Patterns of Dementia Treatment and Frank Prescribing Errors in Older Adults with Parkinson Disease
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
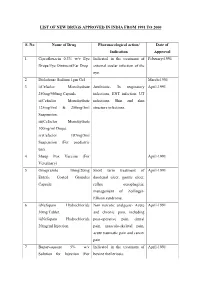
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

Research Article RP-HPLC Method for Determination of Several Nsaids and Their Combination Drugs
Hindawi Publishing Corporation Chromatography Research International Volume 2013, Article ID 242868, 13 pages http://dx.doi.org/10.1155/2013/242868 Research Article RP-HPLC Method for Determination of Several NSAIDs and Their Combination Drugs Prinesh N. Patel, Gananadhamu Samanthula, Vishalkumar Shrigod, Sudipkumar C. Modh, and Jainishkumar R. Chaudhari Department of Pharmaceutical Analysis, National Institute of Pharmaceutical Education and Research (NIPER), Balanagar, Hyderabad, Andhra Pradesh 500037, India Correspondence should be addressed to Gananadhamu Samanthula; [email protected] Received 29 June 2013; Accepted 13 October 2013 Academic Editor: Andrew Shalliker Copyright © 2013 Prinesh N. Patel et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. An RP-HPLC method for simultaneous determination of 9 NSAIDs (paracetamol, salicylic acid, ibuprofen, naproxen, aceclofenac, diclofenac, ketorolac, etoricoxib, and aspirin) and their commonly prescribed combination drugs (thiocolchicoside, moxifloxacin, clopidogrel, chlorpheniramine maleate, dextromethorphan, and domperidone) was established. The separation was performed on ∘ Kromasil C18 (250 × 4.6 mm, 5 m) at 35 C using 15 mM phosphate buffer pH 3.25 and acetonitrile with gradient elution ata flow rate of 1.1 mL/min. The detection was performed by a diode array detector (DAD) at 230 nm with total run time of 30min. 2 Calibration curves were linear with correlation coefficients of determinationr ( ) > 0.999. Limit of detection (LOD) and Limit of quantification (LOQ) ranged from 0.04 to 0.97 g/mL and from 0.64 to 3.24 g/mL, respectively. -

MSM Cross Reference Antihistamine Decongestant 20100701 Final Posted
MISSISSIPPI DIVISION OF MEDICAID Antihistamine/Decongestant Product and Active Ingredient Cross-Reference List The agents listed below are the antihistamine/decongestant drug products listed in the Mississippi Medicaid Preferred Drug List (PDL). This is a cross-reference between the drug product name and its active ingredients to reference the antihistamine/decongestant portion of the PDL. For more information concerning the PDL, including non- preferred agents, the OTC formulary, and other specifics, please visit our website at www.medicaid.ms.gov. List Effective 07/16/10 Therapeutic Class Active Ingredients Preferred Non-Preferred ANTIHISTAMINES - 1ST GENERATION BROMPHENIRAMINE MALEATE BPM BROMAX BROMPHENIRAMINE MALEATE J-TAN PD BROMSPIRO LODRANE 24 LOHIST 12HR VAZOL BROMPHENIRAMINE TANNATE BROMPHENIRAMINE TANNATE J-TAN P-TEX BROMPHENIRAMINE/DIPHENHYDRAM ALA-HIST CARBINOXAMINE MALEATE CARBINOXAMINE MALEATE PALGIC CHLORPHENIRAMINE MALEATE CHLORPHENIRAMINE MALEATE CPM 12 CHLORPHENIRAMINE TANNATE ED CHLORPED ED-CHLOR-TAN MYCI CHLOR-TAN MYCI CHLORPED PEDIAPHYL TANAHIST-PD CLEMASTINE FUMARATE CLEMASTINE FUMARATE CYPROHEPTADINE HCL CYPROHEPTADINE HCL DEXCHLORPHENIRAMINE MALEATE DEXCHLORPHENIRAMINE MALEATE DIPHENHYDRAMINE HCL ALLERGY MEDICINE ALLERGY RELIEF BANOPHEN BENADRYL BENADRYL ALLERGY CHILDREN'S ALLERGY CHILDREN'S COLD & ALLERGY COMPLETE ALLERGY DIPHEDRYL DIPHENDRYL DIPHENHIST DIPHENHYDRAMINE HCL DYTUSS GENAHIST HYDRAMINE MEDI-PHEDRYL PHARBEDRYL Q-DRYL QUENALIN SILADRYL SILPHEN DIPHENHYDRAMINE TANNATE DIPHENMAX DOXYLAMINE SUCCINATE -

2-Bromopyridine Safety Data Sheet Jubilant Ingrevia Limited
2-Bromopyridine Safety Data Sheet According to the federal final rule of hazard communication revised on 2012 (HazCom 2012) Date of Compilation : July 03 ’ 2019 Date of Revision : February 09 ’ 2021 Revision due date : January 2024 Revision Number : 01 Version Name : 0034Gj Ghs01 Div.3 sds 2-Bromopyridine Supersedes date : July 03 ’ 2019 Supersedes version : 0034Gj Ghs00 Div.3 sds 2-Bromopyridine Jubilant Ingrevia Limited Page 1 of 9 2-Bromopyridine Safety Data Sheet According to the federal final rule of hazard communication revised on 2012 (HazCom 2012) SECTION 1: IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING 1.1. Product identifier PRODUCT NAME : 2-Bromopyridine CAS RN : 109-04-6 EC# : 203-641-6 SYNONYMS : 2-Pyridyl bromide, Pyridine, 2-bromo-, beta-Bromopyridine, o-Bromopyridine SYSTEMATIC NAME : 2-Bromopyridine, -Pyridine, 2-bromo- MOLECULAR FORMULA : C5H4BrN STRUCTURAL FORMULA N Br 1.2. Relevant identified uses of the substance or mixture and uses advised against 1.2.1. Relevant identified uses 2-Bromopyridine is used as an intermediate in the pharmaceutical industry for the manufacture of Atazanavir (an antiretroviral drug), Carbinoxamine, Chloropyramine, triprolidine (antihistamine drugs), Disopyramide Phosphate (an antiarrythmic drug), Mefloquine (antimalarial drug), Pipradrol (mild CNS stimulant) etc. Uses advised against: None 1.3. Details of the supplier of the safety data sheet Jubilant Ingrevia Limited REGISTERED & FACTORY OFFICE: Jubilant Ingrevia Limited Bhartiagram, Gajraula , District: Amroha, Uttar Pradesh-244223, India PHONE NO: +91-5924-252353 to 252360 Contact Department-Safety: Ext. 7424 , FAX NO : +91-5924-252352 HEAD OFFICE: Jubilant Ingrevia Limited, Plot 1-A, Sector 16-A,Institutional Area, Noida, Uttar Pradesh, 201301 - India T +91-120-4361000 - F +91-120-4234881 / 84 / 85 / 87 / 95 / 96 [email protected] -www.jubilantingrevia.com 1.4. -

Table 6.12: Deaths from Poisoning, by Sex and Cause, Scotland, 2016
Table 6.12: Deaths from poisoning, by sex and cause, Scotland, 2016 ICD code(s), cause of death and substance(s) 1 Both Males Females ALL DEATHS FROM POISONING 2 1130 766 364 ACCIDENTS 850 607 243 X40 - X49 Accidental poisoning by and exposure to … X40 - Nonopioid analgesics, antipyretics and antirheumatics Paracetamol 2 1 1 Paracetamol, Cocaine, Amphetamine || 1 0 1 X41 - Antiepileptic, sedative-hypnotic, antiparkinsonism and psychotropic drugs, not elsewhere classified Alprazolam, MDMA, Cocaine || Cannabis, Alcohol 1 1 0 Alprazolam, Methadone || Pregabalin, Tramadol, Gabapentin, Cannabis 1 1 0 Alprazolam, Morphine, Heroin, Dihydrocodeine, Buprenorphine || Alcohol 1 1 0 Alprazolam, Oxycodone, Alcohol || Paracetamol 1 0 1 Amitriptyline, Cocaine, Etizolam || Paracetamol, Codeine, Hydrocodone, Alcohol 1 1 0 Amitriptyline, Dihydrocodeine || Diazepam, Paracetamol, Verapamil, Alcohol 1 1 0 Amitriptyline, Fluoxetine, Alcohol 1 0 1 Amitriptyline, Methadone, Diazepam || 1 1 0 Amitriptyline, Methadone, Morphine, Etizolam || Gabapentin, Cannabis, Alcohol 1 1 0 Amitriptyline, Venlafaxine 1 0 1 Amphetamine 1 1 0 Amphetamine || 1 1 0 Amphetamine || Alcohol 1 1 0 Amphetamine || Chlorpromazine 1 1 0 Amphetamine || Fluoxetine 1 1 0 Amphetamine, Dihydrocodeine, Alcohol || Procyclidine, Tramadol, Duloxetine, Haloperidol 1 0 1 Amphetamine, MDMA || Diclazepam, Cannabis, Alcohol 1 1 0 Amphetamine, Methadone || 1 1 0 Amphetamine, Oxycodone, Gabapentin, Zopiclone, Diazepam || Paracetamol, Alcohol 1 0 1 Amphetamine, Tramadol || Mirtazapine, Alcohol 1 1 0 Benzodiazepine -

Medication Instructions for Allergy Patients
MEDICATION INSTRUCTIONS FOR ALLERGY PATIENTS Drugs which contain antihistamine or have antihistaminic effects can result in negative reactions to skin testing. As a result, it may not be possible to properly interpret skin test results, and testing may have to be repeated at a later date. While this list is extensive, it is NOT all inclusive (particularly of the various brand names). Discontinue ALL antihistamines including the following medications seven (7) days prior to skin testing (unless longer time specified): Antihistamines – Generic name (Brand name(s)): Cetirizine (Zyrtec, Zyrtec-D) Hydroxyzine (Vistaril, Atarax) Desloratadine (Clarinex) Levocetirizine (Xyzal) Fexofenadine (Allegra, Allegra-D) Loratadine (Claritin, Claritin-D, Alavert) Diphenhydramine (Aleve PM, Benadryl, Bayer P.M., Benylin, Contac P.M., Doans P.M, Excedrin PM, Legatrin P.M.. Nytol, Tylenol Nighttime, Unisom, Zzzquil) Chlorpheniramine (Aller-Chlor, Allerest, Alka Seltzer Plus, Chlor-Trimeton, Comtrex, Contac, Co-Pyronil, Coricidin, CTM, Deconamine, Dristan, Dura-tap, Naldecon, Ornade Spansules, Rondec, Sinutab, Teldrin, Triaminic, Triaminicin, Tylenol Allergy) Azatadine (Optimine, Trinalin) Doxylamine (Nyquil) Brompheniramine (Bromfed, Dimetane, Dimetapp) Meclizine (Antivert) Carbinoxamine (Clistin, Rondec) Pheniramine Clemastine (Tavist) Phenyltoloxamine (Nadecon) Cyclizine (Marezine) Promethazine (Phenergan) Cyprohepatidine (Periactin) (9 days) Pyrilamine (Mepyramine) Dexbrompheniramine (Drixoral) Quinacrine (Atabrine) Dexchlorpheniramine (Extendryl, Polaramine) -

Marketing Authorisations Granted in December 2020
Marketing authorisations granted in December 2020 PL Number Grant Date MA Holder Licensed Name(s) Active Ingredient Quantity Units Legal Status Territory PL 14251/0100 01/12/2020 MANX HEALTHCARE LIMITED COLCHICINE 500 MICROGRAMS TABLETS COLCHICINE 0.500 MILLIGRAMS POM UK PL 34424/0050 02/12/2020 KEY PHARMACEUTICALS LIMITED SPIRONOLACTONE 25MG FILM-COATED TABLETS SPIRONOLACTONE 25 MILLIGRAMS POM UK PL 34424/0051 02/12/2020 KEY PHARMACEUTICALS LIMITED SPIRONOLACTONE 50MG FILM-COATED TABLETS SPIRONOLACTONE 50 MILLIGRAMS POM UK PL 34424/0052 02/12/2020 KEY PHARMACEUTICALS LIMITED SPIRONOLACTONE 100MG FILM-COATED TABLETS SPIRONOLACTONE 100 MILLIGRAMS POM UK PL 36282/0021 03/12/2020 RIA GENERICS LIMITED COLCHICINE 500 MICROGRAM TABLETS COLCHICINE 500 MICROGRAMS POM UK PL 39352/0439 03/12/2020 KOSEI PHARMA UK LIMITED FROVATRIPTAN 2.5 MG FILM-COATED TABLETS FROVATRIPTAN SUCCINATE MONOHYDRATE 2.5 MILLIGRAMS POM UK PL 31750/0174 04/12/2020 SUN PHARMACEUTICAL INDUSTRIES EUROPE BV CETRORELIX SUN 0.25 MG SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE CETRORELIX ACETATE 0.25 MILLIGRAMS PER MILLILITRE POM UK PL 34424/0054 04/12/2020 KEY PHARMACEUTICALS LIMITED ALIMEMAZINE TARTRATE 10MG FILM COATED TABLETS ALIMEMAZINE TARTRATE 10.00 MILLIGRAMS POM UK PL 17780/0858 07/12/2020 ZENTIVA PHARMA UK LIMITED FINGOLIMOD ZENTIVA 0.5 MG HARD CAPSULES FINGOLIMOD HYDROCHLORIDE 0.56 MILLIGRAMS POM UK PL 01502/0113 08/12/2020 HAMELN PHARMA LTD AMIODARONE HYDROCHLORIDE 20 MG/ML SOLUTION FOR INFUSION AMIODARONE HYDROCHLORIDE 20 MILLIGRAMS POM UK PL 16786/0006 08/12/2020 -

Torolac Combined with Oxycodone in Patient- Controlled Analgesia After Gynecol
Int. J. Med. Sci. 2015, Vol. 12 644 Ivyspring International Publisher International Journal of Medical Sciences 2015; 12(8): 644-649. doi: 10.7150/ijms.11828 Research Paper A Randomized Clinical Trial of Nefopam versus Ke- torolac Combined With Oxycodone in Patient- Controlled Analgesia after Gynecologic Surgery Boo-Young Hwang, Jae-Young Kwon, Do-Won Lee, Eunsoo Kim, Tae-Kyun Kim, Hae-Kyu Kim Department of Anesthesia and Pain Medicine, Biomedical Research Institute, Pusan National University Hospital, Busan, Republic of Korea Corresponding author: Jae-Young Kwon, Department of Anesthesia and Pain Medicine, Biomedical Research Institute, Pusan National University Hospital, 179 Gudeok-ro, Seo-gu, Busan 602-793, Republic of Korea. Tel: +82-51-240-7399; Fax: +82-51-242-7466; E-mail: [email protected] © 2015 Ivyspring International Publisher. Reproduction is permitted for personal, noncommercial use, provided that the article is in whole, unmodified, and properly cited. See http://ivyspring.com/terms for terms and conditions. Received: 2015.02.09; Accepted: 2015.07.18; Published: 2015.07.30 Abstract Objectives: Nefopam is a centrally-acting non-opioid analgesic, which has no effect on bleeding time and platelet aggregation. There has been no study about nefopam and oxycodone combina- tion for postoperative analgesia. In this study, we present efficacy and side effects of nefo- pam/oxycodone compared with ketorolac/oxycodone in patient-controlled analgesia (PCA) after gynecologic surgery. Methods: 120 patients undergoing gynecologic surgery were divided randomly into two groups: Nefopam group treated with oxycodone 1 mg and nefopam 1 mg bolus; and Ketorolac group treated with oxycodone 1 mg and ketorolac 1.5 mg bolus. -

ACUPAN Tablets
Consumer Medicine Information ACUPAN™ Nefopam hydrochloride 30 mg tablets What is in this leaflet This leaflet answers some common questions about ACUPAN tablets. However, it does not contain all the available information. It does not take the place of talking to your doctor or pharmacist. If you have any questions about ACUPAN that are not answered by this leaflet, ask your doctor or pharmacist. All medicines have risks and benefits. Your doctor has weighed the risks of you taking ACUPAN against the benefits before prescribing it to you. If you are worried about taking this medicine, ask your doctor or pharmacist. Keep this leaflet with your medicine as you may want to read it again. What ACUPAN is used for The name of your medicine is ACUPAN. ACUPAN belongs to a group of medicines called analgesics, commonly known as pain killers or pain relievers. It contains nefopam hydrochloride as the active ingredient. Nefopam hydrochloride interrupts the pain messages being sent to your brain, and it also acts in your brain to stop pain messages being felt. This means that ACUPAN does not stop the pain from happening, but you will not be able to feel the pain as much. ACUPAN is used to relieve both short term pain and long lasting pain (for example pain after an operation, dental pain, joint or muscle pain or after an injury). ACUPAN should not be used to treat the pain from a heart attack. Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed ACUPAN for another purpose. -

Antihistamine Therapy in Allergic Rhinitis
CLINICAL REVIEW Antihistamine Therapy in Allergic Rhinitis Paul R. Tarnasky, MD, and Paul P. Van Arsdel, Jr, MD Seattle, Washington Allergic rhinitis is a common disorder that is associated with a high incidence of mor bidity and considerable costs. The symptoms of allergic rhinitis are primarily depen dent upon the tissue effects of histamine. Antihistamines are the mainstay of therapy for allergic rhinitis. Recently, a second generation of antihistamines has become available. These agents lack the adverse effect of sedation, which is commonly associated with older antihistamines. Current practice of antihistamine therapy in allergic rhinitis often involves random selection among the various agents. Based upon the available clinical trials, chlorpheniramine appears to be the most reasonable initial antihistaminic agent. A nonsedating antihis tamine should be used initially if a patient is involved in activities where drowsiness is dangerous. In this comprehensive review of allergic rhinitis and its treatment, the cur rent as well as future options in antihistamine pharmacotherapy are emphasized. J Fam Pract 1990; 30:71-80. llergic rhinitis is a common condition afflicting some defined by the period of exposure to those agents to which A where between 15 and 30 million people in the United a patient is sensitive. Allergens in seasonal allergic rhinitis States.1-3 The prevalence of disease among adolescents is consist of pollens from nonflowering plants such as trees, estimated to be 20% to 30%. Two thirds of the adult grasses, and weeds. These pollens generally create symp allergic rhinitis patients are under 30 years of age.4-6 Con toms in early spring, late spring through early summer, sequently, considerable costs are incurred in days lost and fall, respectively. -

Trends in Prescription Medication Use Among Children and Adolescents— United States, 1999-2014
Supplementary Online Content Hales CM, Kit BK, Gu Q, Ogden CL. Trends in prescription medication use among children and adolescents— United States, 1999-2014. JAMA. doi:10.1001/jama.2018.5690 eTable. Classification of Prescription Medications Reported by NHANES Participants Aged 0-19 Years From 1999- 2000 to 2013-2014 by Therapeutic Class This supplementary material was provided by the authors to give readers additional informtion about their work. © 2018 American Medical Association. All rights reserved. Downloaded From: https://jamanetwork.com/ on 09/27/2021 eTable. Classification of Prescription Medications Reported by NHANES Participants Aged 0-19 Years From 1999- 2000 to 2013-2014 by Therapeutic Class Therapeutic classes are based on the Lexicon Plus prescription medication database and only those classes reported in the manuscript are listed. ADHD Medications Antiadrenergic Agents, Centrally Acting Clonidine Guanfacine CNS Stimulants Amphetamines Amphetamine Amphetamine; Dextroamphetamine Dextroamphetamine Lisdexamfetamine Methylphenidate or Dexmethylphenidate Dexmethylphenidate Methylphenidate Other CNS Stimulant Pemoline Selective Norepinephrine Reuptake Inhibitor Atomoxetine Antibiotics Cephalosporins Cefadroxil Cephalexin Cefaclor Cefprozil Cefuroxime Loracarbef Cefdinir Cefditoren Cefixime Cefpodoxime Ceftibuten Ceftriaxone Glycopeptide Antibiotics Vancomycin H. Pylori Eradication Agents Amoxicillin; Clarithromycin; Lansoprazole Lincomycin Derivatives Clindamycin Macrolide Derivatives Telithromycin Azithromycin Clarithromycin -

Medication Guide for a Safe Recovery
Medication Guide For A Safe Recovery A guide to maintaining sobriety while receiving treatment for other health problems. Revision 1.0 -April 2008 Table of Contents Introduction..................................................................................2 How to Use this Guide..................................................................3 Class A Drugs (Absolutely Avoid)................................................4 Class B Drugs................................................................................8 (With Addiction Medicine Specialist/Doctor Approval Only) Class C Drugs (Generally Safe to Take).....................................12 Alcohol-Free Products..................................................................16 Incidental Exposure Index...........................................................22 www.talbottcampus.com Introduction From the Talbott Recovery Campus Welcome to the Talbott Recovery Campus guide for a safe and sustained recovery. This document was developed through a collaborative effort between some of the best minds in addiction care today and will help you make wise decisions, ensuring that medications you may be prescribed and incidental exposure to alcohol do not threaten your hard won recovery. This guide is divided into three sections and is based on the drug classification system developed nearly 20 years ago by Dr. Paul Earley and recently expanded on by Bruce Merkin, M.D., Renee Enstrom, Nicholas Link and the staff at Glenbeigh hospital. Part one provides a way of categorizing medications