Limited Benefit of Marine Protein Hydrolysate on Physical Function
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Metodisme I Bergen
Metodismen i Bergen 1879 –1914 Resultat av målretta, strategiske tiltak Eller svar på spørsmål i samtida? Thor Bernhard Tobiassen AVH502 – Masteravhandling (55ECTS) Master i teologi/metodisme Veileder: Professor Bengt T. Oftestad Det teologiske menighetsfakultet, Oslo våren Thor Bernhard Tobiassen Metodismen i Bergen 1879 - 1914 Resultatet av målretta strategiske tiltak eller svar på spørsmål i samtida? AVH502 – Masteravhandling (55ECTS) Master i teologi/metodisme Veileder: Professor Bengt T. Oftestad Det teologiske menighetsfakultet, Oslo våren 2008 Innhald I. Innleiing 5 A. Tema og grunngjeving for val av problemstilling 5 B. Problemformulering og avgrensing 5 C. Metode 6 D. kjelder 8 E. Anna forsking 9 F. Definisjonar 9 II. Bergen kring 1880 11 A. Kort demografi 11 1. Det bergenske folk 11 2. Yrke, klassedeling og busetjingsmønster 12 B. Straumdrag i norsk samtid 14 1. Demokratisering og modernisering. 14 2. Internasjonalisering 16 3. Pluralisme og voluntarisme 17 C. Den religiøse landskapen i Bergen kring 1878 18 1. Statskyrkja 18 2. Luthersk lekmannsarbeid og andre organisasjonar 19 3. Dissentarar 20 III. Metodismens ideologi, mål og strategiar 22 A. Metodistkirken i Norge fram til 1879 22 B. Mål og Ideologi 24 1. Mål 24 2. Ideologi 25 C. Metodismen til Bergen – medvite satsing? 26 1. Metodistkirken ville til Bergen 26 2. Planstyrte nyetableringar? 27 3. Det lokale grunnlaget 29 Familien Barratt 30 D. Kyrkja sin strategi 33 Lars Petersen 1854 - 1889 33 IV. Metodistar – Oppvekstvilkår, samfunnsklassar og rekruttering 35 A. Dei første metodistar – mobilitet? 36 1. By og land 36 2. Emigrasjon og anna flytting 37 3. Kyst og innland 39 Jens Johannessen (1855-1927) 40 B. -

Preliminary Programme
JUNE 14th - 17th, 2010 26th CIMAC World Congress on Combustion Engine Technology for Ship propulsion Power generation Rail traction PPRELIMINARYRELIMINARY PPROGRAMROGRAM www.cimac.com Bergen Tourist Board / Willy Haraldsen Board / Tourist Bergen CONTENTS 3 WELCOME ADDRESS 4 INTRODUCTION TO CIMAC 5 CONFERENCE VENUE 6 PRELIMINARY PROGRAMME 6 MONDAY, 14TH JUNE 2010 9 TUESDAY, 15TH JUNE 2010 14 WEDNESDAY, 16TH JUNE 2010 18 THURSDAY, 17TH JUNE 2010 23 EXHIBITION 25 OPTIONAL TOURS MONDAY 14TH JUNE 2010 26 OPTIONAL TOURS TUESDAY 15TH JUNE 2010 27 OPTIONAL TOURS WEDNESDAY 16TH JUNE 2010 28 OPTIONAL TOURS THURSDAY 17TH JUNE 2010 29 TECHNICAL VISITS FRIDAY 18TH JUNE 2010 30 OPTIONAL PRE AND POST CONGRESS TOURS 32 NORWAY 34 BERGEN 36 ACCOMMODATION 37 HOTEL OVERVIEW AND ROOM RATES 39 GETTING TO BERGEN 40 LOCAL BUSES IN BERGEN AND HORDALAND 41 GENERAL INFORMATION 43 REGISTRATION INFORMATION 46 MEMBERS OF CIMAC Come to beautiful Bergen. You’ll be most welcome. CIMAC National Member Association Norway, through CIMAC 2010 AS has the pleasure of organising the 26th CIMAC World Congress on Combustion Engines, scheduled for 14th – 17th June 2010 in Bergen/Norway. It is a special honour for us to have been awarded the next congress by CIMAC, an organization which has been acting for more than 50 years as a lively and attractive forum for engine and turbine builders and users. Today, CIMAC is the most important platform for the dialogue between the engine industry’s technical experts and its customers. The Congress is devoted to the presentation of papers in the fi elds of marine, power generation and locomotive engine engineering covering state-of-the-art technologies as well as the application of such engines. -
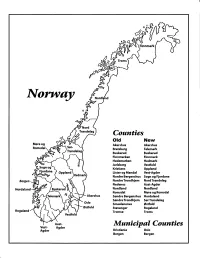
Norway Maps.Pdf
Finnmark lVorwny Trondelag Counties old New Akershus Akershus Bratsberg Telemark Buskerud Buskerud Finnmarken Finnmark Hedemarken Hedmark Jarlsberg Vestfold Kristians Oppland Oppland Lister og Mandal Vest-Agder Nordre Bergenshus Sogn og Fjordane NordreTrondhjem NordTrondelag Nedenes Aust-Agder Nordland Nordland Romsdal Mgre og Romsdal Akershus Sgndre Bergenshus Hordaland SsndreTrondhjem SorTrondelag Oslo Smaalenenes Ostfold Ostfold Stavanger Rogaland Rogaland Tromso Troms Vestfold Aust- Municipal Counties Vest- Agder Agder Kristiania Oslo Bergen Bergen A Feiring ((r Hurdal /\Langset /, \ Alc,ersltus Eidsvoll og Oslo Bjorke \ \\ r- -// Nannestad Heni ,Gi'erdrum Lilliestrom {", {udenes\ ,/\ Aurpkog )Y' ,\ I :' 'lv- '/t:ri \r*r/ t *) I ,I odfltisard l,t Enebakk Nordbv { Frog ) L-[--h il 6- As xrarctaa bak I { ':-\ I Vestby Hvitsten 'ca{a", 'l 4 ,- Holen :\saner Aust-Agder Valle 6rrl-1\ r--- Hylestad l- Austad 7/ Sandes - ,t'r ,'-' aa Gjovdal -.\. '\.-- ! Tovdal ,V-u-/ Vegarshei I *r""i'9^ _t Amli Risor -Ytre ,/ Ssndel Holt vtdestran \ -'ar^/Froland lveland ffi Bergen E- o;l'.t r 'aa*rrra- I t T ]***,,.\ I BYFJORDEN srl ffitt\ --- I 9r Mulen €'r A I t \ t Krohnengen Nordnest Fjellet \ XfC KORSKIRKEN t Nostet "r. I igvono i Leitet I Dokken DOMKIRKEN Dar;sird\ W \ - cyu8npris Lappen LAKSEVAG 'I Uran ,t' \ r-r -,4egry,*T-* \ ilJ]' *.,, Legdene ,rrf\t llruoAs \ o Kirstianborg ,'t? FYLLINGSDALEN {lil};h;h';ltft t)\l/ I t ,a o ff ui Mannasverkl , I t I t /_l-, Fjosanger I ,r-tJ 1r,7" N.fl.nd I r\a ,, , i, I, ,- Buslr,rrud I I N-(f i t\torbo \) l,/ Nes l-t' I J Viker -- l^ -- ---{a - tc')rt"- i Vtre Adal -o-r Uvdal ) Hgnefoss Y':TTS Tryistr-and Sigdal Veggli oJ Rollag ,y Lvnqdal J .--l/Tranbv *\, Frogn6r.tr Flesberg ; \. -

Puddefjorden Sandviken Mulen Skansemyren Nordnes Sandviken Møhlenpris Jekteviken Gyldenpris Laksevåg Fløyfjellet
200 . 175 Blaauwsv 150 Star Odont. Preklinisk Håmanen Inst. inst. vei Alrek Stoltzekleiven 125 gens W Ole Ir Br n Munkebotn Fløyfjellet Fløyen i e n eidabli l e F jellve n v e n e i i e l l . e d g e WC i e sv kk a S n gen FLØYFJELLSTUNNELEN Ka v e j al ALSVEIEN ba t n and Hoffm in mv m F d e kkv s viksl a Ka eien e b r kka ie ien n Øvr Christine- v e lv VED b e e n s r 320m 300 Ka u . Å r s e e rd le N b lv s ve l o am KAL s Sandviksfjellets l Sudmann i v Heien går u N G d je e l den M Mulen i befestningsanlegg i T h F Heien Beiteveien 585 Fløenbakken n i u p G lie Skram Ama s v Form Christinegård eistølen p ei n ei an Br e v e n i s v t s l ei Ballbane u Skansemyren en r M e a ø SAN A K Per ingene Idrettsplass Irg l f l l e n DV m se v ysv Fløibanen n a d IK a Uren ir nb Flø yre K Fløen a SV ke ute ak e em v re l EI li ga G nb ke kans Ø s E e t e i S le v eien N Lærerakademiet e rg n rdre O i e n e sv n Mulelvsm.. s e Hest e b e r gvei e n o l f a r l t i ik S P ve n N K a e e v Sandviksboder k ra i g n n d 585 ram i . -

Bergen-Map-2019.Pdf
Krokane 5 Florø Skei JOSTEDALSBREEN NIGARDS- Stavang t e BREEN Naustdal tn Jølsterva Askrova E39 Svanøybukt 611 5 55 Førde 604 609 Moskog 13 Gaupne Eikenes Fjærland en d Askvoll r Gaularfjellet o j Dale f Gjervik Viken a r Værlandet 55 t n s 13 e u d Hafslo 611 r L jo E39 f Bulandet s Fure d 607 57 Solvorn Ornes 79 Myking m Herdla Museum Westland Hotel Gjervik Tepstad Fjordslottet la 51 Hotel & Bad Haugstveit r Bidogen Abbedissen Brakstad Alver Hotel Hamre Sandal jæ Bruvoll Camping og Hytter THE OFFICIAL MAP 2019 F Sogndal Dale BLOMØYJ Herdla K L M Håland N Grønås Salbu Høyanger Dragsvik Fløksand MELAND KNARVIK Fugledale Kallekleiv A Hopland Bjørnestad Vadheim Hella Oksneset Ådlandsvik Fosse Bleikli Børtveit TOURIST INFORMATION Dale Flatøy Eikeland Gåsvær 5 Berland Mosevoll Nordeide Leikanger H Sagstad LONEVÅG REGION NORTH AND WEST Balestrand Mann- MAIN ATTRACTIONS Hjertås 564 Hordvikneset Osterøy Museum Fitje j Holme Nordhordlands- 55 Kaupanger heller U l v e s u n d e t Fauskanger HOLSNØY Angskår Greve Njåstad Måren ACTIVITIES / MUSEUMS / SIGHTS / VENUES Blomvågnes e Heggernes brua 67 Sula Krakhella E39 45 Alvøen Manor (L3) l Langeland STEINESTØ S Røskeland Låstad 55 Vangsnes t ø Hatland 46 Berg Fritid (J1) Tellevik r Kvammen Borge 606 Rysjedalsvika Fodnes e Fjordside f Ytrøy DEN 47 FREKHAUG j Burkeland Lone Lavik JOR Bergen Trotting Park (M1) f H Lodge o I EF J K j 64 N 48 Bjørn West Museum (J1) e r 567 OG Rong o Træet r d Dalstø Mjåtveit n d Autun 607 S Ortnevik 49 Damsgård Manor (M2) 562 e 36 Halland RONG SENTER r l SALHUS Hylkje e Revheim BERGEN Daløy Frønningen 50 e d Norsk n Lærdal Économusée Hillesvåg Ullvarefabrikk (J2) d f Trikotasjemuseum Falkanger Hagebø Rutledal Vik Kjerrgarden Hanevik r Hardbakke Håbakken51 Économusée Oleana (N1) e j Runnhovda KARTEN | PLAN | KART | PIANTINA | Finden Rongesund o o y Fløibanen n Annekset Veten 66 N Solberg e PHUS Oppedal 52 Fjell Fortress (I2) RONGØY r j Kleiveland S Bjordal A d a Vetrlidsalm. -

Programme 2017
WELCOME TO REGION BERGEN AND NORWEGIAN TRAVEL WORKSHOP BERGEN Norwegian Travel Workshop 2017 24-27 April PROGRAMME 2017 visitBergen.com PLAN & BOOK: visitBergen.com 3 INDEX Norwegian Travel Workshop 2017 2 WELCOME 4 Programme for Norwegian Travel Workshop 4 Saturday 22 April .................................................................................................................................................... 10:00 & 14.00 Fjord cruise Bergen – Mostraumen (3 hours) 4–5 Sunday 23 April ....................................................................................................................................................... 10:00 & 14.00 Fjord cruise Bergen – Mostraumen (3 hours) 18:00 – 23:00 Unique Seafood experience with a boat trip and dinner at Cornelius Seafood Restaurant Monday 24 April ...................................................................................................................................................... 10:00 – 16:00 Suppliers decorate stands at Grieghallen (Dovregubbens hall) 12:00 – 14:00 Bergen Panorama tour by bus 12:00 – 15:00 Bergen Coast Adventure – where the history of fi sheries comes alive 10:00 + 14:00 Fjord cruise Bergen – Mostraumen (3h) 13:30 – 16:00 Site inspection of the new hotels in Bergen city centre and by the airport 17:30 – 19:00 Seminar for suppliers at Grieghallen (Peer Gynt Salen) 6 17:45 – 19:00 Welcome Drink for buyers at KODE – Art Museums of Bergen 19:30 – 20:00 Opening Ceremony at Grieghallen 20:00 Welcome party at Grieghallen (foyer 2nd fl oor) Tuesday -

Kvh-Bergen-Strandsiden.Pdf
KULTURHISTORISK VEGBOK. RUTEDEL RV. 555 BERGEN TEMA: Bergen – bysamfunnet T66, Byen og strilelandet T71 KART: K436, K437 Strandsiden I løpet av 1300-tallet endret MUNKELIV STRANDSIDEN seg fra et landlig OG JONSKLOSTERET klosterlandskap til et pulserende handelssentrum med sjøboder, MUNKELIV kloster lå på Nordnes, på sjøhus og hoper. plas sen som nå heter KLOSTERET. Det var et benediktinerkloster som ble grunn - lagt av kong ØYSTEIN MAGNUSSON (1103-23). I 1426 ble klosteret overtatt av birgittinerne. I 1455 ble det herjet av tyskerne, som satte fyr på både kirken og klosterbyg ningene da de drepte OLAV NILSSON, den norske høvedsmannen på Bergenhus som hadde flyktet inn i kirken. «Muren» fra 1561, slik den ble seende ut Kirken ble gjen reist og fungerte i kort tid etter brannen i 1702. Xylografi ca. 1860. som byens dom kirke, før fransiskaner - kirken i Vågs botn ble domkirke i 1536. RIK ROSENKRANTZ arvet en gård Kirken ble anta kelig påbegynt allerede i på Stranden gjennom sitt slekts - Øystein Magnussons tid (før 1123) og må Eskap med Losneslekten. Etter en ha vært en av de stase ligste kirkebyg - brann i 1561 bygde han her en «grund- ninger i Bergen. muret Vaaning» tvers over Strandgaten, og han hentet byggestein fra det nær - JONSKLOSTERET lå ovenfor Strand - gaten, i området mellom Strandgaten og liggende Munkeliv kloster. «De Rosen- plas sen foran Tinghuset. Det var et augus- krantzers Mur» og «Rosenkrantz Almin- tiner kloster, an takelig grunnlagt i andre ding» er idag kjent som MUREN og MUR- halvdel av 1100-årene. Ingen rester av ALMENNING. klosterbyg nin gen er bevart (K436). -

Byrådet I Bergen Beboere I Strangehagen, Verftsbakken
Byrådet i Bergen Beboere i Strangehagen, Verftsbakken, Galgebakken og Verftstrappen Bergen, 20. august 2019 Vedrørende byrådssak: Bergenhus Gnr 165 Bnr 637 Strangehagen 13 Stans av planinitiativ Vi viser til byrådets møte torsdag 22. august og behandling av sak om bebyggelse av tomt i Strangehagen 13. Som nære naboer til eiendommen ønsker vi å komme med noen innspill før byrådets behandling. Bakgrunn for saken er Larsen og Solheim AS sitt ønske om å fremme et planforslag som legger til rette for bygging av 5 til 8 boenheter på tomten, samt etablering av et utsiktspunkt og ny snarvei fra Haugeveien ned til Strangehagen. Fagetatens behandling Plan- og Bygningsetaten har tidligere stoppet planinitiativet. I sitt brev fra 4. mars 2019 konkluderer de med at oppstart av planarbeid ikke er anbefalt, med følgende begrunnelse: - Området er regulert til spesialområde bevaring etter pbl § 25.6 på grunn av sin høye kulturhistoriske verdi. - Områdets høye antikvariske verdi gjelder både den historiske bebyggelsesstruktur, veifar og grøntstruktur. Det er viktig å se området som en helhet som har en historisk struktur/ kulturminnemiljø. - Gjeldene reguleringsplan åpner ikke opp for bygging på eiendommen. - Fortetting skal i hovedsak skje på de «grå» flatene ikke de «grønne» eiendommen er ikke et prioritert fortettingsområde. - Det opplyses også om at eiendommen er en festetomt og at Bergen kommune står som eier av tomten. I henhold til festeavtalen har ikke fester rett til ytterligere bygging uten tillatelse fra eier. Fester må ta kontakt med Etat for eiendom for nærmere avklaring. Etaten skriver videre om kulturmiljø: Hagen til Strangehagen 13 har etter de historiske kart vi har, ikke vært bebygget tidligere. -

Havet I Byen, Byen Fra Havet Foto: Vill Urbanisme Foto
HAVET I BYEN, BYEN FRA HAVET FOTO: VILL URBANISME FOTO: 1 INNHOLD FORORD - HAVET I BYEN, BYEN FRA HAVET 04-07 AKVARIET, EN VISJONÆR IDÉ TILPASSET DATIDEN 08-15 BERGEN ER VERDENS OG NORGES MARINE HOVEDSTAD 16-21 AKVARIET, EN BY- OG SAMFUNNSUTVIKLER 22-29 FOTO: FRANSISCO MUNOZ FOTO: EN AKSE FOR LÆRELYST 30-39 PROSESSEN FREM MOT ALTERNATIVENE 40-45 ANBEFALNINGEN: SAMLOKALISERING MED 46-59 HAVFORSKNINGSINSTITUTTET PÅ DOKKEN DE ALTERNATIVE KONSEPTENE 50-59 2 3 HAVET I BYEN, BYEN FRA HAVET Bergen ble født av havet, og lever ennå av havet. Nå foreslår Akvariet at Bergen skal få et hav, midt i byen. Bergen skal bli verdensledende på formidling og opplevelse av havet. Det nye Akvariet vil prege en hel ny bydel, og vi vil gi byutvikling av tidligere havnearealer en ny dimensjon: Bergen skal ikke bare åpnes mot havet, men gjennom det nye Akvariet vil havet flytte inn og bli en del av byen. Alle vet at havet er viktig. Fiskeri- og Havbruksnæringen er sentral for verdi- skapningen. På havets bunn borer vi, og henter opp store verdier. INTET ER SÅ ROMMELIG SOM HAVET, INTET SÅ TÅLMODIG. PÅ SIN BREDE RYGG BÆRER DET LIK EN GODSLIG ELEFANT DE SMÅ PUSLINGER SOM BEBOR JORDEN; OG I SITT STORE KJØLIGE DYP EIER DET PLASS FOR ALL VERDENS JAMMER. DET ER IKKE SANT AT HAVET ER TROLØST; FOR DET HAR ALDRI FORORD LOVET NOE: UTEN KRAV, UTEN FORPLIKTELSE, FRITT, RENT OG UFORFALSKET BANKER DET STORE HJERTE – DET SISTE SUNNE I DEN SYKE VERDEN. Havet i Byen, ALEKSANDER KIELLAND På overflaten og i strandsonen nyter vi livet. -

Planbeskrivelse Detaljregulering Med Ku Bergenhus, Gnr
Oppdragsgiver J. Berstad Eiendom AS Rapporttype Planbeskrivelse 2017-12-08 PLANBESKRIVELSE DETALJREGULERING MED KU BERGENHUS, GNR. 165, BNR. 574 MFL., NØSTEGATEN BERGENHUS, GNR. 165, BNR. 574 MFL., NØSTEGATEN 3 Oppdragsnr.: 1350001360 Oppdragsnavn: Nøstet sjøfront Dokument nr.: Filnavn: Revisjon 0 Dato 2017-02-24 Utarbeidet av KRJ/LILD/HST Kontrollert av TSK/OAV Godkjent av Beskrivelse Planbeskrivelse Revisjonsoversikt Revisjon Dato Revisjonen gjelder 1 2017-05-16 Suppleringer i tekst og presisering av tema ihht krav fra PB-etaten etter første samlede innlevering: Kap. 8.3 – 8.7, 8.9, 9.1 – 9.4, 9.6 – 9.8, 10 (Vedlegg) 2 2017-10-12 Supplering nytt kap. 9, forslag om påbygging av Nordre Nøstekai 1, jf. også nytt vedlegg 14 3 2017-12-08 Supplering kap. 8.9 og 10.2, med presiseringer av kotehøyder og grad av utnytting før/etter utbygging. Rambøll Torgny Segerstedts vei 27 Pb 3705 Fyllingsdalen NO-5845 BERGEN T +47 55 17 58 00 F +47 55 17 58 10 www.ramboll.no Ramboll 4 (46) BERGENHUS, GNR. 165, BNR. 574 MFL., NØSTEGATEN INNHOLD 1. SAMMENDRAG ......................................................................... 6 2. NØKKELOPPLYSNINGER .......................................................... 6 3. BAKGRUNN FOR PLANARBEIDET ............................................. 7 Bakgrunn .................................................................................. 7 Intensjonen med planforslaget..................................................... 7 4. PLANPROSESSEN .................................................................... 7 Planoppstart -

Cruisekart 2016 Web.Pdf
13 FREE CITY MAP FOR CRUISE PASSENGERS 2016 Concert series in July and August BERGEN Tuesdays in St. Mary’s Church Wednesdays in St. John’s Church 21 INTERNATIONAL Thursdays and Sundays in Bergen Cathedral ORGAN FESTIVAL More information at the Turist Information and in the churches www.windfjord.com www.kirkemusikkibergen.no Slottsgaten 3 N-5003 Bergen, Norway BERGEN 14 THE GATEWAY TO THE FJORDS OF NORWAY ARCTIC ELEGANCE – EXCLUSIVE JEWELLERY Inspired by the magical nature of the Norwegian Lapland. ATTRACTIONS – MUSEUM – SHOPPING JUHLS SILVERGALLERY – Bryggen 39 Tel. +47 55 32 47 40 www.juhls.no 15 Everybody loves adventures. On the fjords. FJORD CRUISE BERGEN-MOSTRAUMEN – ALL YEAR ROUND MORE INFORMATION rodne.no Tel: +47 51 89 52 70, e-mail: [email protected] AND BOOKING AT: A UNIQUE ATTRACTION – THE HOTTEST PLACE TO COOL DOWN! Magic Ice is a wondrous winter wonderland constructed from several tons of ice. The ice bar is the work of world renowned ice carvers, they re-craft the bar several times every year. Magic Ice is the gateway to an adventure far removed from everyday life with a feeling of being beamed over to the Arctic Circle. We provide warm poncho and gloves before i you step inside the ice bar! Magic Ice has been wowing visitors in northern Norway, Oslo and Caribbean for more than 11 years. Open every day, all year around. 16 i C. Sundtsgate 50, 5004 Bergen • e-mail: [email protected] • www.magicice.no 17 FJORDCRUISES • half day excursions • sightseeing tours • active tours www.norled.no Money exchange – 18 19 Geldwechsel – Cambio de divisas In the Tourist Information you can change the most common currencies. -

Bybane Mellom Bergen Sentrum Og Bergen Vest
NOVEMBER 2016 BERGEN KOMMUNE BYBANE MELLOM BERGEN SENTRUM OG BERGEN VEST KOMMUNEDELPLAN TEMARAPPORT TRASÉER OG VENDEMULIGHETER FOR BANE I SENTRUMSFASE 3 DOKUMENTINFORMASJON Rapporttittel: Bybane mellom Bergen Sentrum og Bergen vest Dato: 10.11.2016 Utgave: 1.0 Filnavn: A068178_F3 SF3 Bybane mellom Bergen Sentrum og Bergen vest Oppdragsgiver: Bergen kommune Kontaktperson hos Svein Steine Bergen kommune: Utfører: COWI AS Prosjektleder COWI AS: Marianne Flø Utarbeidet av: Vibeke Forsting, Henrik Juul Vestergaard, Bent Bertil Jacobsen, Michael Goth-Rindal Sidemannskontroll: Marianne Flø Godkjent av: Henrik Juul Vestergaard BYBANE MELLOM BERGEN SENTRUM OG BERGEN VEST 3 TRASÉER OG VENDEMULIGHETER FOR BANE I SENTRUMSFASE 3 INNHOLD INNHOLD 3 FORORD 4 1 Sammendrag 5 2 Bakgrunn 9 2.1 Bakgrunn for arbeidet med Sentrumsfase 3 9 2.2 Formål 9 2.3 Fremtidig bybanenett 9 2.4 Prosess for arbeidet med sentrumsløsning 11 3 Kobling mod Bergen Vest 12 3.1 Puddefjordskryssing 13 3.2 Sammenhengende bybanesystem 14 3.3 BRT gjennom sentrum 16 4 Bergen sentrum –grunnlag for vurderinger 18 4.1 Bosatte og ansatte 18 4.2 Utdannelse 18 4.3 Reisevaner 19 4.4 Kollektiv trafikk 19 4.5 Biltrafikk 24 4.6 Gang og sykkel 27 4.7 Byvekst 31 4.8 Oppsamling 31 5 Gjennomgang av mulige sentrumsløsninger 32 5.1 Strandgaten-løsninger 34 5.2 Tunnel-løsninger under Nygårdshøyden 49 5.3 Holdeplasser 54 5.4 Vendemuligheter i sentrum 56 6 Konsekvensvurdering og anbefaling 58 6.1 Betjening av Sydnes/Dokken 58 6.2 Vurdering av trasévalg gjennom sentrum 59 6.3 Anbefaling av trasé gjennom sentrum 61 BYBANE MELLOM BERGEN SENTRUM OG BERGEN VEST 4 TRASÉER OG VENDEMULIGHETER FOR BANE I SENTRUMSFASE 3 FORORD Denne temarapport er utarbeidet som en del av arbeidet med kommunedelplan og konsekvensutredning for kollektivsystemet mellom Bergen sentrum og Bergen vest.