Clomipramine 25 Mg Capsules, Hard
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-
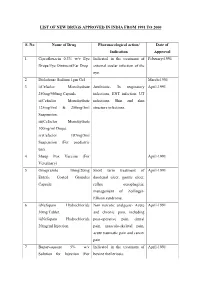
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

Optum Essential Health Benefits Enhanced Formulary PDL January
PENICILLINS ketorolac tromethamineQL GENERIC mefenamic acid amoxicillin/clavulanate potassium nabumetone amoxicillin/clavulanate potassium ER naproxen January 2016 ampicillin naproxen sodium ampicillin sodium naproxen sodium CR ESSENTIAL HEALTH BENEFITS ampicillin-sulbactam naproxen sodium ER ENHANCED PREFERRED DRUG LIST nafcillin sodium naproxen DR The Optum Preferred Drug List is a guide identifying oxacillin sodium oxaprozin preferred brand-name medicines within select penicillin G potassium piroxicam therapeutic categories. The Preferred Drug List may piperacillin sodium/ tazobactam sulindac not include all drugs covered by your prescription sodium tolmetin sodium drug benefit. Generic medicines are available within many of the therapeutic categories listed, in addition piperacillin sodium/tazobactam Fenoprofen Calcium sodium to categories not listed, and should be considered Meclofenamate Sodium piperacillin/tazobactam as the first line of prescribing. Tolmetin Sodium Amoxicillin/Clavulanate Potassium LOW COST GENERIC PREFERRED For benefit coverage or restrictions please check indomethacin your benefit plan document(s). This listing is revised Augmentin meloxicam periodically as new drugs and new prescribing LOW COST GENERIC naproxen kit information becomes available. It is recommended amoxicillin that you bring this list of medications when you or a dicloxacillin sodium CARDIOVASCULAR covered family member sees a physician or other penicillin v potassium ACE-INHIBITORS healthcare provider. GENERIC QUINOLONES captopril ANTI-INFECTIVES -

Research Article RP-HPLC Method for Determination of Several Nsaids and Their Combination Drugs
Hindawi Publishing Corporation Chromatography Research International Volume 2013, Article ID 242868, 13 pages http://dx.doi.org/10.1155/2013/242868 Research Article RP-HPLC Method for Determination of Several NSAIDs and Their Combination Drugs Prinesh N. Patel, Gananadhamu Samanthula, Vishalkumar Shrigod, Sudipkumar C. Modh, and Jainishkumar R. Chaudhari Department of Pharmaceutical Analysis, National Institute of Pharmaceutical Education and Research (NIPER), Balanagar, Hyderabad, Andhra Pradesh 500037, India Correspondence should be addressed to Gananadhamu Samanthula; [email protected] Received 29 June 2013; Accepted 13 October 2013 Academic Editor: Andrew Shalliker Copyright © 2013 Prinesh N. Patel et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. An RP-HPLC method for simultaneous determination of 9 NSAIDs (paracetamol, salicylic acid, ibuprofen, naproxen, aceclofenac, diclofenac, ketorolac, etoricoxib, and aspirin) and their commonly prescribed combination drugs (thiocolchicoside, moxifloxacin, clopidogrel, chlorpheniramine maleate, dextromethorphan, and domperidone) was established. The separation was performed on ∘ Kromasil C18 (250 × 4.6 mm, 5 m) at 35 C using 15 mM phosphate buffer pH 3.25 and acetonitrile with gradient elution ata flow rate of 1.1 mL/min. The detection was performed by a diode array detector (DAD) at 230 nm with total run time of 30min. 2 Calibration curves were linear with correlation coefficients of determinationr ( ) > 0.999. Limit of detection (LOD) and Limit of quantification (LOQ) ranged from 0.04 to 0.97 g/mL and from 0.64 to 3.24 g/mL, respectively. -

Management of Chronic Problems
MANAGEMENT OF CHRONIC PROBLEMS INTERACTIONS BETWEEN ALCOHOL AND DRUGS A. Leary,* T. MacDonald† SUMMARY concerned. Alcohol may alter the effects of the drug; drug In western society alcohol consumption is common as is may change the effects of alcohol; or both may occur. the use of therapeutic drugs. It is not surprising therefore The interaction between alcohol and drug may be that concomitant use of these should occur frequently. The pharmacokinetic, with altered absorption, metabolism or consequences of this combination vary with the dose of elimination of the drug, alcohol or both.2 Alcohol may drug, the amount of alcohol taken, the mode of affect drug pharmacokinetics by altering gastric emptying administration and the pharmacological effects of the drug or liver metabolism. Drugs may affect alcohol kinetics by concerned. Interactions may be pharmacokinetic or altering gastric emptying or inhibiting gastric alcohol pharmacodynamic, and while coincidental use of alcohol dehydrogenase (ADH).3 This may lead to altered tissue may affect the metabolism or action of a drug, a drug may concentrations of one or both agents, with resultant toxicity. equally affect the metabolism or action of alcohol. Alcohol- The results of concomitant use may also be principally drug interactions may differ with acute and chronic alcohol pharmacodynamic, with combined alcohol and drug effects ingestion, particularly where toxicity is due to a metabolite occurring at the receptor level without important changes rather than the parent drug. There is both inter- and intra- in plasma concentration of either. Some interactions have individual variation in the response to concomitant drug both kinetic and dynamic components and, where this is and alcohol use. -

Clomipramine | Memorial Sloan Kettering Cancer Center
PATIENT & CAREGIVER EDUCATION Clomipramine This information from Lexicomp® explains what you need to know about this medication, including what it’s used for, how to take it, its side effects, and when to call your healthcare provider. Brand Names: US Anafranil Brand Names: Canada Anafranil; MED ClomiPRAMINE; TARO-Clomipramine Warning Drugs like this one have raised the chance of suicidal thoughts or actions in children and young adults. The risk may be greater in people who have had these thoughts or actions in the past. All people who take this drug need to be watched closely. Call the doctor right away if signs like low mood (depression), nervousness, restlessness, grouchiness, panic attacks, or changes in mood or actions are new or worse. Call the doctor right away if any thoughts or actions of suicide occur. This drug is not approved for use in all children. Talk with the doctor to be sure that this drug is right for your child. What is this drug used for? It is used to treat obsessive-compulsive problems. It may be given to you for other reasons. Talk with the doctor. Clomipramine 1/8 What do I need to tell my doctor BEFORE I take this drug? If you have an allergy to clomipramine or any other part of this drug. If you are allergic to this drug; any part of this drug; or any other drugs, foods, or substances. Tell your doctor about the allergy and what signs you had. If you have had a recent heart attack. If you are taking any of these drugs: Linezolid or methylene blue. -

Supplementary Materials
Supplementary Materials Table S1. The significant drug pairs in potential DDIs examined by the two databases. Micromedex Drugs.com List of drugs paired PK-PD Mechanism details 1. Amiodarone— PD Additive QT-interval prolongation Dronedarone 2. Amiodarone— PK CYP3A inhibition by Ketoconazole Ketoconazole 3. Ciprofloxacin— PD Additive QT-interval prolongation Dronedarone 4. Cyclosporine— PK CYP3A inhibition by Cyclosporine Dronedarone 5. Dronedarone— PK CYP3A inhibition by Erythromycin Erythromycin 6. Dronedarone— PD Additive QT-interval prolongation Flecainide 7. Dronedarone— PK CYP3A4 inhibition by Itraconazole Itraconazole 8. Dronedarone— PK Contraindication Major CYP3A inhibition by Ketoconazole Ketoconazole 9. Dronedarone— PD Additive QT-interval prolongation Procainamide PD 10. Dronedarone—Sotalol Additive QT-interval prolongation 11. Felodipine— PK CYP3A inhibition by Itraconazole Itraconazole 12. Felodipine— PK CYP3A inhibition by Ketoconazole Ketoconazole 13. Itraconazole— PK CYP3A inhibition by Itraconazole Nisoldipine 14. Ketoconazole— PK CYP3A inhibition by Ketoconazole Nisoldipine 15. Praziquantel— PK CYP induction by Rifampin Rifampin PD 1. Amikacin—Furosemide Additive or synergistic toxicity 2. Aminophylline— Decreased clearance of PK Ciprofloxacin Theophylline by Ciprofloxacin 3. Aminophylline— PK Decreased hepatic metabolism Mexiletine 4. Amiodarone— PD Additive effects on QT interval Ciprofloxacin 5. Amiodarone—Digoxin PK P-glycoprotein inhibition by Amiodarone 6. Amiodarone— PD, PK Major Major Additive effects on QT Erythromycin prolongation, CYP3A inhibition by Erythromycin 7. Amiodarone— PD, PK Flecainide Antiarrhythmic inhibition by Amiodarone, CYP2D inhibition by Amiodarone 8. Amiodarone— PK CYP3A inhibition by Itraconazole Itraconazole 9. Amiodarone— PD Antiarrhythmic inhibition by Procainamide Amiodarone 10. Amiodarone— PK CYP induction by Rifampin Rifampin PD Additive effects on refractory 11. Amiodarone—Sotalol potential 12. Amiodarone— PK CYP3A inhibition by Verapamil Verapamil 13. -

FDA Approved Drugs with Broad Anti-Coronaviral Activity Inhibit SARS-Cov-2 in Vitro
bioRxiv preprint doi: https://doi.org/10.1101/2020.03.25.008482; this version posted March 27, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. FDA approved drugs with broad anti-coronaviral activity inhibit SARS-CoV-2 in vitro Stuart Weston1, Rob Haupt1, James Logue1, Krystal Matthews1 and Matthew B. Frieman*1 1 - Department of Microbiology and Immunology, University of Maryland School of Medicine, 685 W. Baltimore St., Room 380, Baltimore, MD, 21201, USA *Corresponding author. Email: [email protected] Key words: SARS-CoV-2, nCoV-2019, COVID-19, drug repurposing, FDA approved drugs, antiviral therapeutics, pandemic, chloroquine, hydroxychloroquine AbstraCt SARS-CoV-2 emerged in China at the end of 2019 and has rapidly become a pandemic with over 400,000 recorded COVID-19 cases and greater than 19,000 recorded deaths by March 24th, 2020 (www.WHO.org) (1). There are no FDA approved antivirals or vaccines for any coronavirus, including SARS-CoV-2 (2). Current treatments for COVID-19 are limited to supportive therapies and off-label use of FDA approved drugs (3). Rapid development and human testing of potential antivirals is greatly needed. A potentially quicker way to test compounds with antiviral activity is through drug re-purposing (2, 4). Numerous drugs are already approved for use in humans and subsequently there is a good understanding of their safety profiles and potential side effects, making them easier to test in COVID-19 patients. -

Management of Major Depressive Disorder Clinical Practice Guidelines May 2014
Federal Bureau of Prisons Management of Major Depressive Disorder Clinical Practice Guidelines May 2014 Table of Contents 1. Purpose ............................................................................................................................................. 1 2. Introduction ...................................................................................................................................... 1 Natural History ................................................................................................................................. 2 Special Considerations ...................................................................................................................... 2 3. Screening ........................................................................................................................................... 3 Screening Questions .......................................................................................................................... 3 Further Screening Methods................................................................................................................ 4 4. Diagnosis ........................................................................................................................................... 4 Depression: Three Levels of Severity ............................................................................................... 4 Clinical Interview and Documentation of Risk Assessment............................................................... -
667783-Atenolol (ACC-UK)06-16-SPC-PIL
Artwork No. - Colours Used Customer Accord Pantone Black Description Atenolol Market UK Language English Size 170 x 550 mm PIL Min. Font Size 9.5 Version No. 13 (Page 1 of 2) (06-16-Atenolol SPC) Date 07_05_16 (Atenolol (ACC-UK)06-16-SPC-PIL) Prepared By Checked By Approved By Regulatory Affairs Regulatory Affairs Quality Assurance You must consult your doctor if you are If you forget to take Atenolol Tablets 6. Contents of the pack and other planning to combine Atenolol tablets with the If you forget to take a dose of the Atenolol information medicines listed below: Tablet, take it as soon as you remember, PACKAGE LEAFLET: INFORMATION FOR • Clonidine (for high blood pressure or unless it is time for your next dose. What Atenolol Tablet contains THE USER migraine). If you are taking clonidine and Do not take a double dose to make up for a • The active ingredient is Atenolol. Atenolol 25 mg Tablets Atenolol tablets together, do not stop taking forgotten dose. • The other ingredients are Heavy clonidine unless your doctor tells you to do Magnesium Carbonate, Maize Starch, Atenolol 50 mg Tablets so. If you have to stop taking clonidine, your If you stop taking Atenolol Tablets Sodium Lauryl Sulphate, Gelatin, doctor will give you careful instructions Do not stop taking Atenolol Tablets without Magnesium Stearate, Microcrystalline Atenolol 100 mg Tablets about how to do it. talking to your doctor. In some cases, you may Cellulose and Purified Talc. (Atenolol) • Verapamil, diltiazem and nifedipine (for high need to stop taking it gradually over 7-14 days blood pressure or chest pain). -

Prescribing Information for High-Dose Fexofenadine in the Management of Urticaria in Adults
1 Prescribing information for high-dose fexofenadine in the management of urticaria in adults This prescribing information document outlines the prescribing responsibilities between the specialist and GP. GPs are invited to participate. If the GP feels that such prescribing is outside their area of expertise or have clinical concerns about the safe management of the drug in primary care, then he or she is under no obligation to do so. In such an event, clinical responsibility for the patient’s health remains with the specialist. If a specialist asks the GP to prescribe, the GP should reply to this request as soon as practicable. Consultant details GP details Patient details Name: Name: Name: Address: Address: NHS Number: Email: Email: Date of birth: Contact number: Contact number: Contact: Introduction Fexofenadine is a second generation non-sedating H1-antihistamine used in the treatment of allergic disorders. Fexofenadine is a pharmacologically active metabolite of terfenadine. Licensed indication: In adults and children 12 years and older, the licensed dose is 180mg daily for the relief of symptoms associated with chronic idiopathic urticaria. Unlicensed indication (the focus of this document): As advised by Europeani and British Association of Dermatologistsii guidelines, doses of antihistamines up to four times the recommended daily dose may be used in the treatment of urticaria. Adult dosage and administration The licensed maximum recommended daily dose is 180mg once daily taken before a meal. Doses of up to four times the daily recommended dose have been used in the treatment of severe urticaria (unlicensed indication)i Available as: 30mg, 120mg and 180mg tablets Specialist responsibilities Provide patient/carer with relevant written information on the unlicensed use of high-dose fexofenadine and possible side-effects. -

Profile Profile Profile Profile Uses and Administration Adverse Effects And
640 Antihistamines Propiomazine Hydrochloride {BANM, r!NNMJ P..r�p�rc:Jti()n.� ............................ m (details are given in Volume B) propiorf,azina; to o azine; Chiorhydrate ProprietaryPreparations HidrodoruroPropi<;>rn deazini HydrochloridP pium; f1ponvtot,la3Y!Ha China: Qi Qi Hung. : de; Single-ingredient Preparafions. Loderixt. CIH'f); rMAPOX!10p!'\A. C;cJcl1_,--N_,QS,HCI�3.76_91 Rupatadine is an antihistamine with platelet-activating CAS 240) 5-9. factor (PAF) antagonist activity that is used for the Terfenadine (BAN, USAN, r!NN) treatment of allergic rhinitis (p. 612.1) and chronic 1 idiopathic urticaria (p. 612.3). It is given as the fumarate although doses are expressed in terms of the base; rupatadine fumarate 12.8 mg is equivalent to about 10 mg of rupatadine. The usual oral dose is the equivalent of 10 mg once daily of rupatadine. Izquierdo I. et a!. Rupatadine: a new selective histamine HI receptor and platelet-activating factor (PAF) antagonist: a review of pharmacological profile and clinical management of allergic rhinitis. Drugs Today 2003; 39: 451-68. 2. Kearn SJ,Plosker GL. Rupatadine: a review of its use in the management of allergic disorders. Drugs 2007; 67: 457-74. 3. Fantin et at. International Rupatadine study group. A 12-week 5, 8: (Terfenadine). A white or almost white, placebo-controlled study of rupatadine 10 mg once daily compared with Ph. Bur. cetirizine IO mg once daily, in the treatment of persistent allergic crystalline powder. It shows polymorphism. Very slightly rhinitis. Allergy 2008; 63: 924-3 1. soluble in water and in dilute hydrochloric acid; freely 4. -

Data Sheet SURMONTIL Trimipramine (As Maleate) 25 Mg Tablets and 50 Mg Capsules
Data Sheet SURMONTIL trimipramine (as maleate) 25 mg tablets and 50 mg capsules Presentation SURMONTIL tablets are compression coated, white or cream, circular, biconvex, containing the equivalent of 25mg trimipramine (as maleate) with a diameter of about 8.0mm. The face is indented with the name and strength, reverse plain. SURMONTIL capsules are opaque white with opaque green cap, printed SU50, each containing the equivalent of 50mg trimipramine (as maleate). Uses Actions SURMONTIL has a potent anti-depressant action similar to that of other tricyclic anti-depressants. The mechanism of action is not fully understood but it is thought to be via inhibition of neuronal re- uptake of noradrenalin, thereby increasing availability. SURMONTIL also possesses a pronounced sedative action. Pharmacokinetics SURMONTIL is readily absorbed after oral administration, reaching a mean peak plasma level after 3 hours. High first pass hepatic clearance results in a mean bioavailability of about 41% of the oral dose, and trimipramine is extensively protein bound in plasma. Elimination half-life is 24 hours. Metabolism is in the liver to its major metabolite, desmethyltrimipramine, which is excreted mainly in the urine. Indications SURMONTIL is indicated in the treatment of depressive illness, especially where sleep disturbance, anxiety or agitation is a presenting symptom. Sleep disturbance is controlled within 24 hours and true anti-depressant action follows within 7-10 days. Dosage and Administration Adults Mild/Moderate Depression in General Practice: The recommended dosage is 50-75 mg orally given two hours before bedtime, the larger dose (75 mg) being preferable for those patients with more marked sleep disturbance.