placebo-controlled, phase 3 trial Peter Schmid*, Hope S Rugo*, Sylvia Adams, Andreas Schneeweiss, Carlos H Barrios, Hiroji Iwata, Véronique Diéras, Volkmar Henschel, Luciana Molinero, Stephen Y Chui, Vidya Maiya, Amreen Husain, Eric P Winer, Sherene Loi, Leisha A Emens, for the IMpassion130 Investigators† Summary Lancet Oncol 2020; 21: 44–59 Background Immunotherapy in combination with chemotherapy has shown promising efficacy across many different Published Online tumour types. We report the prespecified second interim overall survival analysis of the phase 3 IMpassion130 study November 27, 2019 assessing the efficacy and safety of atezolizumab plus nab-paclitaxel in patients with unresectable, locally advanced or https://doi.org/10.1016/ metastatic triple-negative breast cancer. S1470-2045(19)30689-8 See Comment page 3 Methods In this randomised, placebo-controlled, double-blind, phase 3 trial, done in 246 academic centres and *Contributed equally community oncology practices in 41 countries, patients aged 18 years or older, with previously untreated, histologically †Investigators are listed in the documented, locally advanced or metastatic triple-negative breast cancer, and Eastern Cooperative Oncology Group appendix performance status of 0 or 1 were eligible. Patients were randomly assigned (1:1) using a permuted block method Barts Cancer Institute, Queen Mary University of London, (block size of four) and an interactive voice–web response system. Randomisation was stratified by previous taxane London, UK (Prof P Schmid MD); use, liver metastases, and PD-L1 expression on tumour-infiltrating immune cells. -

The Two Tontti Tudiul Lui Hi Ha Unit
THETWO TONTTI USTUDIUL 20170267753A1 LUI HI HA UNIT ( 19) United States (12 ) Patent Application Publication (10 ) Pub. No. : US 2017 /0267753 A1 Ehrenpreis (43 ) Pub . Date : Sep . 21 , 2017 ( 54 ) COMBINATION THERAPY FOR (52 ) U .S . CI. CO - ADMINISTRATION OF MONOCLONAL CPC .. .. CO7K 16 / 241 ( 2013 .01 ) ; A61K 39 / 3955 ANTIBODIES ( 2013 .01 ) ; A61K 31 /4706 ( 2013 .01 ) ; A61K 31 / 165 ( 2013 .01 ) ; CO7K 2317 /21 (2013 . 01 ) ; (71 ) Applicant: Eli D Ehrenpreis , Skokie , IL (US ) CO7K 2317/ 24 ( 2013. 01 ) ; A61K 2039/ 505 ( 2013 .01 ) (72 ) Inventor : Eli D Ehrenpreis, Skokie , IL (US ) (57 ) ABSTRACT Disclosed are methods for enhancing the efficacy of mono (21 ) Appl. No. : 15 /605 ,212 clonal antibody therapy , which entails co - administering a therapeutic monoclonal antibody , or a functional fragment (22 ) Filed : May 25 , 2017 thereof, and an effective amount of colchicine or hydroxy chloroquine , or a combination thereof, to a patient in need Related U . S . Application Data thereof . Also disclosed are methods of prolonging or increasing the time a monoclonal antibody remains in the (63 ) Continuation - in - part of application No . 14 / 947 , 193 , circulation of a patient, which entails co - administering a filed on Nov. 20 , 2015 . therapeutic monoclonal antibody , or a functional fragment ( 60 ) Provisional application No . 62/ 082, 682 , filed on Nov . of the monoclonal antibody , and an effective amount of 21 , 2014 . colchicine or hydroxychloroquine , or a combination thereof, to a patient in need thereof, wherein the time themonoclonal antibody remains in the circulation ( e . g . , blood serum ) of the Publication Classification patient is increased relative to the same regimen of admin (51 ) Int . -

(12) Patent Application Publication (10) Pub. No.: US 2017/0172932 A1 Peyman (43) Pub
US 20170172932A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2017/0172932 A1 Peyman (43) Pub. Date: Jun. 22, 2017 (54) EARLY CANCER DETECTION AND A 6LX 39/395 (2006.01) ENHANCED IMMUNOTHERAPY A61R 4I/00 (2006.01) (52) U.S. Cl. (71) Applicant: Gholam A. Peyman, Sun City, AZ CPC .......... A61K 9/50 (2013.01); A61K 39/39558 (US) (2013.01); A61K 4I/0052 (2013.01); A61 K 48/00 (2013.01); A61K 35/17 (2013.01); A61 K (72) Inventor: sham A. Peyman, Sun City, AZ 35/15 (2013.01); A61K 2035/124 (2013.01) (21) Appl. No.: 15/143,981 (57) ABSTRACT (22) Filed: May 2, 2016 A method of therapy for a tumor or other pathology by administering a combination of thermotherapy and immu Related U.S. Application Data notherapy optionally combined with gene delivery. The combination therapy beneficially treats the tumor and pre (63) Continuation-in-part of application No. 14/976,321, vents tumor recurrence, either locally or at a different site, by filed on Dec. 21, 2015. boosting the patient’s immune response both at the time or original therapy and/or for later therapy. With respect to Publication Classification gene delivery, the inventive method may be used in cancer (51) Int. Cl. therapy, but is not limited to such use; it will be appreciated A 6LX 9/50 (2006.01) that the inventive method may be used for gene delivery in A6 IK 35/5 (2006.01) general. The controlled and precise application of thermal A6 IK 4.8/00 (2006.01) energy enhances gene transfer to any cell, whether the cell A 6LX 35/7 (2006.01) is a neoplastic cell, a pre-neoplastic cell, or a normal cell. -

Overcoming MET-Dependent Resistance to Selective RET Inhibition in Patients with RET Fusion–Positive Lung Cancer by Combining Selpercatinib with Crizotinib a C Ezra Y
Published OnlineFirst October 20, 2020; DOI: 10.1158/1078-0432.CCR-20-2278 CLINICAL CANCER RESEARCH | RESEARCH BRIEFS: CLINICAL TRIAL BRIEF REPORT Overcoming MET-Dependent Resistance to Selective RET Inhibition in Patients with RET Fusion–Positive Lung Cancer by Combining Selpercatinib with Crizotinib A C Ezra Y. Rosen1, Melissa L. Johnson2, Sarah E. Clifford3, Romel Somwar4, Jennifer F. Kherani5, Jieun Son3, Arrien A. Bertram3, Monika A. Davare6, Eric Gladstone4, Elena V. Ivanova7, Dahlia N. Henry5, Elaine M. Kelley3, Mika Lin3, Marina S.D. Milan3, Binoj C. Nair5, Elizabeth A. Olek5, Jenna E. Scanlon3, Morana Vojnic4, Kevin Ebata5, Jaclyn F. Hechtman4, Bob T. Li1,8, Lynette M. Sholl9, Barry S. Taylor10, Marc Ladanyi4, Pasi A. Janne€ 3, S. Michael Rothenberg5, Alexander Drilon1,8, and Geoffrey R. Oxnard3 ABSTRACT ◥ Purpose: The RET proto-oncogene encodes a receptor tyrosine Results: MET amplification was identified in posttreatment kinase that is activated by gene fusion in 1%–2% of non–small biopsies in 4 patients with RET fusion–positive NSCLC treated with cell lung cancers (NSCLC) and rarely in other cancer types. selpercatinib. In at least one case, MET amplification was clearly Selpercatinib is a highly selective RET kinase inhibitor that has evident prior to therapy with selpercatinib. We demonstrate that recently been approved by the FDA in lung and thyroid cancers increased MET expression in RET fusion–positive tumor cells causes with activating RET gene fusions and mutations. Molecular resistance to selpercatinib, and this can be overcome by combining mechanisms of acquired resistance to selpercatinib are poorly selpercatinib with crizotinib. Using SPPs, selpercatinib with crizo- understood. -

WO 2016/176089 Al 3 November 2016 (03.11.2016) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2016/176089 Al 3 November 2016 (03.11.2016) P O P C T (51) International Patent Classification: BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, A01N 43/00 (2006.01) A61K 31/33 (2006.01) DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, HN, HR, HU, ID, IL, IN, IR, IS, JP, KE, KG, KN, KP, KR, (21) International Application Number: KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, MG, PCT/US2016/028383 MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, (22) International Filing Date: PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, SC, 20 April 2016 (20.04.2016) SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (25) Filing Language: English (84) Designated States (unless otherwise indicated, for every (26) Publication Language: English kind of regional protection available): ARIPO (BW, GH, (30) Priority Data: GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, 62/154,426 29 April 2015 (29.04.2015) US TZ, UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, (71) Applicant: KARDIATONOS, INC. [US/US]; 4909 DK, EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, Lapeer Road, Metamora, Michigan 48455 (US). -

Acquired Resistance to a MET Antibody in Vivo Can Be Overcome by the MET Antibody Mixture Sym015
Author Manuscript Published OnlineFirst on March 15, 2018; DOI: 10.1158/1535-7163.MCT-17-0787 Author manuscripts have been peer reviewed and accepted for publication but have not yet been edited. Acquired resistance to a MET antibody in vivo can be overcome by the MET antibody mixture Sym015 Authors: Sofie Ellebæk Pollmann1, Valerie S. Calvert2, Shruti Rao3, Simina M. Boca3, Subha Madhavan3, Ivan D. Horak1, Andreas Kjær4, Emanuel F. Petricoin2, Michael Kragh1, and Thomas Tuxen Poulsen1 Authors’ affiliations: 1Symphogen A/S, Ballerup, Denmark 2Center for Applied Proteomics and Molecular Medicine, George Mason University, Virginia, USA 3Innovation Center for Biomedical Informatics, Georgetown University, Washington DC, USA 4Dept. of Clinical Physiology, Nuclear Medicine & PET and Cluster for Molecular Imaging, Dept. of Biomedical Sciences, Rigshospitalet and University of Copenhagen, Denmark Running title: In vivo acquired resistance to a MET-targeting antibody. Keywords: Antibody, MET, Resistance, in vivo, RPPA Corresponding author: Sofie Ellebæk Pollmann Symphogen A/S Pederstrupvej 93 DK-2750 Ballerup, Denmark E-mail: [email protected] Tel: +4545265050 Disclosure of potential conflicts of interest: Sofie Ellebæk Pollmann, Ivan D. Horak, Michael Kragh, and Thomas Tuxen Poulsen are all employed by Symphogen A/S. Subha Madhavan is a member of the scientific advisory board of and a consultant for Perthera. Emanuel F. Petricoin is chief scientific officer, co-founder, member of board of directors of Perthera, of which he has ownership interests. He is also a member of the scientific advisory board of/consultant for Perthera and Avant Diagnotics. 1 Downloaded from mct.aacrjournals.org on September 30, 2021. © 2018 American Association for Cancer Research. -

Small-Cell Lung Cancer: Evidence to Date Olivier Bylicki, Nicolas Paleiron, Jean-Baptiste Assié, Christos Chouaid
Targeting the MET-Signaling Pathway in Non- Small-Cell Lung Cancer: Evidence to Date Olivier Bylicki, Nicolas Paleiron, Jean-Baptiste Assié, Christos Chouaid To cite this version: Olivier Bylicki, Nicolas Paleiron, Jean-Baptiste Assié, Christos Chouaid. Targeting the MET-Signaling Pathway in Non- Small-Cell Lung Cancer: Evidence to Date. OncoTargets and Therapy, Dove Medical Press, 2020, Volume 13, pp.5691-5706. 10.2147/OTT.S219959. hal-02886104 HAL Id: hal-02886104 https://hal.sorbonne-universite.fr/hal-02886104 Submitted on 1 Jul 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. OncoTargets and Therapy Dovepress open access to scientific and medical research Open Access Full Text Article REVIEW Targeting the MET-Signaling Pathway in Non- Small–Cell Lung Cancer: Evidence to Date This article was published in the following Dove Press journal: OncoTargets and Therapy Olivier Bylicki 1,2 Abstract: The c-MET proto-oncogene (MET) plays an important role in lung oncogenesis, Nicolas Paleiron1 affecting cancer-cell survival, growth and invasiveness. The MET receptor in non-small–cell – Jean-Baptiste Assié2 4 lung cancer (NSCLC) is a potential therapeutic target. The development of high-output next- fi Christos Chouaïd2,3 generation sequencing techniques has enabled better identi cation of anomalies in the MET pathway, like the MET exon-14 (METex14) mutation. -
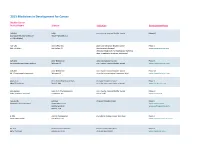
Adis R&D Insight
2015 Medicines in Development for Cancer Bladder Cancer Product Name Sponsor Indication Development Phase ABI-009 AADi non-muscle invasive bladder cancer Phase I/II (nanoparticle albumin-bound Pacific Palisades, CA mTOR inhibitor) ACP-196 Acerta Pharma platinum-refractory bladder cancer Phase II (Btk inhibitor) San Carlos, CA (combination therapy) www.acerta-pharma.com (see also head/neck, hematological, leukemia, lung, lymphoma, myeloma, pancreatic) ALT-801 Altor BioScience advanced bladder cancer, Phase II (immunotherapy fusion protein) Miramar, FL non-muscle invasive bladder cancer www.altorbioscience.com ALT-803 Altor BioScience non-muscle invasive bladder cancer Phase I/II (IL-15 superagonist complex) Miramar, FL (see also hematological, myeloma, skin) www.altorbioscience.com apatorsen OncoGenex Pharmaceuticals metastatic bladder cancer Phase II (Hsp27 inhibitor) Bothell, WA (see also lung, pancreatic, prostate) www.oncogenex.com apaziquone Spectrum Pharmaceuticals non-muscle invasive bladder cancer Phase III (DNA synthesis inhibitor) Henderson, NV (Fast Track) www.sppirx.com ASG-15ME Agensys relapsed bladder cancer Phase I (antibody drug conjugate) Santa Monica, CA www.agensys.com Seattle Genetics www.seattlegenetics.com Bothell, WA B-701 BioClin Therapeutics metastatic bladder cancer (2nd-line) Phase II (anti-FGFR3 mAb) San Ramon, CA www.bioclintherapeutics.com BC-819 BioCancell Therapeutics bladder cancer (2nd-line) Phase II (gene therapy) Jerusalem, Israel (see also pancreatic) www.biocancell.com Bladder Cancer Product Name Sponsor