Mechanistic Studies of Ethylene and Α-Olefin Co-Oligomerization
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Part I: Carbonyl-Olefin Metathesis of Norbornene
Part I: Carbonyl-Olefin Metathesis of Norbornene Part II: Cyclopropenimine-Catalyzed Asymmetric Michael Reactions Zara Maxine Seibel Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate School of Arts and Sciences COLUMBIA UNIVERSITY 2016 1 © 2016 Zara Maxine Seibel All Rights Reserved 2 ABSTRACT Part I: Carbonyl-Olefin Metathesis of Norbornene Part II: Cyclopropenimine-Catalyzed Asymmetric Michael Reactions Zara Maxine Seibel This thesis details progress towards the development of an organocatalytic carbonyl- olefin metathesis of norbornene. This transformation has not previously been done catalytically and has not been done in practical manner with stepwise or stoichiometric processes. Building on the previous work of the Lambert lab on the metathesis of cyclopropene and an aldehyde using a hydrazine catalyst, this work discusses efforts to expand to the less stained norbornene. Computational and experimental studies on the catalytic cycle are discussed, including detailed experimental work on how various factors affect the difficult cycloreversion step. The second portion of this thesis details the use of chiral cyclopropenimine bases as catalysts for asymmetric Michael reactions. The Lambert lab has previously developed chiral cyclopropenimine bases for glycine imine nucleophiles. The scope of these catalysts was expanded to include glycine imine derivatives in which the nitrogen atom was replaced with a carbon atom, and to include imines derived from other amino acids. i Table of Contents List of Abbreviations…………………………………………………………………………..iv Part I: Carbonyl-Olefin Metathesis…………………………………………………………… 1 Chapter 1 – Metathesis Reactions of Double Bonds………………………………………….. 1 Introduction………………………………………………………………………………. 1 Olefin Metathesis………………………………………………………………………… 2 Wittig Reaction…………………………………………………………………………... 6 Tebbe Olefination………………………………………………………………………... 9 Carbonyl-Olefin Metathesis……………………………………………………………. -

Catalytic Pyrolysis of Plastic Wastes for the Production of Liquid Fuels for Engines
Electronic Supplementary Material (ESI) for RSC Advances. This journal is © The Royal Society of Chemistry 2019 Supporting information for: Catalytic pyrolysis of plastic wastes for the production of liquid fuels for engines Supattra Budsaereechaia, Andrew J. Huntb and Yuvarat Ngernyen*a aDepartment of Chemical Engineering, Faculty of Engineering, Khon Kaen University, Khon Kaen, 40002, Thailand. E-mail:[email protected] bMaterials Chemistry Research Center, Department of Chemistry and Center of Excellence for Innovation in Chemistry, Faculty of Science, Khon Kaen University, Khon Kaen, 40002, Thailand Fig. S1 The process for pelletization of catalyst PS PS+bentonite PP ) t e PP+bentonite s f f o % ( LDPE e c n a t t LDPE+bentonite s i m s n HDPE a r T HDPE+bentonite Gasohol 91 Diesel 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) Fig. S2 FTIR spectra of oil from pyrolysis of plastic waste type. Table S1 Compounds in oils (%Area) from the pyrolysis of plastic wastes as detected by GCMS analysis PS PP LDPE HDPE Gasohol 91 Diesel Compound NC C Compound NC C Compound NC C Compound NC C 1- 0 0.15 Pentane 1.13 1.29 n-Hexane 0.71 0.73 n-Hexane 0.65 0.64 Butane, 2- Octane : 0.32 Tetradecene methyl- : 2.60 Toluene 7.93 7.56 Cyclohexane 2.28 2.51 1-Hexene 1.05 1.10 1-Hexene 1.15 1.16 Pentane : 1.95 Nonane : 0.83 Ethylbenzen 15.07 11.29 Heptane, 4- 1.81 1.68 Heptane 1.26 1.35 Heptane 1.22 1.23 Butane, 2,2- Decane : 1.34 e methyl- dimethyl- : 0.47 1-Tridecene 0 0.14 2,2-Dimethyl- 0.63 0 1-Heptene 1.37 1.46 1-Heptene 1.32 1.35 Pentane, -

Title Crystallization of Stereospecific Olefin Copolymers (Special Issue on Physical Chemistry) Author(S) Sakaguchi, Fumio; Kita
Crystallization of Stereospecific Olefin Copolymers (Special Title Issue on Physical Chemistry) Author(s) Sakaguchi, Fumio; Kitamaru, Ryozo; Tsuji, Waichiro Bulletin of the Institute for Chemical Research, Kyoto Citation University (1966), 44(4): 295-315 Issue Date 1966-10-31 URL http://hdl.handle.net/2433/76134 Right Type Departmental Bulletin Paper Textversion publisher Kyoto University Crystallization of Stereospecifie Olefin Copolymers Fumio SAKAGUCHI,Ryozo KITAMARU and Waichiro TSUJI* (Tsuji Laboratory) Received August 13, 1966 The stereoregularity of isotactic poly(4-methyl-1-pentene) was characterized and isomorphism phenomena were examined for the copolymeric systems of 4-methyl-1-pentene with several olefins in order to study the crystallization phenomena in these olefin copoly- mers polymerized with stereospecific catalysts. The structural heterogeneity or the fine crystalline structure of poly(4-methyl-1-pentene) could be correlated with its molecular structure by viewing this stereoregular homopolymer as if it were a copolymer. Cocrystallization or isomorphism phenomenon was recognized for the copolymeric systems of 4-methyl-1-pentene with butene-1, pentene-1, decene-1 and 3-methyl-1-butene, while no evidence of the phenomenon was obtained for the copolymeric systems with styrene and propylene. The degree of the isomorphism of those copolymers was discussed with the informations on the crystalline phases obtained from the X-ray study, on the constitution of the copolymeric chains in the amorphous phases obtained from the viscoelastic studies and on the other thermodynamical properties of these systems. INTRODUCTION Many works have been made with regard to the homopolymerization of olefins with stereospecific catalysts, i. e. complex catalysts composed of the combination of organometallic compound and transitional metallic compound. -

Highly Efficient Olefin Isomerization Catalyzed by Metal Hydrides Derives from Dirhodium(Ii) Carboxylates and Catecholborane
HIGHLY EFFICIENT OLEFIN ISOMERIZATION CATALYZED BY METAL HYDRIDES DERIVES FROM DIRHODIUM(II) CARBOXYLATES AND CATECHOLBORANE Gene A. Devora and Michael P. DoyleL' * Department of Chemistry, Trinity University, San Antonio, Texas 78212, USA Abstract. Dirhodium(ll) tetraacetate in combination with catecholborane catalyzes the iso- merization of alkenes and dienes. Effective isomerization occurs at 80-135°C with the use of only 0.1 mol % rhodium acetate. With 2-methyl-1,5-hexadiene the disubstituted double bond is prefer- entially isomerized. In addition, hydrogen transfer hydrogenation occurs with 1,4-cyclohexadienes. The mechanism of these reactions is proposed to involve organoborane addition across a Rh-0 bond which activates the catalyst for isomerization and hydrogenation. INTRODUCTION Catalytic isomerization of alkenes is a characteristic transformation of transition metal hy- drides that often accompanies hydrogenation1 and is one of the most thoroughly studied catalytic reactions.2"4 Compounds of cobalt, nickel, palladium, platinum, rhodium, and ruthenium are effective,2 but other transition metal compounds have also been employed for catalytic isomeriza- tions.2"4 Although the nature of this transformation is dependent on the catalyst, selectivity for alkene isomerization generally favors reactions with monosubstituted ethylenes over di- and tri-sub- stituted ethylenes. In the course of our investigations of the catalytic effectiveness of dirhodium(ll) tetrakis(carboxylates) we have uncovered a useful methodology for the generation of rhodium hydride species that, as we now report, are surprisingly effective for the isomerization of alkenes as well as for hydrogen transfer hydrogenation. MATERIALS AND METHODS Reactions were performed in a round bottom flask equipped with a screw cap that was fitted with a septum for convenient withdrawal of aliquots. -

September 17, 2007
Pre-Feasibility Report M/s. Neogen Chemicals Ltd. 1 1. Introduction M/s. Neogen Chemicals Ltd. is a new unit located at Plot No. Z/96/B SEZ Dahej, District: Bharuch, Gujarat. Now, the unit proposes to manufacture different type of synthetic organic dyes and pesticide products at above sited address. 2. Cost of Project Cost of existing project is 55 crore &, out of which 5 crore will be used for Environment Management System. 3. Production Capacity Production capacity is prescribe below: List of Products Sr. Name of Products Quantity No. (MT/Year) (MT/month) 1 Bromination and Chlorination of Alcohols 1.1. Ethyl Bromide 3500 291.67 1.2. n-Propyl Bromide 1.3. Iso Propyl Bromide 1.4. n-Butyl Bromide 1.5. Iso Butyl Bromide 1.6. Sec-Butyl Bromide 1.7. n-Hexyl Bromide 1.8. n-Heptyl Bromide 1.9. n-Octyl Bromide 1.10. n-Decyl Bromide 1.11. Lauryl Bromide 1.12. Cetyl Bromide 1.13. Myristyl Bromide 1.14. Stearyl Bromide 1.15. 1,2 Dibromo Ethane 1.16. 1,3 Dibromo Propane 1.17. 1,4 Dibromo Butane 1.18. 1,5 Dibromo pentane M/s. Neogen Chemicals Ltd. 2 Sr. Name of Products Quantity No. (MT/Year) (MT/month) 1.19. 1,6 Dibromo Hexane 1.20. 1 Chloro 2 Ethyl Hexane 1.21. 6 Chloro 1 Hexanol 1.22. 3 Chloro Propanol 1.23. 1,6 Dichloro Hexane 1.24. Cyclo Propyl Methyl Bromide 1.25. Cyclo Pentyl Bromide 1.26. Cyclo Pentyl Chloride 2. Bromination of Organic Acids and Esterification thereof 2.1. -

Conformational Studies of Aliphatic Secondary Ozonides (Propene, 1-Butene and 1-Heptene) by Means of FTIR Spectroscopy
DOI: 10.2478/s11532-006-0031-3 Research article CEJC 4(4) 2006 578–591 Conformational studies of aliphatic secondary ozonides (propene, 1-butene and 1-heptene) by means of FTIR spectroscopy Ruta Bariseviciute, Justinas Ceponkus, Alytis Gruodis, Valdas Sablinskas∗ Department of General Physics and Spectroscopy, Vilnius University, Vilnius-01513, Lithuania Received 6 January 2006; accepted 31 May 2006 Abstract: Ozonization reaction of simple alkenes was studied by means of FT infrared absorption gas spectroscopy. The reaction was performed at 95 K in neat films of the reactants. IR absorption spectra of the gaseous products were recorded. The spectra were analyzed combining experimental results with theoretical calculations performed at B3LYP 6-311++G (3df, 3pd) level. We found that among all theoretically predicted conformers of propene secondary ozonide, only one which has the O-O half-chair configuration for the five membered ring and the radical attached in the equatorial position was present in the sample. Samples of 1-butene and 1-heptene secondary ozonides consist from two conformers of very similar energy (ΔH =0.3 kJ/mol). The most stable conformer for both ozonides is the one with O-O half-chair configuration of the five membered ring and the radical attached in equatorial position and the aliphatic chain in gauche position. The second stable conformer has the aliphatic chain in anti position. c Versita Warsaw and Springer-Verlag Berlin Heidelberg. All rights reserved. Keywords: Ozonization, conformational analysis, secondary ozonides, IR spectroscopy 1 Introduction The ability of ozone to cleave double bonds in olefins has been known for more than 50 years. -

' United "States Patent Office
Patented on. 20, 1.942 I . 2,299,411 ' UNITED "STATES PATENT OFFICE CATALYZED HYDROBROMINATION OF UN SATURATED ORGANIC COMPOUNDS Fredericlr Rust and William E. Vaughan, ' Berkeley, Calif., assignors. to Shell Develop ment Company, San Francisco, Calif., a corpo ration of Delaware No Drawing. 4 Application August 25, 1941, Serial No. 408,212 - 9'Claims. (01. 260-663) ' This invention relates to an improved process for the hydrobromination of unsaturated organic hydes and metal alkyls which tend to initiate I compounds, and more particularly to improve the reaction chains. v ments in the method of controlling the addition It is known that the presence of peroxide: orv of hydrogen bromide to unsymmetrical organic of peroxide-forming compounds in unsaturated compounds containing at least one ole?nic or organic compounds, e. g. unsaturated hydrocar acetylenic linkage to produce addition products bons, is undesirable. For example, organic per of a predetermined character. oxides, when present even in relatively small con It is known that hydrogen halides may be centrations, tend to catalyze the polymerization of a large number of unsaturated hydrocarbons, . added to unsaturated hydrocarbons and to var 10 ious unsaturated derivatives thereof. In fact, and particularly diole?ns. As to the “abnormal” in 1870 Markowniko? stated that “if an addition ofhydrogen bromide to unsaturates by metrical hydrocarbon combines with a halogen effecting the reaction under the deliberate in acid, the halogen adds to the carbon atom with ?uence, of light, and particularly of ultra-violet fewer hydrogen atoms, i. e. to the carbon atom radiations having wave-lengths of below about which is more'under the in?uence of other car' 2900 to 3000 Angstrom units, such processes ne bon atoms.” The same investigator further de cessitate the use of special equipment, such as termined that when a hydrogen halide is added reaction vessels provided with or containing to a halogenated unsaturated compound such as lamps made of quartz Or other suitable mate rials, e. -

Chemical Compatibility Chart X
Chemical Compatibility Chart Below is a chart adapted from the CRC Laboratory Handbook, which groups various chemicals in to 23 groups with examples and incompatible chemical groups. This chart is by no means complete but it will aid in making decisions about storage. For more complete information please refer to the MSDS for the specific chemical. Examples of each group can be found on the next pages. 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 Monomers Polymerizable Esters Alcohols, Glycols, Glycol Ether Amines and Alkanolamines Halogenated Compounds Aldehydes Acetaldehyde Saturated Hydrocar Aromatic Hydrocarbons Acid Anhydrides Alkylene Oxides Inorganic Acids Petrolium Oils Organic Acids Cyanohydrins Phosphorus Ammonia Group Halogens Ketones Caustics Phenols Nitriles Olefins Ethers Number/Chemical Esters Type bons Inorganic 1 x x x x x x x x x x x x x x x x x Acids 2 Organic Acids x x x x x x x x x x 3 Caustics x x x x x x x x x x x x x Amines and 4 x x x x x x x x x x x x Alkanolamines Halogenated 5 x x x x x x Compounds Alcohols, 6 Glycols, Glycol x x x x x x Ether Aldehydes 7 x x x x x x x x x x x x Acetaldehyde 8 Ketones x x x x x x Saturated 9 x Hydrocarbons Aromatic 10 x x Hydrocarbons 11 Olefins x x x 12 Petrolium Oils x 13 Esters x x x x x Monomers 14 Polymerizable x x x x x x x x x x x x Esters 15 Phenols x x x x x x x Alkylene 16 x x x x x x x x x x x x Oxides 17 Cyanohydrins x x x x x x x x x 18 Nitriles x x x x x x 19 Ammonia x x x x x x x x x x x 20 Halogens x x x x x x x x x x x x x x 21 Ethers x x x 22 Phosphorus x x x x Acid 23 x x x x x x x x x x Anhydrides X - Indicates chemicals that are incompatible and should not be stored together. -

Properties and Synthesis. Elimination Reactions of Alkyl Halides
P1: PBU/OVY P2: PBU/OVY QC: PBU/OVY T1: PBU Printer: Bind Rite JWCL234-07 JWCL234-Solomons-v1 December 8, 2009 21:37 7 ALKENES AND ALKYNES I: PROPERTIES AND SYNTHESIS. ELIMINATION REACTIONS OF ALKYL HALIDES SOLUTIONS TO PROBLEMS 7.1 (a) ( E )-1-Bromo-1-chloro-1-pentene or ( E )-1-Bromo-1-chloropent-1-ene (b) ( E )-2-Bromo-1-chloro-1-iodo-1-butene or ( E )-2-Bromo-1-chloro-1-iodobut-1-ene (c) ( Z )-3,5-Dimethyl-2-hexene or ( Z )-3,5-Dimethylhex-2-ene (d) ( Z )-1-Chloro-1-iodo-2-methyl-1-butene or ( Z )-1-Chloro-1-iodo-2-methylbut-1-ene (e) ( Z,4 S )-3,4-Dimethyl-2-hexene or ( Z,4 S )-3,4-Dimethylhex-2-ene (f) ( Z,3 S )-1-Bromo-2-chloro-3-methyl-1-hexene or (Z,3 S )-1-Bromo-2-chloro-3-methylhex-1-ene 7.2 < < Order of increasing stability 7.3 (a), (b) H − 2 ∆ H° = − 119 kJ mol 1 Pt 2-Methyl-1-butene pressure (disubstituted) H2 − ∆ H° = − 127 kJ mol 1 Pt 3-Methyl-1-butene pressure (monosubstituted) H2 − ∆ H° = − 113 kJ mol 1 Pt 2-Methyl-2-butene pressure (trisubstituted) (c) Yes, because hydrogenation converts each alkene into the same product. 106 CONFIRMING PAGES P1: PBU/OVY P2: PBU/OVY QC: PBU/OVY T1: PBU Printer: Bind Rite JWCL234-07 JWCL234-Solomons-v1 December 8, 2009 21:37 ALKENES AND ALKYNES I: PROPERTIES AND SYNTHESIS 107 H H (d) > > H H H (trisubstituted) (disubstituted) (monosubstituted) Notice that this predicted order of stability is confirmed by the heats of hydro- genation. -
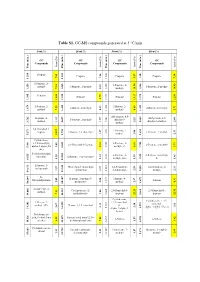
Table S1. GC-MS Compounds Generated at 5 °C/Min
Table S1. GC-MS compounds generated at 5 °C/min. 0 wt.% 10 wt.% 30 wt.% 50 wt.% GC GC GC GC Compounds Compounds Compounds Compounds Area (%) Area (%) Area (%) Area (%) Time (min.) (min.) Time Time (min.) (min.) Time (min.) Time (min.) Time Propene Propene Propene Propene 7.42 7.42 1.26 5.47 1.20 7.38 1.200 1.200 1.200 1.214 1.214 1-Propene, 2- 1-Propene, 2- methyl- 1-Propene, 2-methyl- 1-Propene, 2-methyl- 7.34 7.34 2.41 methyl- 1.245 1.245 1.245 1.259 1.259 10.54 1.246 14.90 Pentane Pentane Pentane Pentane 3.79 3.79 1.375 1.375 1.395 1.395 11.81 1.381 13.17 1.375 12.83 1-Pentene, 2- 2-Butene, 2- 2-Butene, 2-methyl- 2-Butene, 2-methyl- methyl- 7.25 0.93 methyl- 4.65 5.71 1.426 1.426 1.420 1.420 1.679 1.679 1H-Pyrazole, 4,5- Heptane, 4- 1H-Pyrazole, 4,5- 1-Pentene, 2-methyl- dihydro-5- methyl- 1.92 1.57 2.25 dihydro-5-methyl- 2.44 1.659 1.659 1.562 1.569 3.762 3.762 methyl- 2,4-Dimethyl-1- 1-Pentene, 2- heptene 2-Butene, 2,3-dimethyl- 1-Pentene, 2-methyl- 1.27 1.27 methyl- 3.08 2.33 1.653 1.653 1.653 5.671 5.671 34.57 1.750 Cyclohexane, 1,3,5-trimethyl-, 2-Pentene, 3- 2,4-Dimethyl-1-heptene 2-Pentene, 2-methyl- alpha.,3.alpha.,5.b 2.03 9.11 methyl-, €- 6.11 5.73 5.605 5.605 1.743 1.744 5.981 5.981 eta.)- 5-Aminoisoxazole 2-Pentene, 3- 2-Pentene, 3-methyl-, Spermine Ethanone, 1-cyclopentyl- 1.55 1.55 1.32 methyl-, (Z)- 1.59 €- 1.46 1.828 1.828 1.828 6.925 6.925 10.560 Ethanone, 1- 1R,2c,3t,4t-Tetramethyl- 1,3-Pentadiene, 1,4-Hexadiene, 5- cyclopentyl- 3.57 3.57 cyclohexan 0.97 2,3-dimethyl- 2.28 methyl- 2.31 2.584 2.584 2.585 10.613 10.613 10.638 N- Heptane, 2-methyl-3- Heptane, 4- Methylallylamine Toluene 2.49 2.49 methylene- 2.50 methyl- 2.97 4.31 3.697 3.697 3.697 14.047 14.047 10.684 10.684 2-Undecene, 4- Cyclopentane, (2- 2,4-Dimethyl-1- 2,4-Dimethyl-1- methyl- 7.81 7.81 methylbutyl)- 1.00 heptene heptene 5.605 5.605 22.29 22.29 5.605 12.95 14.086 14.086 14.163 Cyclohexane, Cyclohexane, 1,3,5- 2-Decene, 7- 1,3,5-trimethyl-, trimethyl- methyl-, (Z)- Hexane, 2,3,4-trimethyl- (1. -

Linear Alpha-Olefins (681.5030)
IHS Chemical Chemical Economics Handbook Linear alpha-Olefins (681.5030) by Elvira O. Camara Greiner with Yoshio Inoguchi Sample Report from 2010 November 2010 ihs.com/chemical November 2010 LINEAR ALPHA-OLEFINS Olefins 681.5030 B Page 2 The information provided in this publication has been obtained from a variety of sources which SRI Consulting believes to be reliable. SRI Consulting makes no warranties as to the accuracy completeness or correctness of the information in this publication. Consequently SRI Consulting will not be liable for any technical inaccuracies typographical errors or omissions contained in this publication. This publication is provided without warranties of any kind either express or implied including but not limited to implied warranties of merchantability fitness for a particular purpose or non-infringement. IN NO EVENT WILL SRI CONSULTING BE LIABLE FOR ANY INCIDENTAL CONSEQUENTIAL OR INDIRECT DAMAGES (INCLUDING BUT NOT LIMITED TO DAMAGES FOR LOSS OF PROFITS BUSINESS INTERRUPTION OR THE LIKE) ARISING OUT OF THE USE OF THIS PUBLICATION EVEN IF IT WAS NOTIFIED ABOUT THE POSSIBILITY OF SUCH DAMAGES. BECAUSE SOME STATES DO NOT ALLOW THE EXCLUSION OR LIMITATION OF LIABILITY FOR CONSEQUENTIAL OR INCIDENTAL DAMAGES THE ABOVE LIMITATION MAY NOT APPLY TO YOU. IN SUCH STATES SRI CONSULTING’S LIABILITY IS LIMITED TO THE MAXIMUM EXTENT PERMITTED BY SUCH LAW. Certain statements in this publication are projections or other forward-looking statements. Any such statements contained herein are based upon SRI Consulting’s current knowledge and assumptions about future events including without limitation anticipated levels of global demand and supply expected costs trade patterns and general economic political and marketing conditions. -

United States Patent Office
Patented July 16, 1957 United States Patent Office 1. 2 be isolated from a solution in petroleum ether by frac tional crystallization. 2,799,685 The homologues, i. e. for example the pentachloro and tetrachloro-derivatives and the corresponding ana UNSATURATEDPROCESS FOR POLYCYCLIC THEIR MANUFACTURE SULFITES AND A logues of methyl and ethyl can be obtained in a corre einz Frensch, Helmut Goebel, Wilhelm Staudermann, sponding manner. : and Walter Finkenbrink, Frankfurt am Main, Ger. As Diels-Alder-components suitable for the manufac many, assignors to Farbwerke Hoechst Aktiengesell ture of Sulfites of the kind described above, there come schaft yormals Meister Lucius & Brining, Frankfurt also into consideration the halogenated alkyl derivatives, am Main, Germany; a corporation of the Federal Re O for example, dimethylcyclopentadiene. public of Germany The polycyclic halogenated sulfites are readily crystal lizing colorless compounds. No Drawing. Application April 8, 1955, The products of this invention may be used for pro Serial No. 500,264 tecting wood, paper, textiles and leather and as inter Claims priority, application Germany April 17, 1954 5 mediates for the manufacture of drugs. In the pure state, they are practically, odourless and are, therefore, 9 Claims. (CI. 260-327) especially suitable for the mentioned purposes. They The present invention provides new unsaturated poly have a curative and prophylactic effect and are prefer cyclic sulfites corresponding to the following general ably used, if desired in admixture with other insecticides, 20 ovicides, fungicides, herbicides or fertilizers, for combat formula: ing animal and fungoid pests. Y - In addition to their excellent action as pesticides the substances hereinbefore described are distinguished by 4R/V their chemical stability.