Looking Back at the Early Stages of Redox Biology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Copper Protein and a Cytochrome Bind at the Same Site on Bacterial Cytochrome C Peroxidase† Sofia R
14566 Biochemistry 2004, 43, 14566-14576 A Copper Protein and a Cytochrome Bind at the Same Site on Bacterial Cytochrome c Peroxidase† Sofia R. Pauleta,‡,§ Alan Cooper,⊥ Margaret Nutley,⊥ Neil Errington,| Stephen Harding,| Francoise Guerlesquin,3 Celia F. Goodhew,‡ Isabel Moura,§ Jose J. G. Moura,§ and Graham W. Pettigrew‡ Veterinary Biomedical Sciences, Royal (Dick) School of Veterinary Studies, UniVersity of Edinburgh, Summerhall, Edinburgh EH9 1QH, U.K., Department of Chemistry, UniVersity of Glasgow, Glasgow G12 8QQ, U.K., Centre for Macromolecular Hydrodynamics, UniVersity of Nottingham, Sutton Bonington, Nottingham LE12 5 RD, U.K., Unite de Bioenergetique et Ingenierie des Proteines, IBSM-CNRS, 31 chemin Joseph Aiguier, 13402 Marseilles cedex 20, France, Requimte, Departamento de Quimica, CQFB, UniVersidade NoVa de Lisboa, 2829-516 Monte de Caparica, Portugal ReceiVed July 5, 2004; ReVised Manuscript ReceiVed September 9, 2004 ABSTRACT: Pseudoazurin binds at a single site on cytochrome c peroxidase from Paracoccus pantotrophus with a Kd of 16.4 µMat25°C, pH 6.0, in an endothermic reaction that is driven by a large entropy change. Sedimentation velocity experiments confirmed the presence of a single site, although results at higher pseudoazurin concentrations are complicated by the dimerization of the protein. Microcalorimetry, ultracentrifugation, and 1H NMR spectroscopy studies in which cytochrome c550, pseudoazurin, and cytochrome c peroxidase were all present could be modeled using a competitive binding algorithm. Molecular docking simulation of the binding of pseudoazurin to the peroxidase in combination with the chemical shift perturbation pattern for pseudoazurin in the presence of the peroxidase revealed a group of solutions that were situated close to the electron-transferring heme with Cu-Fe distances of about 14 Å. -

The Role of Glutathionperoxidase 4 (GPX4) in Hematopoiesis and Leukemia
The role of glutathionperoxidase 4 (GPX4) in hematopoiesis and leukemia von Kira Célénie Stahnke Inaugral- Dissertation zur Erlangung der Doktorwürde der TierärztliChen Fakultät der Ludwig-Maximilians-Universität MünChen The role of glutathionperoxidase 4 (GPX4) in hematopoiesis and leukemia von Kira Célénie Stahnke aus Dortmund München 2016 Aus dem VeterinärwissensChaftliChen Department der TierärztliChen Fakultät der Ludwig-Maximilians-Universität München Lehrstuhl für Molekulare Tierzucht und Biotechnologie Arbeit angefertigt unter der Leitung von Univ.-Prof. Dr. Eckhard Wolf Angefertigt am Institut für Experimentelle TumorforsChung Universitätsklinikum Ulm Mentor: Prof. Dr. Christian Buske GedruCkt mit Genehmigung der TierärztliChen Fakultät der Ludwig-Maximilians-Universität München Dekan: Univ.-Prof. Dr. JoaChim Braun Berichterstatter: Univ.-Prof. Dr. Eckard Wolf Korreferent/en: Priv.-Doz. Dr. Bianka Schulz Tag der Promotion: 06.02.2016 Table of contents List of Abbreviations..............................................................................2 1 Introduction ........................................................................................6 1.1 ROS and oxidative stress in physiology and pathology..................................................... 6 1.1.1 SourCes of Reactive Oxygen SpeCies..................................................................................................6 1.1.2 PhysiologiCal role of ROS........................................................................................................................8 -

(ER) Membrane Contact Sites (MCS) Uses Toxic Waste to Deliver Messages Edgar Djaha Yoboue1, Roberto Sitia1 and Thomas Simmen2
Yoboue et al. Cell Death and Disease (2018) 9:331 DOI 10.1038/s41419-017-0033-4 Cell Death & Disease REVIEW ARTICLE Open Access Redox crosstalk at endoplasmic reticulum (ER) membrane contact sites (MCS) uses toxic waste to deliver messages Edgar Djaha Yoboue1, Roberto Sitia1 and Thomas Simmen2 Abstract Many cellular redox reactions housed within mitochondria, peroxisomes and the endoplasmic reticulum (ER) generate hydrogen peroxide (H2O2) and other reactive oxygen species (ROS). The contribution of each organelle to the total cellular ROS production is considerable, but varies between cell types and also over time. Redox-regulatory enzymes are thought to assemble at a “redox triangle” formed by mitochondria, peroxisomes and the ER, assembling “redoxosomes” that sense ROS accumulations and redox imbalances. The redoxosome enzymes use ROS, potentially toxic by-products made by some redoxosome members themselves, to transmit inter-compartmental signals via chemical modifications of downstream proteins and lipids. Interestingly, important components of the redoxosome are ER chaperones and oxidoreductases, identifying ER oxidative protein folding as a key ROS producer and controller of the tri-organellar membrane contact sites (MCS) formed at the redox triangle. At these MCS, ROS accumulations could directly facilitate inter-organellar signal transmission, using ROS transporters. In addition, ROS influence the flux 2+ 2+ of Ca ions, since many Ca handling proteins, including inositol 1,4,5 trisphosphate receptors (IP3Rs), SERCA pumps or regulators of the mitochondrial Ca2+ uniporter (MCU) are redox-sensitive. Fine-tuning of these redox and ion signaling pathways might be difficult in older organisms, suggesting a dysfunctional redox triangle may accompany 1234567890 1234567890 the aging process. -

Independent Evolution of Four Heme Peroxidase Superfamilies
Archives of Biochemistry and Biophysics xxx (2015) xxx–xxx Contents lists available at ScienceDirect Archives of Biochemistry and Biophysics journal homepage: www.elsevier.com/locate/yabbi Independent evolution of four heme peroxidase superfamilies ⇑ Marcel Zámocky´ a,b, , Stefan Hofbauer a,c, Irene Schaffner a, Bernhard Gasselhuber a, Andrea Nicolussi a, Monika Soudi a, Katharina F. Pirker a, Paul G. Furtmüller a, Christian Obinger a a Department of Chemistry, Division of Biochemistry, VIBT – Vienna Institute of BioTechnology, University of Natural Resources and Life Sciences, Muthgasse 18, A-1190 Vienna, Austria b Institute of Molecular Biology, Slovak Academy of Sciences, Dúbravská cesta 21, SK-84551 Bratislava, Slovakia c Department for Structural and Computational Biology, Max F. Perutz Laboratories, University of Vienna, A-1030 Vienna, Austria article info abstract Article history: Four heme peroxidase superfamilies (peroxidase–catalase, peroxidase–cyclooxygenase, peroxidase–chlo- Received 26 November 2014 rite dismutase and peroxidase–peroxygenase superfamily) arose independently during evolution, which and in revised form 23 December 2014 differ in overall fold, active site architecture and enzymatic activities. The redox cofactor is heme b or Available online xxxx posttranslationally modified heme that is ligated by either histidine or cysteine. Heme peroxidases are found in all kingdoms of life and typically catalyze the one- and two-electron oxidation of a myriad of Keywords: organic and inorganic substrates. In addition to this peroxidatic activity distinct (sub)families show pro- Heme peroxidase nounced catalase, cyclooxygenase, chlorite dismutase or peroxygenase activities. Here we describe the Peroxidase–catalase superfamily phylogeny of these four superfamilies and present the most important sequence signatures and active Peroxidase–cyclooxygenase superfamily Peroxidase–chlorite dismutase superfamily site architectures. -
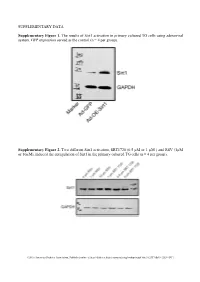
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -

GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis Giovanni C
REVIEW GPX4 www.proteomics-journal.com GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis Giovanni C. Forcina and Scott J. Dixon* formation of toxic radicals (e.g., R-O•).[5] Oxygen is necessary for aerobic metabolism but can cause the harmful The eight mammalian GPX proteins fall oxidation of lipids and other macromolecules. Oxidation of cholesterol and into three clades based on amino acid phospholipids containing polyunsaturated fatty acyl chains can lead to lipid sequence similarity: GPX1 and GPX2; peroxidation, membrane damage, and cell death. Lipid hydroperoxides are key GPX3, GPX5, and GPX6; and GPX4, GPX7, and GPX8.[6] GPX1–4 and 6 (in intermediates in the process of lipid peroxidation. The lipid hydroperoxidase humans) are selenoproteins that contain glutathione peroxidase 4 (GPX4) converts lipid hydroperoxides to lipid an essential selenocysteine in the enzyme + alcohols, and this process prevents the iron (Fe2 )-dependent formation of active site, while GPX5, 6 (in mouse and toxic lipid reactive oxygen species (ROS). Inhibition of GPX4 function leads to rats), 7, and 8 use an active site cysteine lipid peroxidation and can result in the induction of ferroptosis, an instead. Unlike other family members, GPX4 (PHGPx) can act as a phospholipid iron-dependent, non-apoptotic form of cell death. This review describes the hydroperoxidase to reduce lipid perox- formation of reactive lipid species, the function of GPX4 in preventing ides to lipid alcohols.[7,8] Thus,GPX4ac- oxidative lipid damage, and the link between GPX4 dysfunction, lipid tivity is essential to maintain lipid home- oxidation, and the induction of ferroptosis. ostasis in the cell, prevent the accumula- tion of toxic lipid ROS and thereby block the onset of an oxidative, iron-dependent, non-apoptotic mode of cell death termed 1. -

Thiol Peroxidases Mediate Specific Genome-Wide Regulation of Gene Expression in Response to Hydrogen Peroxide
Thiol peroxidases mediate specific genome-wide regulation of gene expression in response to hydrogen peroxide Dmitri E. Fomenkoa,1,2, Ahmet Koca,1, Natalia Agishevaa, Michael Jacobsena,b, Alaattin Kayaa,c, Mikalai Malinouskia,c, Julian C. Rutherfordd, Kam-Leung Siue, Dong-Yan Jine, Dennis R. Winged, and Vadim N. Gladysheva,c,2 aDepartment of Biochemistry, University of Nebraska, Lincoln, NE 68588-0664; bDepartment of Life Sciences, Wayne State College, Wayne, NE 68787; dDepartment of Medicine, University of Utah Health Sciences Center, Salt Lake City, UT 84132; eDepartment of Biochemistry, University of Hong Kong, Hong Kong, China; and cDivision of Genetics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA 02115 Edited by Joan Selverstone Valentine, University of California, Los Angeles, CA, and approved December 22, 2010 (received for review July 21, 2010) Hydrogen peroxide is thought to regulate cellular processes by and could withstand significant oxidative stress. It responded to direct oxidation of numerous cellular proteins, whereas antioxi- several redox stimuli by robust transcriptional reprogramming. dants, most notably thiol peroxidases, are thought to reduce However, it was unable to transcriptionally respond to hydrogen peroxides and inhibit H2O2 response. However, thiol peroxidases peroxide. The data suggested that thiol peroxidases transfer have also been implicated in activation of transcription factors oxidative signals from peroxides to target proteins, thus activating and signaling. It remains unclear if these enzymes stimulate or various transcriptional programs. This study revealed a previously inhibit redox regulation and whether this regulation is widespread undescribed function of these proteins, in addition to their roles or limited to a few cellular components. -

Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications
724 Journal of Food Protection, Vol. 47. No. 9, Pages 724-732 (September 1984) Copyright®, International Association of Milk, Food, and Environmental Sanitarians Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications BRUNO REITER1 and GORAN HARNULV2* Downloaded from http://meridian.allenpress.com/jfp/article-pdf/47/9/724/1650811/0362-028x-47_9_724.pdf by guest on 29 September 2021 National Institute for Research in Dairying, Shinfield, Reading, RG2 9AT, England and Alfa-Laval Agri International AS, P.O. Box 39, S-I47 00 Tumba, Sweden (Received for publication January 30, 1984) ABSTRACT (37,80). The various biological functions of the LP sys tem, which have now been established, will be discussed In the present review dealing with the antibacterial lac below. toperoxidase (LP) system, it is shown that the two reactants In recent years, much work has been devoted to practi thiocyanate (SCN~) and hydrogen peroxide (H202) as well as cal applications of the LP system. The effect against oral the catalytic enzyme lactoperoxidase (LP) are widely distributed streptococci (5,19,33,101,105,107) has led to the de in nature and that evidence for the activity of the LP system velopment and marketing of a toothpaste containing nec in animals, including man, is accumulating. The in vitro effects on bacterial and animal cells are discussed and the unique ac essary ingredients to activate the LP system. Promising tion of the LP system on the bacterial cytoplasmic membrane results have also been obtained when including activating is pointed out. Some practical applications are also presented, components in the feed to calves (81,83,86) with the aim with particular emphses on the possibility of utilizing the LP of potentiating the LP system in the intestinal tract. -

( 12 ) United States Patent
US010208322B2 (12 ) United States Patent ( 10 ) Patent No. : US 10 ,208 , 322 B2 Coelho et al. (45 ) Date of Patent: * Feb . 19, 2019 ( 54 ) IN VIVO AND IN VITRO OLEFIN ( 56 ) References Cited CYCLOPROPANATION CATALYZED BY HEME ENZYMES U . S . PATENT DOCUMENTS 3 , 965 ,204 A 6 / 1976 Lukas et al. (71 ) Applicant: California Institute of Technology , 4 , 243 ,819 A 1 / 1981 Henrick Pasadena , CA (US ) 5 ,296 , 595 A 3 / 1994 Doyle 5 , 703 , 246 A 12 / 1997 Aggarwal et al. 7 , 226 , 768 B2 6 / 2007 Farinas et al. ( 72 ) Inventors : Pedro S . Coelho , Los Angeles, CA 7 , 267 , 949 B2 9 / 2007 Richards et al . (US ) ; Eric M . Brustad , Durham , NC 7 ,625 ,642 B2 12 / 2009 Matsutani et al. (US ) ; Frances H . Arnold , La Canada , 7 ,662 , 969 B2 2 / 2010 Doyle et al. CA (US ) ; Zhan Wang , San Jose , CA 7 ,863 ,030 B2 1 / 2011 Arnold (US ) ; Jared C . Lewis , Chicago , IL 8 ,247 ,430 B2 8 / 2012 Yuan 8 , 993 , 262 B2 * 3 / 2015 Coelho . .. .. .. • * • C12P 7 /62 (US ) 435 / 119 9 ,399 , 762 B26 / 2016 Farwell et al . (73 ) Assignee : California Institute of Technology , 9 , 493 ,799 B2 * 11 /2016 Coelho .. C12P 7162 Pasadena , CA (US ) 2006 / 0030718 AL 2 / 2006 Zhang et al. 2006 / 0111347 A1 5 / 2006 Askew , Jr . et al. 2007 /0276013 AL 11 /2007 Ebbinghaus et al . ( * ) Notice : Subject to any disclaimer , the term of this 2009 /0238790 A2 9 /2009 Sommadosi et al. patent is extended or adjusted under 35 2010 / 0056806 A1 3 / 2010 Warren U . -

The C718T Polymorphism in the 3″-Untranslated
Hypertension Research (2012) 35, 507–512 & 2012 The Japanese Society of Hypertension All rights reserved 0916-9636/12 www.nature.com/hr ORIGINAL ARTICLE The C718T polymorphism in the 3¢-untranslated region of glutathione peroxidase-4 gene is a predictor of cerebral stroke in patients with essential hypertension Alexey V Polonikov1, Ekaterina K Vialykh2, Mikhail I Churnosov3, Thomas Illig4, Maxim B Freidin5, Oksana V Vasil¢eva1, Olga Yu Bushueva1, Valentina N Ryzhaeva1, Irina V Bulgakova1 and Maria A Solodilova1 In the present study we have investigated the association of three single nucleotide polymorphisms in glutathione peroxidase (GPx) genes GPX1 rs1050450 (P198L), GPX3 rs2070593 (G930A) and GPX4 rs713041 (T718C) with the risk of cerebral stroke (CS) in patients with essential hypertension (EH). A total of 667 unrelated EH patients of Russian origin, including 306 hypertensives (the EH–CS group) who suffered from CS and 361 people (the EH–CS group) who did not have cerebrovascular accidents, were enrolled in the study. The variant allele 718C of the GPX4 gene was found to be significantly associated with an increased risk of CS in hypertensive patients (odds ratio (OR) 1.53, 95% confidence interval (CI) 1.23–1.90, Padj¼0.0003). The prevalence of the 718TC and 718CC genotypes of the GPX4 gene was higher in the EH–CS group than the EH-alone group (OR¼2.12, 95%CI 1.42–3.16, Padj¼0.0018). The association of the variant GPX4 genotypes with the increased risk of CS in hypertensives remained statistically significant after adjusting for confounding variables such as sex, body mass index (BMI), blood pressure and antihypertensive medication use (OR¼2.18, 95%CI 1.46–3.27, P¼0.0015). -

Antioxidative Defense Mechanisms in the Aging Brain
Arch. Biol. Sci., Belgrade, 66 (1), 245-252, 2014 DOI:10.2298/ABS1401245J ANTIOXIDATIVE DEFENSE MECHANISMS IN THE AGING BRAIN ZORICA JOVANOVIĆ Department of Pathophysiology, Faculty of Medicine, 34000 Kragujevac, Serbia Abstract - Aging is an extremely complex, multifactorial process that is characterized by a gradual and continuous loss of physiological functions and responses, particularly marked in the brain. A common hallmark in aging and age-related diseases is an increase in oxidative stress and the failure of antioxidant defense systems. Current knowledge indicates that the level of glutathione progressively declines during aging. Because nerve cells are the longest-living cells that exhibit a high consumption rate of oxygen throughout an individual’s lifetime, the brain may be especially vulnerable to oxidative damage and this vulnerability increases during aging. In addition, the brain contains high concentrations of polyunsatu- rated fatty acids and transition metals and low antioxidative defense mechanisms. Although aging is an inevitable event, a growing volume of data confirms that antioxidant supplementation in combination with symptomatic drug treatments reduces oxidative stress and improves cognitive function in aging and age-related diseases. The present review discusses the neuroprotective effects of antioxidants in the aging brain. Key words: Aging, brain, oxidative stress, glutathione, polyphenols OXIDATIVE STRESS AND AGING cells and tissues with advancing age that increase the risk of disease and death”. Later, Harman added The aging process is associated with a progressive a slight modification to this theory to bring special decline in multiple aspects of cognitive perform- attention to the role of the mitochondria in the ag- ance, including reductions in mental speed, atten- ing process because these organelles are a major site tion, memory, hearing, vision and stamina, as well of reactive oxygen species (ROS) generation (Har- as decreases in brain structure size and white mat- man, 1972). -

The Catalytic Role of the Distal Site Asparagine-Histidine Couple in Catalase-Peroxidases
Eur. J. Biochem. 270, 1006–1013 (2003) Ó FEBS 2003 doi:10.1046/j.1432-1033.2003.03476.x The catalytic role of the distal site asparagine-histidine couple in catalase-peroxidases Christa Jakopitsch1, Markus Auer1,Gu¨ nther Regelsberger1, Walter Jantschko1, Paul G. Furtmu¨ ller1, Florian Ru¨ ker2 and Christian Obinger1 1Institute of Chemistry and 2Institute of Applied Microbiology, University of Agricultural Sciences, Vienna, Austria Catalase-peroxidases (KatGs) are unique in exhibiting an 6% and that of Asn153fiAsp is 16.5% of wild-type activity. overwhelming catalase activity and a peroxidase activity of Stopped-flow analysis of the reaction of the ferric forms with broad specificity. Similar to other peroxidases the distal H2O2 suggest that exchange of Asn did not shift significantly histidine in KatGs forms a hydrogen bond with an adjacent the ratio of rates of H2O2-mediated compound I formation conserved asparagine. To investigate the catalytic role(s) of and reduction. Both rates seem to be reduced most probably this potential hydrogen bond in the bifunctional activity of because (a) the lower basicity of His123 hampers its function KatGs, Asn153 in Synechocystis KatG was replaced with as acid-base catalyst and (b) Asn153 is part of an extended either Ala (Asn153fiAla) or Asp (Asn153fiAsp). Both KatG-typical H-bond network, the integrity of which seems variants exhibit an overall peroxidase activity similar with to be essential to provide optimal conditions for binding and wild-type KatG. Cyanide binding is monophasic, however, oxidation of the second H2O2 molecule necessary in the the second-order binding rates are reduced to 5.4% catalase reaction.