The Role of Peroxiredoxin 6 in Cell Signaling
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Formaldehyde Induces Bone Marrow Toxicity in Mice by Inhibiting Peroxiredoxin 2 Expression
MOLECULAR MEDICINE REPORTS 10: 1915-1920, 2014 Formaldehyde induces bone marrow toxicity in mice by inhibiting peroxiredoxin 2 expression GUANGYAN YU1, QIANG CHEN1, XIAOMEI LIU1, CAIXIA GUO2, HAIYING DU1 and ZHIWEI SUN1,2 1Department of Preventative Medicine, School of Public Health, Jilin University, Changchun, Jilin 130021; 2 Department of Hygenic Toxicology, School of Public Health, Capital Medical University, Beijing 100069, P.R. China Received November 4, 2013; Accepted June 5, 2014 DOI: 10.3892/mmr.2014.2473 Abstract. Peroxiredoxin 2 (Prx2), a member of the perox- Introduction iredoxin family, regulates numerous cellular processes through intracellular oxidative signal transduction pathways. Formaldehyde (FA) is an environmental agent commonly Formaldehyde (FA)-induced toxic damage involves reactive found in numerous products, including paint, cloth and oxygen species (ROS) that trigger subsequent toxic effects and exhaust gas, as well as other medicinal and industrial products. inflammatory responses. The present study aimed to inves- FA exposure has raised significant concerns due to mounting tigate the role of Prx2 in the development of bone marrow evidence suggesting its carcinogenic potential and severe toxicity caused by FA and the mechanism underlying FA effects on human health (1). Epidemiological and experi- toxicity. According to the results of the preliminary inves- mental data have demonstrated that FA may cause leukemia, tigations, the mice were divided into four groups (n=6 per particularly myeloid leukemia; however, the mechanism group). One group was exposed to ambient air and the other underlying this effect remains unclear (2-4). In the process three groups were exposed to different concentrations of FA of tumor formation, DNA damage may be the initiating (20, 40, 80 mg/m3) for 15 days in the respective inhalation factor (5), while myeloperoxidase (MPO) activity is associ- chambers, for 2 h a day. -

Peroxiredoxins in Neurodegenerative Diseases
antioxidants Review Peroxiredoxins in Neurodegenerative Diseases Monika Szeliga Mossakowski Medical Research Centre, Department of Neurotoxicology, Polish Academy of Sciences, 5 Pawinskiego Street, 02-106 Warsaw, Poland; [email protected]; Tel.: +48-(22)-6086416 Received: 31 October 2020; Accepted: 27 November 2020; Published: 30 November 2020 Abstract: Substantial evidence indicates that oxidative/nitrosative stress contributes to the neurodegenerative diseases. Peroxiredoxins (PRDXs) are one of the enzymatic antioxidant mechanisms neutralizing reactive oxygen/nitrogen species. Since mammalian PRDXs were identified 30 years ago, their significance was long overshadowed by the other well-studied ROS/RNS defense systems. An increasing number of studies suggests that these enzymes may be involved in the neurodegenerative process. This article reviews the current knowledge on the expression and putative roles of PRDXs in neurodegenerative disorders such as Alzheimer’s disease, Parkinson’s disease and dementia with Lewy bodies, multiple sclerosis, amyotrophic lateral sclerosis and Huntington’s disease. Keywords: peroxiredoxin (PRDX); oxidative stress; nitrosative stress; neurodegenerative disease 1. Introduction Under physiological conditions, reactive oxygen species (ROS, e.g., superoxide anion, O2 -; · hydrogen peroxide, H O ; hydroxyl radical, OH; organic hydroperoxide, ROOH) and reactive nitrogen 2 2 · species (RNS, e.g., nitric oxide, NO ; peroxynitrite, ONOO-) are constantly produced as a result of normal · cellular metabolism and play a crucial role in signal transduction, enzyme activation, gene expression, and regulation of immune response [1]. The cells are endowed with several enzymatic (e.g., glutathione peroxidase (GPx); peroxiredoxin (PRDX); thioredoxin (TRX); catalase (CAT); superoxide dismutase (SOD)), and non-enzymatic (e.g., glutathione (GSH); quinones; flavonoids) antioxidant systems that minimize the levels of ROS and RNS. -

The Role of Glutathionperoxidase 4 (GPX4) in Hematopoiesis and Leukemia
The role of glutathionperoxidase 4 (GPX4) in hematopoiesis and leukemia von Kira Célénie Stahnke Inaugral- Dissertation zur Erlangung der Doktorwürde der TierärztliChen Fakultät der Ludwig-Maximilians-Universität MünChen The role of glutathionperoxidase 4 (GPX4) in hematopoiesis and leukemia von Kira Célénie Stahnke aus Dortmund München 2016 Aus dem VeterinärwissensChaftliChen Department der TierärztliChen Fakultät der Ludwig-Maximilians-Universität München Lehrstuhl für Molekulare Tierzucht und Biotechnologie Arbeit angefertigt unter der Leitung von Univ.-Prof. Dr. Eckhard Wolf Angefertigt am Institut für Experimentelle TumorforsChung Universitätsklinikum Ulm Mentor: Prof. Dr. Christian Buske GedruCkt mit Genehmigung der TierärztliChen Fakultät der Ludwig-Maximilians-Universität München Dekan: Univ.-Prof. Dr. JoaChim Braun Berichterstatter: Univ.-Prof. Dr. Eckard Wolf Korreferent/en: Priv.-Doz. Dr. Bianka Schulz Tag der Promotion: 06.02.2016 Table of contents List of Abbreviations..............................................................................2 1 Introduction ........................................................................................6 1.1 ROS and oxidative stress in physiology and pathology..................................................... 6 1.1.1 SourCes of Reactive Oxygen SpeCies..................................................................................................6 1.1.2 PhysiologiCal role of ROS........................................................................................................................8 -

Oxidative Stress Modulates the Expression Pattern of Peroxiredoxin-6 in Peripheral Blood Mononuclear Cells of Asthmatic Patients and Bronchial Epithelial Cells
Allergy Asthma Immunol Res. 2020 May;12(3):523-536 https://doi.org/10.4168/aair.2020.12.3.523 pISSN 2092-7355·eISSN 2092-7363 Original Article Oxidative Stress Modulates the Expression Pattern of Peroxiredoxin-6 in Peripheral Blood Mononuclear Cells of Asthmatic Patients and Bronchial Epithelial Cells Hyun Jae Shim ,1 So-Young Park ,2 Hyouk-Soo Kwon ,1 Woo-Jung Song ,1 Tae-Bum Kim ,1 Keun-Ai Moon ,1 Jun-Pyo Choi ,1 Sin-Jeong Kim ,1 You Sook Cho 1* 1Division of Allergy and Clinical Immunology, Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea 2Division of Pulmonary, Allergy and Critical Care Medicine, Department of Internal Medicine, Konkuk University Medical Center, Seoul, Korea Received: Jul 8, 2019 ABSTRACT Revised: Nov 29, 2019 Accepted: Dec 23, 2019 Purpose: Reduction-oxidation reaction homeostasis is vital for regulating inflammatory Correspondence to conditions and its dysregulation may affect the pathogenesis of chronic airway inflammatory You Sook Cho, MD, PhD diseases such as asthma. Peroxiredoxin-6, an important intracellular anti-oxidant molecule, Division of Allergy and Clinical Immunology, Department of Internal Medicine, Asan is reported to be highly expressed in the airways and lungs. The aim of this study was to Medical Center, University of Ulsan College of analyze the expression pattern of peroxiredoxin-6 in the peripheral blood mononuclear cells Medicine, 88 Olympic-ro 43-gil, Songpa-gu, (PBMCs) of asthmatic patients and in bronchial epithelial cells (BECs). Seoul 05505, Korea. Methods: The expression levels and modifications of peroxiredoxin-6 were evaluated in Tel: +82-2-3010-3285 PBMCs from 22 asthmatic patients. -

1L1 an Outsider S Take on Autism Spectrum Disorders
Plenary Lecture 特別講演 1L1 An outsider’s take on autism spectrum disorders ○Martin Raff MRC LMCB, University College London Autism spectrum disorders(ASDs)are among the commonest neuropsychiatric disorders. Although, until re- cently, ASDs were one of the least understood of these disorders, they are now one of the best understood. Pro- gress has come largely through recent advances in human genetics that have identified rare large!effect muta- tions that cause or greatly increase the risk of these disorders, together with studies of genetic mouse models based on these mutations. Remarkably, in a number of mouse models caused by single mutations, correction of the problem in the adult brain(with either drugs or genetic manipulations)largely reverses many of the behav- ioral and neurobiological abnormalities, providing hope for the development of therapies for individuals with ASDs. In my talk, I will review the basic features of ASDs, using home videos of the development of my 13!year! old autistic grandson as an example, and I will discuss some of the recent advances in ASD research, consider current puzzles, and speculate on possible ways forward. Plenary Lecture 特別講演 2L1 A role of drebrin A in the activity!dependent trafficking of NMDA receptors to the plasma membrane ○Chiye Aoki1,Sho Fujisawa1,Kei Tateyama1,Yi!Wen Chen1,Tomoaki Shirao2 1Center for Neural Science, New York University 2Dept of Neurobiology and Behavior, Gunma Univ Graduate School of Medicine NMDA receptors(NMDARs)are central molecular agents that enable the activity!dependent modification of synaptic strengths at excitatory synapses. However, relatively little is known about the molecular mechanisms regulating NMDAR levels at each synapse. -

Overview on Peroxiredoxin
Mol. Cells 2016; 39(1): 1-5 http://dx.doi.org/10.14348/molcells.2016.2368 Molecules and Cells http://molcells.org Overview Established in 1990 Overview on Peroxiredoxin Sue Goo Rhee* Peroxiredoxins (Prxs) are a very large and highly con- 2-Cys Prxs Tsa1 and Tsa2, two atypical 2-Cys Prxs Ahp1 and served family of peroxidases that reduce peroxides, with a nTpx (where n stands for nucleus), and one 1-Cys mTpx conserved cysteine residue, designated the “peroxidatic” (where m stands for mitochondria) [see the review by by Tole- Cys (CP) serving as the site of oxidation by peroxides (Hall dano and Huang (2016)]. et al., 2011; Rhee et al., 2012). Peroxides oxidize the CP-SH to cysteine sulfenic acid (CP–SOH), which then reacts with CLASSIFICATION another cysteine residue, named the “resolving” Cys (CR) to form a disulfide that is subsequently reduced by an The CP residue is conserved in all Prx enzymes. On the basis appropriate electron donor to complete a catalytic cycle. of the location or absence of the CR, Prxs are classified into 2- This overview summarizes the status of studies on Prxs Cys, atypical 2-Cys, and 1-Cys Prx subfamilies (Chae et al., and relates the following 10 minireviews. 1994b; Rhee et al., 2001; Wood et al., 2003b). 2-Cys Prx en- 1 zymes are homodimeric and contain two conserved (CP and CR) cysteine residues per subunit. The CP–SOH reacts with the NOMENCLATURE CR–SH of the other subunit to form an intersubunit disulfide. In atypical 2-Cys PrxV, the CP–SOH reacts with the CR–SH of the As in all biology, acronyms are overwhelming in Prx literature. -
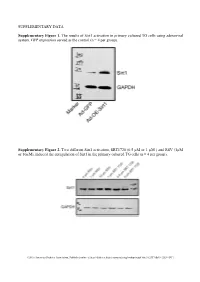
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -

Role of Peroxiredoxin 6 in Human Melanoma
Role of Peroxiredoxin 6 in human melanoma Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen Julius-Maximilians-Universität Würzburg vorgelegt von Alexandra Schmitt aus Würzburg Würzburg 2015 Eingereicht am:__________________________ Mitglieder der Promotionskommission: Vorsitzender:____________________________ Gutachter:______________________________ Gutachter:______________________________ Tag des Promotionskolloquiums:___________________ Doktorurkunde ausgehändigt am:___________________ Eidesstattliche Erklärung Gemäß §4, Abs. 3, Ziff. 3, 5 und 8 der Promotionsordnung der Fakultät für Biologie der Bayerischen Julius-Maximilians-Universität Würzburg Hiermit erkläre ich ehrenwörtlich, dass ich die vorliegende Dissertation selbständig angefertigt und keine anderen als die angegebenen Quellen und Hilfsmittel verwendet habe. Ich erkläre weiterhin, dass die vorliegende Dissertation weder in gleicher, noch in ähnlicher Form bereits in einem anderen Prüfungsverfahren vorgelegen hat. Weiterhin erkläre ich, dass ich außer den mit dem Zulassungsantrag urkundlich vorgelegten Graden keine weiteren akademischen Grade erworben oder zu erwerben versucht habe. Würzburg, Januar 2015 ______________________________________ Alexandra Schmitt Table of contents 1. Abstract ................................................................................................................. 1 2. Zusammenfassung ............................................................................................... 3 3. Introduction .......................................................................................................... -

Kinetic Approaches to Measuring Peroxiredoxin Reactivity
Mol. Cells 2016; 39(1): 26-30 http://dx.doi.org/10.14348/molcells.2016.2325 Molecules and Cells http://molcells.org Established in 1990 Kinetic Approaches to Measuring Peroxiredoxin Reactivity Christine C. Winterbourn*, and Alexander V. Peskin Peroxiredoxins are ubiquitous thiol proteins that catalyse demonstrated surprisingly low reactivity with thiol reagents such the breakdown of peroxides and regulate redox activity in as iodoacetamide and other oxidants such as chloramines the cell. Kinetic analysis of their reactions is required in (Peskin et al., 2007) and it is clear that the low pKa of the active order to identify substrate preferences, to understand how site thiol is insufficient to confer the high peroxide reactivity. In molecular structure affects activity and to establish their fact, typical low molecular weight and protein thiolates react -1 -1 physiological functions. Various approaches can be taken, with H2O2 with a rate constant of 20 M s whereas values of including the measurement of rates of individual steps in Prxs are 105-106 fold higher (Winterbourn and Hampton, 2008). the reaction pathway by stopped flow or competitive kinet- An elegant series of structural and mutational studies (Hall et al., ics, classical enzymatic analysis and measurement of pe- 2010; Nakamura et al., 2010; Nagy et al., 2011) have shown roxidase activity. Each methodology has its strengths and that to get sufficient rate enhancement, it is necessary to acti- they can often give complementary information. However, vate the peroxide. As discussed in detail elsewhere (Hall et al., it is important to understand the experimental conditions 2010; 2011), this involves formation of a transition state in of the assay so as to interpret correctly what parameter is which hydrogen bonding of the peroxide to conserved Arg and being measured. -

GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis Giovanni C
REVIEW GPX4 www.proteomics-journal.com GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis Giovanni C. Forcina and Scott J. Dixon* formation of toxic radicals (e.g., R-O•).[5] Oxygen is necessary for aerobic metabolism but can cause the harmful The eight mammalian GPX proteins fall oxidation of lipids and other macromolecules. Oxidation of cholesterol and into three clades based on amino acid phospholipids containing polyunsaturated fatty acyl chains can lead to lipid sequence similarity: GPX1 and GPX2; peroxidation, membrane damage, and cell death. Lipid hydroperoxides are key GPX3, GPX5, and GPX6; and GPX4, GPX7, and GPX8.[6] GPX1–4 and 6 (in intermediates in the process of lipid peroxidation. The lipid hydroperoxidase humans) are selenoproteins that contain glutathione peroxidase 4 (GPX4) converts lipid hydroperoxides to lipid an essential selenocysteine in the enzyme + alcohols, and this process prevents the iron (Fe2 )-dependent formation of active site, while GPX5, 6 (in mouse and toxic lipid reactive oxygen species (ROS). Inhibition of GPX4 function leads to rats), 7, and 8 use an active site cysteine lipid peroxidation and can result in the induction of ferroptosis, an instead. Unlike other family members, GPX4 (PHGPx) can act as a phospholipid iron-dependent, non-apoptotic form of cell death. This review describes the hydroperoxidase to reduce lipid perox- formation of reactive lipid species, the function of GPX4 in preventing ides to lipid alcohols.[7,8] Thus,GPX4ac- oxidative lipid damage, and the link between GPX4 dysfunction, lipid tivity is essential to maintain lipid home- oxidation, and the induction of ferroptosis. ostasis in the cell, prevent the accumula- tion of toxic lipid ROS and thereby block the onset of an oxidative, iron-dependent, non-apoptotic mode of cell death termed 1. -

K746-200 Myeloperoxidase (MPO) Inhibitor Screening
FOR RESEARCH USE ONLY! Myeloperoxidase (MPO) Inhibitor Screening Kit (Fluorometric) rev. 08/15 (Catalog # K746-200; 200 assays; Store at -20°C) I. Introduction: Myeloperoxidase (MPO: EC 1.11.2.2) is a peroxidase abundantly expressed in neutrophil granulocytes. It catalyzes the hydrogen peroxide dependent oxidation of halide ions to generate hypochlorite (HClO), the reaction by which MPO exhibits cytotoxic activity against tumor cells and microorganisms. MPO also oxidizes various substances such as phenol and anilines. MPO undergoes chlorination or peroxidation reaction depending upon the relative concentrations of chloride and the reducing substrates. Recent studies suggest that increased levels of MPO are associated with an increased risk for cardiovascular disease and myocardial infarction, thus development of novel and specific inhibitors of MPO is critical for therapeutic purposes. BioVision’s MPO Inhibitor Screening Kit provides screening assays for both MPO chlorination and peroxidation activities. In the chlorination assay (specific for MPO), MPO converts hydrogen peroxide and sodium chloride to sodium hypochlorite, which reacts with Chlorination substrate to give an intensely fluorescent product (Ex/Em = 480/520 nm). In the Peroxidation Assay, MPO oxidizes peroxidation substrate to generate fluorescence (Ex/Em = 535/587 nm). The fluorescence generated is directly proportional to any peroxidase activity present. In the presence of MPO inhibitor, reactions are impeded, thus decreasing the rate and/or extent of generation of MPO-dependent fluorescence. This kit provides a sensitive, quick, and easy method for screening potential inhibitors of MPO, and identifying whether one or both activities are inhibited. MPO Inhibitor Control is included to compare the efficacy of test inhibitors. -

The C718T Polymorphism in the 3″-Untranslated
Hypertension Research (2012) 35, 507–512 & 2012 The Japanese Society of Hypertension All rights reserved 0916-9636/12 www.nature.com/hr ORIGINAL ARTICLE The C718T polymorphism in the 3¢-untranslated region of glutathione peroxidase-4 gene is a predictor of cerebral stroke in patients with essential hypertension Alexey V Polonikov1, Ekaterina K Vialykh2, Mikhail I Churnosov3, Thomas Illig4, Maxim B Freidin5, Oksana V Vasil¢eva1, Olga Yu Bushueva1, Valentina N Ryzhaeva1, Irina V Bulgakova1 and Maria A Solodilova1 In the present study we have investigated the association of three single nucleotide polymorphisms in glutathione peroxidase (GPx) genes GPX1 rs1050450 (P198L), GPX3 rs2070593 (G930A) and GPX4 rs713041 (T718C) with the risk of cerebral stroke (CS) in patients with essential hypertension (EH). A total of 667 unrelated EH patients of Russian origin, including 306 hypertensives (the EH–CS group) who suffered from CS and 361 people (the EH–CS group) who did not have cerebrovascular accidents, were enrolled in the study. The variant allele 718C of the GPX4 gene was found to be significantly associated with an increased risk of CS in hypertensive patients (odds ratio (OR) 1.53, 95% confidence interval (CI) 1.23–1.90, Padj¼0.0003). The prevalence of the 718TC and 718CC genotypes of the GPX4 gene was higher in the EH–CS group than the EH-alone group (OR¼2.12, 95%CI 1.42–3.16, Padj¼0.0018). The association of the variant GPX4 genotypes with the increased risk of CS in hypertensives remained statistically significant after adjusting for confounding variables such as sex, body mass index (BMI), blood pressure and antihypertensive medication use (OR¼2.18, 95%CI 1.46–3.27, P¼0.0015).