Binaural Interaction in Superior Olivary Nucleus
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Neural Sensitivity to Interaural Time Differences: Beyond the Jeffress Model
The Journal of Neuroscience, February 15, 2000, 20(4):1605–1615 Neural Sensitivity to Interaural Time Differences: Beyond the Jeffress Model Douglas C. Fitzpatrick, Shigeyuki Kuwada, and Ranjan Batra Department of Anatomy, University of Connecticut Health Center, Farmington, Connecticut 06030-3405 Interaural time differences (ITDs) are a major cue for localizing with large differences in path lengths between the two ears. Our the azimuthal position of sounds. The dominant models for results suggest instead that sensitivity to large ITDs is created processing ITDs are based on the Jeffress model and predict with short delay lines, using neurons that display intermediate- neurons that fire maximally at a common ITD across their and trough-type responses. We demonstrate that a neuron’s responsive frequency range. Such neurons are indeed found in best ITD can be predicted from (1) its characteristic delay, a the binaural pathways and are referred to as “peak-type.” rough measure of the delay line, (2) its characteristic phase, However, other neurons discharge minimally at a common ITD which defines the response type, and (3) its best frequency for (trough-type), and others do not display a common ITD at the ITD sensitivity. The intermediate- and trough-type neurons that maxima or minima (intermediate-type). From recordings of neu- have large best ITDs are predicted to be most active when rons in the auditory cortex of the unanesthetized rabbit to sounds at the two ears are decorrelated and may transmit low-frequency tones and envelopes of high-frequency sounds, information about auditory space other than sound localization. we show that the different response types combine to form a continuous axis of best ITD. -

Auditory Processing Disorders Assessment and Intervention: Best Practices Task Force on Auditory Processing Disorders, Minnesota Speech-Language-Hearing Assoc
Auditory Processing Disorders Assessment and Intervention: Best Practices Task Force on Auditory Processing Disorders, Minnesota Speech-Language-Hearing Assoc. Introduction The Minnesota Speech-Language-Hearing Association developed the Task Force on Auditory Processing Disorders to survey practices pertaining to auditory processing assessment and intervention among audiologists and speech-language pathologists in Minnesota. This document summarizes the auditory processing protocols and practices endorsed by the following members of the task force: Vicki Anderson, AuD, CCC-A, FAAA, HealthPartners Sarah Angerman, PhD, CCC-A, University of Minnesota Cassandra Billiet, AuD, CCC-A, FAAA, Oakdale Ear, Nose, & Throat Clinic Sarah Hanson, MA, CCC-SLP, Associated Speech & Language Specialists, LLC Linda Kalweit, AuD, CCC-A, FAAA, Duluth Public Schools ISD #709 Anne Lindgren, MA, CCC-SLP, Anoka-Hennepin School District #11 Amy Sievert O’Keefe, AuD, CCC-A, FAAA, HealthPartners Statement of Purpose The purpose of this document is to supplement the consensus statements provided by the national professional associations for audiology and speech-language pathology, namely the American Academy of Audiology (AAA) and the American Speech-Language-Hearing Association (ASHA): American Academy of Audiology. (2010). Diagnosis, treatment, and management of children and adults with central auditory processing disorder [Clinical Practice Guidelines]. Available from http://audiology.org/resources/documentlibrary/Pages/default.aspx American Speech-Language-Hearing Association. (2005). (Central) auditory processing disorders [Technical Report]. Available from www.asha.org/policy. American Speech-Language-Hearing Association. (2005). (Central) auditory processing disorders—the role of the audiologist [Position Statement]. Available from www.asha.org/policy. This document is also meant to serve in conjunction with, and as an update to, the document cited below: Minnesota Department of Education. -

Binaural Interaction Component in Speech Evoked Auditory Brainstem Responses
J Int Adv Otol 2015; 11(2): 114-7 • DOI: 10.5152/iao.2015.426 Original Article Binaural Interaction Component in Speech Evoked Auditory Brainstem Responses Ajith Kumar Uppunda, Jayashree Bhat, Pearl Edna D'costa, Muthu Raj, Kaushlendra Kumar Department of Audiology, All India Institute of Speech and Hearing, Mysore, Karnataka, India (AKU) Kasturba Medical College, Mangalore, Manipal University, Manipal, Audiology and Speech Language Pathology, Karnataka, India (JB, PED, MR, KK) OBJECTIVE: This article aims to describe the characteristics of the binaural interaction component (BIC) of speech-evoked auditory brainstem response (ABR). MATERIALS and METHODS: All 15 subjects had normal peripheral hearing sensitivity. ABRs were elicited by speech stimulus /da/. RESULTS: The first BIC (BIC-SP1) in the speech-evoked ABR occurred at around 6 ms in the region of peak V. The second BIC (BIC-SP2) was present around 8 ms in the latency region of peak A. The third and fourth BICs of speech-evoked ABR (BIC-SP3 & BIC-SP4) were observed at around 36 ms and 46 ms, respectively, in the latency regions of peaks E and F, respectively. BIC-SP1 and BIC-SP2 were present in all subjects tested (100%), whereas BIC-SP3 and BIC-SP4 were present in 11 (73%). CONCLUSION: Because ABRs are not affected by sleep and mature early, this tool can be evaluated in identifying binaural interaction in younger and difficult-to-test populations. KEYWORDS: Binaural interaction component, auditory brainstem response, speech, latency, amplitude INTRODUCTION The binaural interaction component (BIC) of auditory brainstem responses (ABR) has been a subject of investigation since the 1970s. -

Auditory and Vestibular Systems Objective • to Learn the Functional
Auditory and Vestibular Systems Objective • To learn the functional organization of the auditory and vestibular systems • To understand how one can use changes in auditory function following injury to localize the site of a lesion • To begin to learn the vestibular pathways, as a prelude to studying motor pathways controlling balance in a later lab. Ch 7 Key Figs: 7-1; 7-2; 7-4; 7-5 Clinical Case #2 Hearing loss and dizziness; CC4-1 Self evaluation • Be able to identify all structures listed in key terms and describe briefly their principal functions • Use neuroanatomy on the web to test your understanding ************************************************************************************** List of media F-5 Vestibular efferent connections The first order neurons of the vestibular system are bipolar cells whose cell bodies are located in the vestibular ganglion in the internal ear (NTA Fig. 7-3). The distal processes of these cells contact the receptor hair cells located within the ampulae of the semicircular canals and the utricle and saccule. The central processes of the bipolar cells constitute the vestibular portion of the vestibulocochlear (VIIIth cranial) nerve. Most of these primary vestibular afferents enter the ipsilateral brain stem inferior to the inferior cerebellar peduncle to terminate in the vestibular nuclear complex, which is located in the medulla and caudal pons. The vestibular nuclear complex (NTA Figs, 7-2, 7-3), which lies in the floor of the fourth ventricle, contains four nuclei: 1) the superior vestibular nucleus; 2) the inferior vestibular nucleus; 3) the lateral vestibular nucleus; and 4) the medial vestibular nucleus. Vestibular nuclei give rise to secondary fibers that project to the cerebellum, certain motor cranial nerve nuclei, the reticular formation, all spinal levels, and the thalamus. -

Anatomy of the Superior Olivary Complex.Pdf
Douglas Oliver University of Connecticut Health Center SUPERIOR OLIVE Auditory Pathways Auditory CORTEX GLUT Cortex GABA GLY Medial Geniculate MGB Body Inferior IC Colliculus DLL DLL COCHLEA VLL VLL DCN VCN SOC Auditory Pathways IC Organization of Superior Olivary Complex . Subdivisions and Cytoarchitecture . Neuron types . Inputs . Outputs . Synapses . Basic Circuit Cytoarchitecture of Superior Olivary Complex LSO LSO MSO MSO MNTB D MNTB M (somata & dendrites) (axons & endings) Tsuchitani, 1978, Fig. 10 Comparative anatomy of SOC Tetsufumi Ito & Shig Kuwada Binaural Basic Circuits 8 ‐ 9 Brodal Fig MSO: medial superior olive; LSO: lateral superior olive NTB: nucleus of trapezoid body; IC: inferior colliculus MSO Principle glutamate Cells . Fusiform . Bipolar . Disc‐shaped . Each dendrite innervated by a different side MSO‐In situ hybridization RPO MSO MNTB SPO LSO VGLUT1 VGLUT2 VIAAT NISSL MSO Inputs and Synapses H=high frequency EI - ILD L=low frequency EE - ITD LSO MSO L L B H B B H G LNTB TO LSO MNTB E=Excitation (glutamate) ‐‐‐ I=Inhibition (glycine) ITD CODING Unlike retinal targets, the cochlear nuclei contain maps of frequency, not location. So how does the auditory system know ‘where’ a sound is coming from? T + ITD T By comparing the interaural time differences (ITD) between the ears How is this accomplished?... LSO MSO Right Input A Right Input B C Time Code Time Code E E A A B B C C D D E E Output Output abcde Place Code abcde Place Code Excitation MSO creates a response to Left Input Left Input Inhibition interaural time differences I Time Code E Time Code DEMSO "peak" unit LSO "trough" unit ITD ITD Figure 14.2 Binaural Responses in MSO MSO Summary . -

The Superior Olivary Complex +
Excitatory and inhibitory transmission in the superior olivary complex. Ian D. Forsythe, Matt Barker, Margaret Barnes-Davies, Brian Billups, Paul Dodson, Fatima Osmani, Steven Owens and Adrian Wong. Department of Cell Physiology and Pharmacology, University of Leicester, Leicester LE1 9HN. UK. The timing and pattern of action potentials propagating into the brainstem from both cochleae contain information about the azimuth location of that sound in auditory space. This binaural information is integrated in the superior olivary complex. This part of the auditory pathway is adapted for fast conduction speeds and the preservation of timing information with several complimentary mechanisms (see Oertel, 1999; Trussell, 1999). There are large diameter axons terminating in giant somatic synapses that activate receptor ion channels with fast kinetics. The resultant postsynaptic potentials generated in the receiving neuron are integrated with a suite of voltage-gated ion channels that determine the action potential threshold, duration and repetitive firing properties. We have studied presynaptic and postsynaptic mechanisms that regulate efficacy, timing and integration of synaptic responses in the medial nucleus of the trapezoid body and the medial and lateral superior olives. Presynaptic calcium currents in the calyx of Held. The calyx of Held is a giant synaptic terminal that forms around the soma of principal cells in the Medial Nucleus of the Trapezoid Body (MNTB) (Forsythe, 1994). Each MNTB neuron receives a single calyx. Action potentials propagating into the synaptic terminal trigger the opening of P-type calcium channels (Forsythe et al. 1998) which in turn trigger the release of glutamate into the synaptic cleft (Borst et al., 1995). -
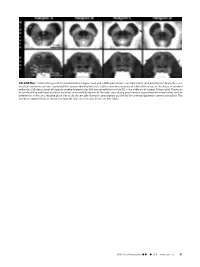
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

Accuracy of Recorded Measures of the Willeford Central Auditory Processing Test Battery
University of Montana ScholarWorks at University of Montana Graduate Student Theses, Dissertations, & Professional Papers Graduate School 1981 Accuracy of recorded measures of the Willeford Central Auditory Processing test battery Susan L. Shea The University of Montana Follow this and additional works at: https://scholarworks.umt.edu/etd Let us know how access to this document benefits ou.y Recommended Citation Shea, Susan L., "Accuracy of recorded measures of the Willeford Central Auditory Processing test battery" (1981). Graduate Student Theses, Dissertations, & Professional Papers. 1471. https://scholarworks.umt.edu/etd/1471 This Thesis is brought to you for free and open access by the Graduate School at ScholarWorks at University of Montana. It has been accepted for inclusion in Graduate Student Theses, Dissertations, & Professional Papers by an authorized administrator of ScholarWorks at University of Montana. For more information, please contact [email protected]. COPYRIGHT ACT OF 1976 THIS IS AN UNPUELISHED MANUSCRIPT IN WHICH COPYRIGHT SUB SISTS. ANY FURTHER REPRINTING OF ITS CONTENTS MUST BE APPROVED BY THE AUTHOR. MANSFIELD LIBRARY UNIVERSITY QI; MONTANA DATE I 1^ ^ \ „ ACCURACY OF RECORDED MEASURES OF THE WILLEFORD CENTRAL AUDITORY PROCESSING TEST BATTERY by Susan L. Shea B.Sc., Speech Pathology & Audiology University of Alberta, 1979 A thesis submitted in partial fulfillment of the requirements for the degree of Master of Arts in the Department of Communication Sciences and Disorders in the Graduate School of the University of Montana June, 1981 Approved by: Chairman, oi Examiners Dean, Graduate School SLJL-JJL Date UMI Number: EP35122 All rights reserved INFORMATION TO ALL USERS The quality of this reproduction is dependent upon the quality of the copy submitted. -

The Challenges of Restoring Binaural Hearing: a Study of Binaural Fusion, Listening Effort, and Music Perception in Children Using Bilateral Cochlear Implants
The challenges of restoring binaural hearing: A study of binaural fusion, listening effort, and music perception in children using bilateral cochlear implants Written by Morrison Mansour Steel A thesis submitted in conformity with the requirements for the degree of Master of Science Institute of Medical Science University of Toronto © Copyright by Morrison M Steel, 2014 The challenges of restoring binaural hearing: A study of binaural fusion, listening effort, and music perception in children using bilateral cochlear implants Morrison M Steel Master of Science Institute of Medical Science The University of Toronto 2014 Benefits from bilateral implantation vary and are limited by mismatched devices, stimulation schemes, and abnormal development. We asked whether bilateral implant use promotes binaural fusion, reduces listening effort, or enhances music perception in children. Binaural fusion (perception of 1 vs. 2 sounds) was assessed behaviourally and reaction times and pupillary changes were recorded simultaneously to measure effort. The child’s Montreal Battery of Evaluation of Amusia was modified and administered to evaluate music perception. Bilaterally implanted children heard a fused percept less frequently than normal hearing peers, possibly due to impaired cortical integration and device limitations, but fusion improved in children who were older and had less brainstem asymmetry and when there was a level difference. Poorer fusion was associated with increased effort, which may have resulted from diminished bottom-up processing. Children with bilateral implants perceived music as accurately as unilaterally implanted peers and improved performance with greater bilateral implant use. ii Acknowledgments I would not have been able to persevere through this process without continual support and encouragement from many important people in my life. -

Evolution of Mammalian Sound Localization Circuits: a Developmental Perspective
Progress in Neurobiology 141 (2016) 1–24 Contents lists available at ScienceDirect Progress in Neurobiology journal homepage: www.elsevier.com/locate/pneurobio Review article Evolution of mammalian sound localization circuits: A developmental perspective a,b, Hans Gerd Nothwang * a Neurogenetics group, Center of Excellence Hearing4All, School of Medicine and Health Sciences, Carl von Ossietzky University Oldenburg, 26111 Oldenburg, Germany b Research Center for Neurosensory Science, Carl von Ossietzky University Oldenburg, 26111 Oldenburg, Germany A R T I C L E I N F O A B S T R A C T Article history: Localization of sound sources is a central aspect of auditory processing. A unique feature of mammals is Received 29 May 2015 the smooth, tonotopically organized extension of the hearing range to high frequencies (HF) above Received in revised form 27 February 2016 10 kHz, which likely induced positive selection for novel mechanisms of sound localization. How this Accepted 27 February 2016 change in the auditory periphery is accompanied by changes in the central auditory system is unresolved. Available online 28 March 2016 + I will argue that the major VGlut2 excitatory projection neurons of sound localization circuits (dorsal cochlear nucleus (DCN), lateral and medial superior olive (LSO and MSO)) represent serial homologs with Keywords: modifications, thus being paramorphs. This assumption is based on common embryonic origin from an Brainstem + + Atoh1 /Wnt1 cell lineage in the rhombic lip of r5, same cell birth, a fusiform cell morphology, shared Evolution Homology genetic components such as Lhx2 and Lhx9 transcription factors, and similar projection patterns. Such a Innovation parsimonious evolutionary mechanism likely accelerated the emergence of neurons for sound Rhombomere localization in all three dimensions. -

Binaural Sound Localization
CHAPTER 5 BINAURAL SOUND LOCALIZATION 5.1 INTRODUCTION We listen to speech (as well as to other sounds) with two ears, and it is quite remarkable how well we can separate and selectively attend to individual sound sources in a cluttered acoustical environment. In fact, the familiar term ‘cocktail party processing’ was coined in an early study of how the binaural system enables us to selectively attend to individual con- versations when many are present, as in, of course, a cocktail party [23]. This phenomenon illustrates the important contributionthat binaural hearing makes to auditory scene analysis, byenablingusto localizeandseparatesoundsources. Inaddition, the binaural system plays a major role in improving speech intelligibility in noisy and reverberant environments. The primary goal of this chapter is to provide an understanding of the basic mechanisms underlying binaural localization of sound, along with an appreciation of how binaural pro- cessing by the auditory system enhances the intelligibility of speech in noisy acoustical environments, and in the presence of competing talkers. Like so many aspects of sensory processing, the binaural system offers an existence proof of the possibility of extraordinary performance in sound localization, and signal separation, but it does not yet providea very complete picture of how this level of performance can be achieved with the contemporary tools of computational auditory scene analysis (CASA). We first summarize in Sec. 5.2 the major physical factors that underly binaural percep- tion, and we briefly summarize some of the classical physiological findings that describe mechanisms that could support some aspects of binaural processing. In Sec. 5.3 we briefly review some of the basic psychoacoustical results which have motivated the development Computational Auditory Scene Analysis. -

Binaural Cross-Correlation Predicts the Responses of Neurons in the Owl’S Auditory Space Map Under Conditions Simulating Summing Localization
The Journal of Neuroscience, July 1, 1996, 16(13):4300–4309 Binaural Cross-Correlation Predicts the Responses of Neurons in the Owl’s Auditory Space Map under Conditions Simulating Summing Localization Clifford H. Keller and Terry T. Takahashi Institute of Neuroscience, University of Oregon, Eugene, Oregon 97403-1254 Summing localization describes the perceptions of human lis- stimulus delays. The resulting binaural cross-correlation sur- teners to two identical sounds from different locations pre- face strongly resembles the pattern of activity across the space sented with delays of 0–1 msec. Usually a single source is map inferred from recordings of single space-specific neurons. perceived to be located between the two actual source loca- Four peaks are observed in the cross-correlation surface for tions, biased toward the earlier source. We studied neuronal any nonzero delay. One peak occurs at the correlation delay responses within the space map of the barn owl to sounds equal to the ITD of each speaker. Two additional peaks reflect presented with this same paradigm. The owl’s primary cue for “phantom sources” occurring at correlation delays that match localization along the azimuth, interaural time difference (ITD), is the signal of the left speaker in one ear with the signal of the based on a cross-correlation-like treatment of the signals ar- right speaker in the other ear. At zero delay, the two phantom riving at each ear. The output of this cross-correlation is dis- peaks coincide. The surface features are complicated further played as neural activity across the auditory space map in the by the interactions of the various correlation peaks.