Dondorp, Background on Malaria and Combination Anti-Malarial Drug Therapy
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Neena Valecha1, Deepali Savargaonkar1, Bina Srivastava1, B
Valecha et al. Malar J (2016) 15:42 DOI 10.1186/s12936-016-1084-1 Malaria Journal RESEARCH Open Access Comparison of the safety and efficacy of fixed‑dose combination of arterolane maleate and piperaquine phosphate with chloroquine in acute, uncomplicated Plasmodium vivax malaria: a phase III, multicentric, open‑label study Neena Valecha1, Deepali Savargaonkar1, Bina Srivastava1, B. H. Krishnamoorthy Rao2, Santanu K. Tripathi3, Nithya Gogtay4, Sanjay Kumar Kochar5, Nalli Babu Vijaya Kumar6, Girish Chandra Rajadhyaksha7, Jitendra D. Lakhani8, Bhagirath B. Solanki9, Rajinder K. Jalali10, Sudershan Arora10, Arjun Roy10, Nilanjan Saha10, Sunil S. Iyer10, Pradeep Sharma10 and Anupkumar R. Anvikar1* Abstract Background: Chloroquine has been the treatment of choice for acute vivax malaria for more than 60 years. Malaria caused by Plasmodium vivax has recently shown resistance to chloroquine in some places. This study compared the efficacy and safety of fixed dose combination (FDC) of arterolane maleate and piperaquine phosphate (PQP) with chloroquine in the treatment of uncomplicated vivax malaria. Methods: Patients aged 13–65 years with confirmed mono-infection of P. vivax along with fever or fever in the previ- ous 48 h were included. The 317 eligible patients were randomly assigned to receive FDC of arterolane maleate and PQP (n 159) or chloroquine (n 158) for 3 days. Primaquine was given as an anti-relapse measure on day 3 and continued= for 14 consecutive days.= Primary efficacy analysis included assessment of the proportion of aparasitaemic and afebrile patients at 72 h. Safety endpoints were analysis of adverse events, vital signs, laboratory data, and abnor- malities on electrocardiograph. Patients participated in the study for at least 42 days. -

Eurartesim, INN-Piperaquine & INN-Artenimol
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Eurartesim 160 mg/20 mg film-coated tablets. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 160 mg piperaquine tetraphosphate (as the tetrahydrate; PQP) and 20 mg artenimol. For the full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM Film-coated tablet (tablet). White oblong biconvex film-coated tablet (dimension 11.5x5.5mm / thickness 4.4mm) with a break-line and marked on one side with the letters “S” and “T”. The tablet can be divided into equal doses. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Eurartesim is indicated for the treatment of uncomplicated Plasmodium falciparum malaria in adults, adolescents, children and infants 6 months and over and weighing 5 kg or more. Consideration should be given to official guidance on the appropriate use of antimalarial medicinal products, including information on the prevalence of resistance to artenimol/piperaquine in the geographical region where the infection was acquired (see section 4.4). 4.2 Posology and method of administration Posology Eurartesim should be administered over three consecutive days for a total of three doses taken at the same time each day. 2 Dosing should be based on body weight as shown in the table below. Body weight Daily dose (mg) Tablet strength and number of tablets per dose (kg) PQP Artenimol 5 to <7 80 10 ½ x 160 mg / 20 mg tablet 7 to <13 160 20 1 x 160 mg / 20 mg tablet 13 to <24 320 40 1 x 320 mg / 40 mg tablet 24 to <36 640 80 2 x 320 mg / 40 mg tablets 36 to <75 960 120 3 x 320 mg / 40 mg tablets > 75* 1,280 160 4 x 320 mg / 40 mg tablets * see section 5.1 If a patient vomits within 30 minutes of taking Eurartesim, the whole dose should be re-administered; if a patient vomits within 30-60 minutes, half the dose should be re-administered. -

Pharmacological and Cardiovascular Perspectives on the Treatment of COVID-19 with Chloroquine Derivatives
www.nature.com/aps REVIEW ARTICLE Pharmacological and cardiovascular perspectives on the treatment of COVID-19 with chloroquine derivatives Xiao-lei Zhang1, Zhuo-ming Li1, Jian-tao Ye1, Jing Lu1, Lingyu Linda Ye2, Chun-xiang Zhang3, Pei-qing Liu1 and Dayue D Duan2 The novel severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) causes coronavirus disease 2019 (COVID-19) and an ongoing severe pandemic. Curative drugs specific for COVID-19 are currently lacking. Chloroquine phosphate and its derivative hydroxychloroquine, which have been used in the treatment and prevention of malaria and autoimmune diseases for decades, were found to inhibit SARS-CoV-2 infection with high potency in vitro and have shown clinical and virologic benefits in COVID-19 patients. Therefore, chloroquine phosphate was first used in the treatment of COVID-19 in China. Later, under a limited emergency- use authorization from the FDA, hydroxychloroquine in combination with azithromycin was used to treat COVID-19 patients in the USA, although the mechanisms of the anti-COVID-19 effects remain unclear. Preliminary outcomes from clinical trials in several countries have generated controversial results. The desperation to control the pandemic overrode the concerns regarding the serious adverse effects of chloroquine derivatives and combination drugs, including lethal arrhythmias and cardiomyopathy. The risks of these treatments have become more complex as a result of findings that COVID-19 is actually a multisystem disease. While respiratory symptoms are the major clinical manifestations, cardiovascular abnormalities, including arrhythmias, myocarditis, heart failure, and ischemic stroke, have been reported in a significant number of COVID-19 patients. Patients with preexisting cardiovascular conditions (hypertension, arrhythmias, etc.) are at increased risk of severe COVID-19 and death. -

Review Article Efforts Made to Eliminate Drug-Resistant Malaria and Its Challenges
Hindawi BioMed Research International Volume 2021, Article ID 5539544, 12 pages https://doi.org/10.1155/2021/5539544 Review Article Efforts Made to Eliminate Drug-Resistant Malaria and Its Challenges Wote Amelo 1,2,3 and Eyasu Makonnen 1,2 1Department of Pharmacology and Clinical Pharmacy, School of Pharmacy, Addis Ababa University, Addis Ababa, Ethiopia 2Center for Innovative Drug Development and Therapeutic Trials for Africa (CDT-Africa), Addis Ababa University, Addis Ababa, Ethiopia 3Department of Pharmacology and Toxicology, School of Pharmacy, Jimma University, Jimma, Ethiopia Correspondence should be addressed to Wote Amelo; [email protected] Received 21 January 2021; Accepted 9 August 2021; Published 30 August 2021 Academic Editor: Jane Hanrahan Copyright © 2021 Wote Amelo and Eyasu Makonnen. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Since 2000, a good deal of progress has been made in malaria control. However, there is still an unacceptably high burden of the disease and numerous challenges limiting advancement towards its elimination and ultimate eradication. Among the challenges is the antimalarial drug resistance, which has been documented for almost all antimalarial drugs in current use. As a result, the malaria research community is working on the modification of existing treatments as well as the discovery and development of new drugs to counter the resistance challenges. To this effect, many products are in the pipeline and expected to be marketed soon. In addition to drug and vaccine development, mass drug administration (MDA) is under scientific scrutiny as an important strategy for effective utilization of the developed products. -

Plasmodium Falciparum Clinical Isolates: in Vitro Genotypic and Phenotypic Characterization Nonlawat Boonyalai1* , Brian A
Boonyalai et al. Malar J (2020) 19:269 https://doi.org/10.1186/s12936-020-03339-w Malaria Journal RESEARCH Open Access Piperaquine resistant Cambodian Plasmodium falciparum clinical isolates: in vitro genotypic and phenotypic characterization Nonlawat Boonyalai1* , Brian A. Vesely1, Chatchadaporn Thamnurak1, Chantida Praditpol1, Watcharintorn Fagnark1, Kirakarn Kirativanich1, Piyaporn Saingam1, Chaiyaporn Chaisatit1, Paphavee Lertsethtakarn1, Panita Gosi1, Worachet Kuntawunginn1, Pattaraporn Vanachayangkul1, Michele D. Spring1, Mark M. Fukuda1, Chanthap Lon1, Philip L. Smith2, Norman C. Waters1, David L. Saunders3 and Mariusz Wojnarski1 Abstract Background: High rates of dihydroartemisinin–piperaquine (DHA–PPQ) treatment failures have been documented for uncomplicated Plasmodium falciparum in Cambodia. The genetic markers plasmepsin 2 (pfpm2), exonuclease (pfexo) and chloroquine resistance transporter (pfcrt) genes are associated with PPQ resistance and are used for moni- toring the prevalence of drug resistance and guiding malaria drug treatment policy. Methods: To examine the relative contribution of each marker to PPQ resistance, in vitro culture and the PPQ survival assay were performed on seventeen P. falciparum isolates from northern Cambodia, and the presence of E415G-Exo and pfcrt mutations (T93S, H97Y, F145I, I218F, M343L, C350R, and G353V) as well as pfpm2 copy number polymor- phisms were determined. Parasites were then cloned by limiting dilution and the cloned parasites were tested for drug susceptibility. Isobolographic analysis of several drug combinations for standard clones and newly cloned P. falciparum Cambodian isolates was also determined. Results: The characterization of culture-adapted isolates revealed that the presence of novel pfcrt mutations (T93S, H97Y, F145I, and I218F) with E415G-Exo mutation can confer PPQ-resistance, in the absence of pfpm2 amplifcation. -

Update Tot 30-04-2020 1. Chloroquine and Hydroxychloroquine for The
Update tot 30-04-2020 1. Chloroquine and Hydroxychloroquine for the Prevention or Treatment of Novel Coronavirus Disease (COVID-19) in Africa: Caution for Inappropriate Off-Label Use in Healthcare Settings. Abena PM, Decloedt EH, Bottieau E, Suleman F, Adejumo P, Sam-Agudu NA, et al. Am j trop med hyg. 2020. 2. Evaluation of Hydroxychloroquine Retinopathy Using Ultra-Widefield Fundus Autofluorescence: Peripheral Findings in the Retinopathy. Ahn SJ, Joung J, Lee BR. American journal of ophthalmology. 2020;209:35-44. http://dx.doi.org/10.1016/j.ajo.2019.09.008. Epub 2019 Sep 14. 3. COVID-19 research has overall low methodological quality thus far: case in point for chloroquine/hydroxychloroquine. Alexander PE, Debono VB, Mammen MJ, Iorio A, Aryal K, Deng D, et al. J clin epidemiol. 2020. 4. Chloroquine and Hydroxychloroquine in the Era of SARS - CoV2: Caution on Their Cardiac Toxicity. Bauman JL, Tisdale JE. Pharmacotherapy. 2020. 5. Repositioned chloroquine and hydroxychloroquine as antiviral prophylaxis for COVID-19: A protocol for rapid systematic review of randomized controlled trials. Chang R, Sun W-Z. medRxiv. 2020:2020.04.18.20071167. 6. Chloroquine and hydroxychloroquine as available weapons to fight COVID-19. Colson P, Rolain J-M, Lagier J-C, Brouqui P, Raoult D. Int J Antimicrob Agents. 2020:105932-. 7. Dose selection of chloroquine phosphate for treatment of COVID-19 based on a physiologically based pharmacokinetic model. Cui C, Zhang M, Yao X, Tu S, Hou Z, Jie En VS, et al. Acta Pharmaceutica Sinica B. 2020. 8. Hydroxychloroquine; Why It Might Be Successful and Why It Might Not Be Successful in the Treatment of Covid-19 Pneumonia? Could It Be A Prophylactic Drug? Deniz O. -
Fixed Dose Combinations Approved by Dcg (I) Since 1961 Till February, 2013
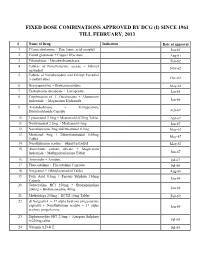
FIXED DOSE COMBINATIONS APPROVED BY DCG (I) SINCE 1961 TILL FEBRUARY, 2013 # Name of Drug Indication Date of approval 1. Cyanocobalamine + Zinc tannic acid complex Jan-61 2. Cobalt glutamate + Copper Glycinate Aug-61 3. Fibrinolysin + Desoxyribonuclease Feb-62 4. Tablets of Norethisterone acetate + Ethinyl Nov-62 oestradiol 5. Tablets of Norethynodrel and Ethinyl Estradiol 3-methyl ether Dec-62 6. Broxyquinoline + Brobenzoxalidine May-63 7. Testosterone decanoate + Isocaproate Jan-64 8. Combination of L Oxethazaine + Aluminium hydroxide + Magnesium Hydroxide Jun-66 9. Amylobarbitone + Trifluperazine Dihydrochloride Capsule Feb-67 10. Lynestronol 2.5mg + Mestranol 0.075mg Tablet Apr-67 11. Northynodrel 2.5mg + Mestranol 0.1mg Jun-67 12. Norethisterone 2mg and Mestranol 0.1mg May-67 13. Mestranol 4mg + Ethinyloestradial 0.05mg May-67 Tablet 14. Norethisterane acetate + ethinyl estradiol May-67 15. Aluminium sodium silicate + Magnesium hydroxide + Methypolysiloxane Tablet Jun-67 16. Ammoidin + Amidine Jul-67 17. Fluocortolene + Flucortolene Caproate Jul-68 18. Norgestrel + Ethinyloestradiol Tablet Aug-68 19. Folic Acid 0.5mg + Ferrous Sulphate 150mg Jan-69 Capsule 20. Tetracycline HCl 250mg + Broxyquinoline 200mg + Brobenzoxadine 40mg Jan-69 21. Methyldopa 250mg + HCTZ 15mg Tablet Feb-69 22. dl Norgestrel + 17 alpha hydroxy progesterone caproate + Norethisterone acetate + 17 alpha Jan-69 acetoxy progesterone 23. Diphenoxylate HCl 2.5mg + Atropine Sulphate 0.025mg tablet Jul-69 24. Vitamin A,D & E Jul-69 25. Lutin 0.1gm + Vit 0.1gm + Vit K1 2.5mg + Dicalcium Phosphate 0.1gm + Carlozochrome Jul-69 Salicylate 1mg tablet 26. Vit K 1 5mg + Calcium Lactolionate 100 m g+ Carlozocrome Salicylate 2.5mg + Phenol 0.5% Jul-69 + Lignocaine Hcl 1% injection 27. -

Downloaded and Saved in PDB Format
bioRxiv preprint doi: https://doi.org/10.1101/833145; this version posted November 6, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 Full title: Efficacy of Lumefantrine against piperaquine resistant Plasmodium berghei parasites is 2 selectively restored by probenecid, verapamil, and cyproheptadine through ferredoxin NADP+- 3 reductase and cysteine desulfurase 4 5 Short title: Mechanisms of Lumefantrine resistance and reversal in Plasmodium berghei ANKA 6 7 Authors: Fagdéba David Bara1,2,3, Loise Ndung’u1, Noah Machuki Onchieku1, Beatrice Irungu2, 8 Simplice Damintoti Karou3, Francis Kimani4, Damaris Matoke-Muhia4, Peter Mwitari2, Gabriel 9 Magoma1,5, Alexis Nzila6, Daniel Kiboi5* 10 11 Affiliations: 1Department of Molecular Biology and Biotechnology, Pan African University 12 Institute for Basic Sciences, Technology and Innovation (PAUSTI), Nairobi, Kenya. 2Centre for 13 Traditional Medicine and Drug Research, Kenya Medical Research Institute, Nairobi, Kenya. 14 3School of Food and Biology Technology, Universite du Lome, Lome, Togo. 4Centre for 15 Biotechnology Research and Development, Kenya Medical Research Institute, Nairobi, Kenya. 16 5Department of Biochemistry, Jomo Kenyatta University of Agriculture and Technology 17 (JKUAT), Nairobi, Kenya. 6Department of Life Sciences, King Fahd University of Petroleum and 18 Minerals, Dharam, Saudi Arabia. 19 20 Corresponding author: [email protected] ; [email protected] 1 bioRxiv preprint doi: https://doi.org/10.1101/833145; this version posted November 6, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -

Treatment Failure Due to the Potential Under-Dosing of Dihydroartemisinin-Piperaquine in a Patient with Plasmodium Falciparum Uncomplicated Malaria
INFECT DIS TROP MED 2019; 5: E525 Treatment failure due to the potential under-dosing of dihydroartemisinin-piperaquine in a patient with Plasmodium falciparum uncomplicated malaria I. De Benedetto1, F. Gobbi2, S. Audagnotto1, C. Piubelli2, E. Razzaboni3, R. Bertucci1, G. Di Perri1, A. Calcagno1 1Department of Medical Sciences, Unit of Infectious Diseases, University of Torino, Amedeo di Savoia Hospital, Torino, Italy 2Department of Infectious–Tropical Diseases and Microbiology, IRCCS Sacro Cuore Don Calabria Hospital, Verona, Italy 3Unit of Infectious Diseases, Azienda Ospedaliera Universitaria Integrata di Verona, Verona, Italy ABSTRACT: — Background: Dihydroartemisinin/piperaquine (DHA-PPQ) 40/320 mg is approved for the treatment of Plasmodium falciparum uncomplicated malaria. Different recommendations are provided by WHO guidelines and drug data sheet about dosing in overweight patients. We report here a treatment failure likely caused by sub-optimal dosing of dihydroartemisinin-piperaquine in a case of uncomplicated P. fal- ciparum malaria in a returning traveler from Burkina Faso. INTRODUCTION kg). They, therefore, provided an updated dosing body weight dosing schedule in their 2015 guidelines for Dihydroartemisinin/piperaquine (DHA-PPQ) 40/320 malaria treatment that provides for a dose of 200/1600 mg tablet formulation is approved for the treatment mg (5 tablets) in individuals > 80 kg1. of Plasmodium falciparum uncomplicated malaria in adults and children > 6 months and > 5 kg of body weight. Following WHO guidelines, the daily -
![Ehealth DSI [Ehdsi V2.2.2-OR] Ehealth DSI – Master Value Set](https://docslib.b-cdn.net/cover/8870/ehealth-dsi-ehdsi-v2-2-2-or-ehealth-dsi-master-value-set-1028870.webp)
Ehealth DSI [Ehdsi V2.2.2-OR] Ehealth DSI – Master Value Set
MTC eHealth DSI [eHDSI v2.2.2-OR] eHealth DSI – Master Value Set Catalogue Responsible : eHDSI Solution Provider PublishDate : Wed Nov 08 16:16:10 CET 2017 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 1 of 490 MTC Table of Contents epSOSActiveIngredient 4 epSOSAdministrativeGender 148 epSOSAdverseEventType 149 epSOSAllergenNoDrugs 150 epSOSBloodGroup 155 epSOSBloodPressure 156 epSOSCodeNoMedication 157 epSOSCodeProb 158 epSOSConfidentiality 159 epSOSCountry 160 epSOSDisplayLabel 167 epSOSDocumentCode 170 epSOSDoseForm 171 epSOSHealthcareProfessionalRoles 184 epSOSIllnessesandDisorders 186 epSOSLanguage 448 epSOSMedicalDevices 458 epSOSNullFavor 461 epSOSPackage 462 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 2 of 490 MTC epSOSPersonalRelationship 464 epSOSPregnancyInformation 466 epSOSProcedures 467 epSOSReactionAllergy 470 epSOSResolutionOutcome 472 epSOSRoleClass 473 epSOSRouteofAdministration 474 epSOSSections 477 epSOSSeverity 478 epSOSSocialHistory 479 epSOSStatusCode 480 epSOSSubstitutionCode 481 epSOSTelecomAddress 482 epSOSTimingEvent 483 epSOSUnits 484 epSOSUnknownInformation 487 epSOSVaccine 488 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 3 of 490 MTC epSOSActiveIngredient epSOSActiveIngredient Value Set ID 1.3.6.1.4.1.12559.11.10.1.3.1.42.24 TRANSLATIONS Code System ID Code System Version Concept Code Description (FSN) 2.16.840.1.113883.6.73 2017-01 A ALIMENTARY TRACT AND METABOLISM 2.16.840.1.113883.6.73 2017-01 -

Medical Review(S) Review of Request for Priority Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 22-268 MEDICAL REVIEW(S) REVIEW OF REQUEST FOR PRIORITY REVIEW To: Edward Cox, MD, MPH Director, Office of Antimicrobial Products Through: Renata Albrecht, M.D Director, DSPTP, OAP From: Joette M. Meyer, Pharm.D. Acting Medical Team Leader, DSPTP, OAP NDA: 22-268 Submission Date: 6/27/08 Date Review Completed 7/25/08 Product: Coartem (artemether/lumefantrine) Sponsor: Novartis Pharmaceuticals Corporation East Hanover, NJ Proposed Indication: Treatment of malaria in patients of 5kg body weight and above with acute, uncomplicated infections due to Plasmodium falciparum or mixed infections including P. falciparum Proposed Dosing Regimen: A standard 3-day treatment schedule with a total of 6 doses is recommended and dosed based on bodyweight: 5 kg to < 15 kg: One tablet as an initial dose, 1 tablet again after 8 hours and then 1 tablet twice daily (morning and evening) for the following two days 15 kg to < 25 kg bodyweight: Two tablets as an initial dose, 2 tablets again after 8 hours and then 2 tablets twice daily (morning and evening) for the following two days 25 kg to < 35 kg bodyweight: Three tablets as an initial dose, 3 tablets again after 8 hours and then 3 tablets twice daily (morning and evening) for the following two days 35 kg bodyweight and above: Four tablets as a single initial dose, 4 tablets again after 8 hours and then 4 tablets twice daily (morning and evening) for the following two days Abbreviations A artemether ACT artemisinin-based combination therapy AL artemether-lumefantrine -

Current Antimalarial Therapies and Advances in the Development of Semi-Synthetic Artemisinin Derivatives
Anais da Academia Brasileira de Ciências (2018) 90(1 Suppl. 2): 1251-1271 (Annals of the Brazilian Academy of Sciences) Printed version ISSN 0001-3765 / Online version ISSN 1678-2690 http://dx.doi.org/10.1590/0001-3765201820170830 www.scielo.br/aabc | www.fb.com/aabcjournal Current Antimalarial Therapies and Advances in the Development of Semi-Synthetic Artemisinin Derivatives LUIZ C.S. PINHEIRO1, LÍVIA M. FEITOSA1,2, FLÁVIA F. DA SILVEIRA1,2 and NUBIA BOECHAT1 1Fundação Oswaldo Cruz, Instituto de Tecnologia em Fármacos Farmanguinhos, Fiocruz, Departamento de Síntese de Fármacos, Rua Sizenando Nabuco, 100, Manguinhos, 21041-250 Rio de Janeiro, RJ, Brazil 2Universidade Federal do Rio de Janeiro, Programa de Pós-Graduação em Química, Avenida Athos da Silveira Ramos, 149, Cidade Universitária, 21941-909 Rio de Janeiro, RJ, Brazil Manuscript received on October 17, 2017; accepted for publication on December 18, 2017 ABSTRACT According to the World Health Organization, malaria remains one of the biggest public health problems in the world. The development of resistance is a current concern, mainly because the number of safe drugs for this disease is limited. Artemisinin-based combination therapy is recommended by the World Health Organization to prevent or delay the onset of resistance. Thus, the need to obtain new drugs makes artemisinin the most widely used scaffold to obtain synthetic compounds. This review describes the drugs based on artemisinin and its derivatives, including hybrid derivatives and dimers, trimers and tetramers that contain an endoperoxide bridge. This class of compounds is of extreme importance for the discovery of new drugs to treat malaria. Key words: malaria, Plasmodium falciparum, artemisinin, hybrid.