Epithelial-Derived IL-18 Regulates Th17 Cell Differentiation and Foxp3 Þ Treg Cell Function in the Intestine
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

DS Human IL-18
® MSD Human IL-18 Kit For quantitative determination in human serum, plasma, and tissue culture supernatants Alzheimer’s Disease IL-18 BioProcess Cardiac Cell Signaling Clinical Immunology Cytokines Growth Factors Hypoxia Immunogenicity Inflammation Metabolic Oncology Interleukin-18 (IL-18) is an 18 kDa cytokine and a co-stimulatory factor that is produced in Kuppfer cells, activated macrophages, Toxicology keratinocytes, and intestinal epithelial cells.1 One of the main functions of IL-18 is to promote the production of IFN-γ from T and NK Vascular cells, particularly in the presence of IL-12p70. IL-18 also promotes the secretion of other proinflammatory cytokines like TNF-α, IL-1β, 2 and GM-CSF that enhance the migration and activtion of neutrophils during microbial infections. IL-18 enhances cytotoxic activity and 2,3 Catalog Numbers proliferation of CD8+ T and NK cells and has been shown to stimulate the production of IL-13 and other Th2 cytokines. Dysregulation of IL-18 may therefore contribute to inflammatory-associated disorders, unchecked infections, autoimmune diseases such as 2,3 Human IL-18 Kit rheumatoid arthritis, acute and chronic kidney injury, cancer, and pathogenic conditions related to metabolic syndrome. Kit size The MSD Human IL - 18 assay is available on 96-well 4-spot plates. This datasheet outlines the performance of the assay. 1 plate K151MCD-1 5 plates K151MCD-2 Assay Sensitivity 25 plates K151MCD-4 IL-18 LLOD (pg/mL) 0.71 The lower limit of detection (LLOD) is a calculated concentration based on a signal Ordering information 2.5 standard deviations above the background (zero calibrator blank). -

Role of IL-4 Receptor &Alpha
Role of IL-4 receptor a–positive CD41 T cells in chronic airway hyperresponsiveness Frank Kirstein, PhD, Natalie E. Nieuwenhuizen, PhD,* Jaisubash Jayakumar, PhD, William G. C. Horsnell, PhD, and Frank Brombacher, PhD Cape Town, South Africa, and Berlin, Germany Background: TH2 cells and their cytokines are associated with IL-17–producing T cells and, consequently, increased airway allergic asthma in human subjects and with mouse models of neutrophilia. allergic airway disease. IL-4 signaling through the IL-4 receptor Conclusion: IL-4–responsive T helper cells are dispensable for 1 a (IL-4Ra) chain on CD4 T cells leads to TH2 cell acute OVA-induced airway disease but crucial in maintaining differentiation in vitro, implying that IL-4Ra–responsive CD41 chronic asthmatic pathology. (J Allergy Clin Immunol T cells are critical for the induction of allergic asthma. However, 2016;137:1852-62.) mechanisms regulating acute and chronic allergen-specific T 2 H Key words: responses in vivo remain incompletely understood. TH2 cell, acute allergic airway disease, chronic asthma, Objective: This study defines the requirements for IL-4Ra– cytokine receptors, IL-4, IL-13, gene-deficient mice responsive CD41 T cells and the IL-4Ra ligands IL-4 and IL-13 in the development of allergen-specific TH2 responses during the Allergic asthma is a chronic inflammatory disease of the airways onset and chronic phase of experimental allergic airway disease. characterized by an inappropriate immune response to harmless Methods: Development of acute and chronic ovalbumin environmental antigens. T 2 cells regulate adaptive immune 1 H (OVA)–induced allergic asthma was assessed weekly in CD4 T responses to allergens, and their presence correlates with disease 2 cell–specific IL-4Ra–deficient BALB/c mice (LckcreIL-4Ra /lox) symptoms in human subjects and mice.1 IL-4 plays a crucial role and respective control mice in the presence or absence of IL-4 in the in vitro and in vivo differentiation of TH2 cells, suggesting or IL-13. -

Genetics of Interleukin 1 Receptor-Like 1 in Immune and Inflammatory Diseases
Current Genomics, 2010, 11, 591-606 591 Genetics of Interleukin 1 Receptor-Like 1 in Immune and Inflammatory Diseases Loubna Akhabir and Andrew Sandford* Department of Medicine, University of British Columbia, UBC James Hogg Research Centre, Providence Heart + Lung Institute, Room 166, St. Paul's Hospital, 1081 Burrard Street, Vancouver, BC V6Z 1Y6, Canada Abstract: Interleukin 1 receptor-like 1 (IL1RL1) is gaining in recognition due to its involvement in immune/inflamma- tory disorders. Well-designed animal studies have shown its critical role in experimental allergic inflammation and human in vitro studies have consistently demonstrated its up-regulation in several conditions such as asthma and rheumatoid ar- thritis. The ligand for IL1RL1 is IL33 which emerged as playing an important role in initiating eosinophilic inflammation and activating other immune cells resulting in an allergic phenotype. An IL1RL1 single nucleotide polymorphism (SNP) was among the most significant results of a genome-wide scan inves- tigating eosinophil counts; in the same study, this SNP associated with asthma in 10 populations. The IL1RL1 gene resides in a region of high linkage disequilibrium containing interleukin 1 receptor genes as well as in- terleukin 18 receptor and accessory genes. This poses a challenge to researchers interested in deciphering genetic associa- tion signals in the region as all of the genes represent interesting candidates for asthma and allergic disease. The IL1RL1 gene and its resulting soluble and receptor proteins have emerged as key regulators of the inflammatory proc- ess implicated in a large variety of human pathologies We review the function and expression of the IL1RL1 gene. -
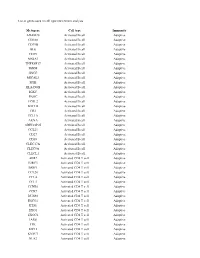
List of Genes Used in Cell Type Enrichment Analysis
List of genes used in cell type enrichment analysis Metagene Cell type Immunity ADAM28 Activated B cell Adaptive CD180 Activated B cell Adaptive CD79B Activated B cell Adaptive BLK Activated B cell Adaptive CD19 Activated B cell Adaptive MS4A1 Activated B cell Adaptive TNFRSF17 Activated B cell Adaptive IGHM Activated B cell Adaptive GNG7 Activated B cell Adaptive MICAL3 Activated B cell Adaptive SPIB Activated B cell Adaptive HLA-DOB Activated B cell Adaptive IGKC Activated B cell Adaptive PNOC Activated B cell Adaptive FCRL2 Activated B cell Adaptive BACH2 Activated B cell Adaptive CR2 Activated B cell Adaptive TCL1A Activated B cell Adaptive AKNA Activated B cell Adaptive ARHGAP25 Activated B cell Adaptive CCL21 Activated B cell Adaptive CD27 Activated B cell Adaptive CD38 Activated B cell Adaptive CLEC17A Activated B cell Adaptive CLEC9A Activated B cell Adaptive CLECL1 Activated B cell Adaptive AIM2 Activated CD4 T cell Adaptive BIRC3 Activated CD4 T cell Adaptive BRIP1 Activated CD4 T cell Adaptive CCL20 Activated CD4 T cell Adaptive CCL4 Activated CD4 T cell Adaptive CCL5 Activated CD4 T cell Adaptive CCNB1 Activated CD4 T cell Adaptive CCR7 Activated CD4 T cell Adaptive DUSP2 Activated CD4 T cell Adaptive ESCO2 Activated CD4 T cell Adaptive ETS1 Activated CD4 T cell Adaptive EXO1 Activated CD4 T cell Adaptive EXOC6 Activated CD4 T cell Adaptive IARS Activated CD4 T cell Adaptive ITK Activated CD4 T cell Adaptive KIF11 Activated CD4 T cell Adaptive KNTC1 Activated CD4 T cell Adaptive NUF2 Activated CD4 T cell Adaptive PRC1 Activated -

Role of Interleukin 36Γ in Host Defense Against Tuberculosis Fadhil Ahsan,1,2,A Pedro Moura-Alves,1,A Ute Guhlich-Bornhof,1 Marion Klemm,1 Stefan H
The Journal of Infectious Diseases MAJOR ARTICLE Role of Interleukin 36γ in Host Defense Against Tuberculosis Fadhil Ahsan,1,2,a Pedro Moura-Alves,1,a Ute Guhlich-Bornhof,1 Marion Klemm,1 Stefan H. E. Kaufmann,1 and Jeroen Maertzdorf1 Downloaded from https://academic.oup.com/jid/article-abstract/214/3/464/2577344 by Deutsches Rheumaforschungs Zentrum user on 26 June 2019 1Department of Immunology, Max Planck Institute for Infection Biology, and 2ZIBI Graduate School Berlin, Germany Tuberculosis remains a major killer worldwide, not the least because of our incomplete knowledge of protective and pathogenic immune mechanism. The roles of the interleukin 1 (IL-1) and interleukin 18 pathways in host defense are well established, as are their regulation through the inflammasome complex. In contrast, the regulation of interleukin 36γ (IL-36γ), a recently described member of the IL-1 family, and its immunological relevance in host defense remain largely unknown. Here we show that Myco- bacterium tuberculosis infection of macrophages induces IL-36γ production in a 2-stage-regulated fashion. In the first stage, mi- crobial ligands trigger host Toll-like receptor and MyD88-dependent pathways, leading to IL-36γ secretion. In the second stage, endogenous IL-1β and interleukin 18 further amplify IL-36γ synthesis. The relevance of this cytokine in the control of M. tuber- culosis is demonstrated by IL-36γ–induced antimicrobial peptides and IL-36 receptor–dependent restriction of M. tuberculosis growth. Thus, we provide first insight into the induction and regulation of the proinflammatory cytokine IL-36γ during tuberculosis. Keywords. IL-36γ; Mycobacterium tuberculosis; TLR; inflammasome; antimicrobial peptide. -

The Role of Interleukin-18 in the Metabolic Syndrome Marius Trøseid1*, Ingebjørg Seljeflot1,2, Harald Arnesen1,2
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Springer - Publisher Connector Trøseid et al. Cardiovascular Diabetology 2010, 9:11 CARDIO http://www.cardiab.com/content/9/1/11 VASCULAR DIABETOLOGY REVIEW Open Access The role of interleukin-18 in the metabolic syndrome Marius Trøseid1*, Ingebjørg Seljeflot1,2, Harald Arnesen1,2 Abstract The metabolic syndrome is thought to be associated with a chronic low-grade inflammation, and a growing body of evidence suggests that interleukin-18 (IL-18) might be closely related to the metabolic syndrome and its conse- quences. Circulating levels of IL-18 have been reported to be elevated in subjects with the metabolic syndrome, to be closely associated with the components of the syndrome, to predict cardiovascular events and mortality in populations with the metabolic syndrome and to precede the development of type 2 diabetes. IL-18 is found in the unstable atherosclerotic plaque, in adipose tissue and in muscle tissue, and is subject to several regulatory steps including cleavage by caspase-1, inactivation by IL-18 binding protein and the influence of other cytokines in modulating its interaction with the IL-18 receptor. The purpose of this review is to outline the role of IL-18 in the metabolic syndrome, with particular emphasis on cardiovascular risk and the potential effect of life style interventions. Introduction developing CVD is approximately doubled in the meta- The metabolic syndrome is a cluster of risk factors that bolic syndrome [10]. In a meta-analysis including identifies a population with increased risk for developing 43 cohorts, the relative risk for cardiovascular events type 2 diabetes mellitus and cardiovascular disease and death was 1.78, with the highest risk in women [11]. -

Single-Cell RNA Sequencing Demonstrates the Molecular and Cellular Reprogramming of Metastatic Lung Adenocarcinoma
ARTICLE https://doi.org/10.1038/s41467-020-16164-1 OPEN Single-cell RNA sequencing demonstrates the molecular and cellular reprogramming of metastatic lung adenocarcinoma Nayoung Kim 1,2,3,13, Hong Kwan Kim4,13, Kyungjong Lee 5,13, Yourae Hong 1,6, Jong Ho Cho4, Jung Won Choi7, Jung-Il Lee7, Yeon-Lim Suh8,BoMiKu9, Hye Hyeon Eum 1,2,3, Soyean Choi 1, Yoon-La Choi6,10,11, Je-Gun Joung1, Woong-Yang Park 1,2,6, Hyun Ae Jung12, Jong-Mu Sun12, Se-Hoon Lee12, ✉ ✉ Jin Seok Ahn12, Keunchil Park12, Myung-Ju Ahn 12 & Hae-Ock Lee 1,2,3,6 1234567890():,; Advanced metastatic cancer poses utmost clinical challenges and may present molecular and cellular features distinct from an early-stage cancer. Herein, we present single-cell tran- scriptome profiling of metastatic lung adenocarcinoma, the most prevalent histological lung cancer type diagnosed at stage IV in over 40% of all cases. From 208,506 cells populating the normal tissues or early to metastatic stage cancer in 44 patients, we identify a cancer cell subtype deviating from the normal differentiation trajectory and dominating the metastatic stage. In all stages, the stromal and immune cell dynamics reveal ontological and functional changes that create a pro-tumoral and immunosuppressive microenvironment. Normal resident myeloid cell populations are gradually replaced with monocyte-derived macrophages and dendritic cells, along with T-cell exhaustion. This extensive single-cell analysis enhances our understanding of molecular and cellular dynamics in metastatic lung cancer and reveals potential diagnostic and therapeutic targets in cancer-microenvironment interactions. 1 Samsung Genome Institute, Samsung Medical Center, Seoul 06351, Korea. -

B Cell Checkpoints in Autoimmune Rheumatic Diseases
REVIEWS B cell checkpoints in autoimmune rheumatic diseases Samuel J. S. Rubin1,2,3, Michelle S. Bloom1,2,3 and William H. Robinson1,2,3* Abstract | B cells have important functions in the pathogenesis of autoimmune diseases, including autoimmune rheumatic diseases. In addition to producing autoantibodies, B cells contribute to autoimmunity by serving as professional antigen- presenting cells (APCs), producing cytokines, and through additional mechanisms. B cell activation and effector functions are regulated by immune checkpoints, including both activating and inhibitory checkpoint receptors that contribute to the regulation of B cell tolerance, activation, antigen presentation, T cell help, class switching, antibody production and cytokine production. The various activating checkpoint receptors include B cell activating receptors that engage with cognate receptors on T cells or other cells, as well as Toll-like receptors that can provide dual stimulation to B cells via co- engagement with the B cell receptor. Furthermore, various inhibitory checkpoint receptors, including B cell inhibitory receptors, have important functions in regulating B cell development, activation and effector functions. Therapeutically targeting B cell checkpoints represents a promising strategy for the treatment of a variety of autoimmune rheumatic diseases. Antibody- dependent B cells are multifunctional lymphocytes that contribute that serve as precursors to and thereby give rise to acti- cell- mediated cytotoxicity to the pathogenesis of autoimmune diseases -

Cd4/Cd8 Panel, Blood
Lab Dept: Flow and Immunology Test Name: CD4/CD8 PANEL, BLOOD General Information Lab Order Codes: C48P Synonyms: Helper/Suppressor Ratio; T- cells; T-cell subsets; T-cell phenotyping See also: Immune Status Panel and Comprehensive Immune Status Panel CPT Codes: 86359 – T cells, total count 86360 – T cells; absolute CD4 and CD8 count, including ratio Test Includes: CD4(CD3+) and CD8(CD3+) relative percentages, absolute values and a calculated Helper/Suppressor ratio. Logistics Test Indications: This is a minimal antibody panel to monitor immune status. Lab Testing Sections: Flow Cytometry Phone Numbers: MIN Lab: 612-813-6280 STP Lab: 651-220-6550 Test Availability: 3 times weekly determined by volume. Transport collected specimen immediately to Flow Cytometry. Routine testing is not available on weekends or holidays. Therefore, specimens cannot be used if drawn the day before a 3 day weekend such as Memorial Day, Labor Day or major holiday that falls on a Monday or Friday. Turnaround Time: 1 – 3 days Special Instructions: See Test Availability Specimen Specimen Type: Whole blood Container: Lavender top (EDTA) tube Draw Volume: 2 mL blood in a 2 mL Lavender (EDTA) tube Minimum volume: 0.5 mL in an EDTA microtainer Collection: Routine venipuncture Special Processing: Keep sample at room temperature and forward promptly to the laboratory. Do not centrifuge, refrigerate, or freeze sample. Patient Preparation: None Sample Rejection: Specimens will not be processed that are clotted; hemolyzed; greater than 72 hours old; collected in the wrong tube type (0.5 mL in a 2 mL tube), or that have been held or handled at a temperature other than room temperature Interpretive Reference Range: Age-dependant reference ranges provided with results. -

How Relevant Are Bone Marrow-Derived Mast Cells (Bmmcs) As Models for Tissue Mast Cells? a Comparative Transcriptome Analysis of Bmmcs and Peritoneal Mast Cells
cells Article How Relevant Are Bone Marrow-Derived Mast Cells (BMMCs) as Models for Tissue Mast Cells? A Comparative Transcriptome Analysis of BMMCs and Peritoneal Mast Cells 1, 2, 1 1 2,3 Srinivas Akula y , Aida Paivandy y, Zhirong Fu , Michael Thorpe , Gunnar Pejler and Lars Hellman 1,* 1 Department of Cell and Molecular Biology, Uppsala University, The Biomedical Center, Box 596, SE-751 24 Uppsala, Sweden; [email protected] (S.A.); [email protected] (Z.F.); [email protected] (M.T.) 2 Department of Medical Biochemistry and Microbiology, Uppsala University, The Biomedical Center, Box 589, SE-751 23 Uppsala, Sweden; [email protected] (A.P.); [email protected] (G.P.) 3 Department of Anatomy, Physiology and Biochemistry, Swedish University of Agricultural Sciences, Box 7011, SE-75007 Uppsala, Sweden * Correspondence: [email protected]; Tel.: +46-(0)18-471-4532; Fax: +46-(0)18-471-4862 These authors contributed equally to this work. y Received: 29 July 2020; Accepted: 16 September 2020; Published: 17 September 2020 Abstract: Bone marrow-derived mast cells (BMMCs) are often used as a model system for studies of the role of MCs in health and disease. These cells are relatively easy to obtain from total bone marrow cells by culturing under the influence of IL-3 or stem cell factor (SCF). After 3 to 4 weeks in culture, a nearly homogenous cell population of toluidine blue-positive cells are often obtained. However, the question is how relevant equivalents these cells are to normal tissue MCs. By comparing the total transcriptome of purified peritoneal MCs with BMMCs, here we obtained a comparative view of these cells. -

IL-33 Can Promote Survival, Adhesion and Cytokine Production in Human
Laboratory Investigation (2007) 87, 971–978 & 2007 USCAP, Inc All rights reserved 0023-6837/07 $30.00 IL-33 can promote survival, adhesion and cytokine production in human mast cells Motoyasu Iikura1,2, Hajime Suto1,3, Naoki Kajiwara4, Keisuke Oboki5, Tatsukuni Ohno5, Yoshimichi Okayama4, Hirohisa Saito3,5, Stephen J Galli1 and Susumu Nakae1,3,5 IL-33 is a recently identified member of the IL-1 family of molecules, which also includes IL-1 and IL-18. IL-33 binds to the receptor, T1/ST2/IL-1R4, and can promote cytokine secretion by Th2 cells and NF-kB phosphorylation in mouse mast cells. However, the effects of these molecules, especially IL-33, in human mast cells are poorly understood. Expression of the receptors for IL-1 family molecules, specifically, IL-1R1, IL-18R and T1/ST2, was detectable intracellularly in human umbilical cord blood-derived mast cells (HUCBMCs) by flow cytometry, but was scarcely detectable on the cells’ surface. However, IL-1b, IL-18 or IL-33 induced phosphorylation of Erk, p38 and JNK in naı¨ve HUCBMCs, and IL-33 or IL-1b, but not IL-18, enhanced the survival of naive HUCBMCs and promoted their adhesion to fibronectin. IL-33 or IL-1b also induced IL-8 and IL-13 production in naı¨ve HUCBMCs, and enhanced production of these cytokines in IgE/anti-IgE-stimulated HUCBMCs, without enhancing secretion of either PGD2 or histamine. Moreover, IL-33-mediated IL-8 production by HUCBMCs was markedly reduced by the p38 MAPK inhibitor, SB203580. In contrast to findings with mouse mast cells, IL-18 neither induced nor enhanced secretion of the mediators PGD2 or histamine by HUCBMCs. -

Rational Design of a CD4 Mimic That Inhibits HIV-1 Entry and Exposes Cryptic Neutralization Epitopes
RESEARCH ARTICLE Rational design of a CD4 mimic that inhibits HIV-1 entry and exposes cryptic neutralization epitopes Loïc Martin1, François Stricher1, Dorothée Missé2, Francesca Sironi3, Martine Pugnière4, Philippe Barthe5, Rafael Prado-Gotor5, Isabelle Freulon1, Xavier Magne2, Christian Roumestand5, André Ménez1, Paolo Lusso3, Francisco Veas2, and Claudio Vita1* Published online 16 December 2002; doi:10.1038/nbt768 The conserved surfaces of the human immunodeficiency virus (HIV)-1 envelope involved in receptor binding represent potential targets for the development of entry inhibitors and neutralizing antibodies. Using structural information on a CD4-gp120-17b antibody complex, we have designed a 27-amino acid CD4 mimic, CD4M33, that presents optimal interactions with gp120 and binds to viral particles and diverse HIV-1 envelopes with CD4-like affinity.This mini-CD4 inhibits infection of both immortalized and primary cells by HIV-1, including pri- http://www.nature.com/naturebiotechnology mary patient isolates that are generally resistant to inhibition by soluble CD4. Furthermore, CD4M33 pos- sesses functional properties of CD4, including the ability to unmask conserved neutralization epitopes of gp120 that are cryptic on the unbound glycoprotein. CD4M33 is a prototype of inhibitors of HIV-1 entry and, in complex with envelope proteins, a potential component of vaccine formulations, or a molecular target in phage display technology to develop broad-spectrum neutralizing antibodies. HIV-1 avoids the host immune surveillance by exposing