Do Chlorophyllous Orchids Heterotrophically Use Mycorrhizal Fungal Carbon?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Guide to the Flora of the Carolinas, Virginia, and Georgia, Working Draft of 17 March 2004 -- LILIACEAE
Guide to the Flora of the Carolinas, Virginia, and Georgia, Working Draft of 17 March 2004 -- LILIACEAE LILIACEAE de Jussieu 1789 (Lily Family) (also see AGAVACEAE, ALLIACEAE, ALSTROEMERIACEAE, AMARYLLIDACEAE, ASPARAGACEAE, COLCHICACEAE, HEMEROCALLIDACEAE, HOSTACEAE, HYACINTHACEAE, HYPOXIDACEAE, MELANTHIACEAE, NARTHECIACEAE, RUSCACEAE, SMILACACEAE, THEMIDACEAE, TOFIELDIACEAE) As here interpreted narrowly, the Liliaceae constitutes about 11 genera and 550 species, of the Northern Hemisphere. There has been much recent investigation and re-interpretation of evidence regarding the upper-level taxonomy of the Liliales, with strong suggestions that the broad Liliaceae recognized by Cronquist (1981) is artificial and polyphyletic. Cronquist (1993) himself concurs, at least to a degree: "we still await a comprehensive reorganization of the lilies into several families more comparable to other recognized families of angiosperms." Dahlgren & Clifford (1982) and Dahlgren, Clifford, & Yeo (1985) synthesized an early phase in the modern revolution of monocot taxonomy. Since then, additional research, especially molecular (Duvall et al. 1993, Chase et al. 1993, Bogler & Simpson 1995, and many others), has strongly validated the general lines (and many details) of Dahlgren's arrangement. The most recent synthesis (Kubitzki 1998a) is followed as the basis for familial and generic taxonomy of the lilies and their relatives (see summary below). References: Angiosperm Phylogeny Group (1998, 2003); Tamura in Kubitzki (1998a). Our “liliaceous” genera (members of orders placed in the Lilianae) are therefore divided as shown below, largely following Kubitzki (1998a) and some more recent molecular analyses. ALISMATALES TOFIELDIACEAE: Pleea, Tofieldia. LILIALES ALSTROEMERIACEAE: Alstroemeria COLCHICACEAE: Colchicum, Uvularia. LILIACEAE: Clintonia, Erythronium, Lilium, Medeola, Prosartes, Streptopus, Tricyrtis, Tulipa. MELANTHIACEAE: Amianthium, Anticlea, Chamaelirium, Helonias, Melanthium, Schoenocaulon, Stenanthium, Veratrum, Toxicoscordion, Trillium, Xerophyllum, Zigadenus. -

Managing Scotland's Pinewoods for Their Wild Flowers
Managing Scotland’s pinewoods for their wild fl owers Back from the Brink Management Series Back from the Brink Management Series Over the last two decades, there has been welcome enthusiasm for revitalising Scotland’s Old Caledonian pinewoods. Management has focused on regenerating trees within the few remaining natural woods and establishing trees to create ‘new native woodlands’ but with relatively little attention to the wider plant community of these woods. This booklet provides land managers with advice on managing native and semi-natural pinewoods for their vascular plant communities, based on recent and ongoing management trials. It focuses especially on the characteristic pinewood herbs. Plantlife is the organisation that is Wild plants have been marginalised and speaking up for the nation’s wild plants. taken for granted for too long. Please help We work hard to protect wild plants on the us by supporting our work. ground and to build understanding of the vital role they play in everyone’s lives. Wild plants To find out more, please visit our website are essential to life – they clean our air and or contact us at the office below. water, provide food and shelter for our insects, birds and animals and are critical in the fight Plantlife Scotland against climate change. Balallan House Allan Park Plantlife carries out practical conservation Stirling work across Scotland, manages nature FK8 2QG reserves, influences policy and legislation, Tel. 01786 478509/479382 runs events and activities that help people discover wild plants and works with others to promote the conservation of wild plants www.plantlife.org.uk for the benefit of all. -

New Record of Goodyera Repens (L.) R. Br
Life Science Journal 2013;10(4) http://www.lifesciencesite.com New record of Goodyera repens (L.) R. Br. (Orchidaceae) on Przedborska Upland (Poland) Andrzej Grzyl 1, Agnieszka Rewicz 2, Spyros Tsiftsis 3 1,2 Department of Geobotany and Plant Ecology, Faculty of Biology and Environmental Protection, University of Łódź, Banacha 12/16, 90-237 Łódź, Poland 3 Department of Botany, School of Biology, Aristotle University of Thessaloniki, 54124 Thessaloniki, Greece [email protected] Abstract: Goodyera repens in Poland occur mainly in the northern and north-eastern parts of the country, as well as in the Carpathians and the Sudeten Mountains. In central Poland it could be considered as a rare species which has been found only in a limited number of sites with a small number of individuals. In Poland it is characterized as a critically endangered species which faces the danger of extinction in many of its sites. A new site of G. repen has been found in central Poland hosting a relatively large number of individuals. Data about its habitat, its population size as well as its spatial structure are provided. [Grzyl, A, Rewicz, A, Tsiftsis, S. New record of Goodyera repens (L.) R. Br. (Orchidaceae) on Przedborska Upland (Poland). Life Sci J 2013;10(4):2993-2995]. (ISSN:1097-8135). http://www.lifesciencesite.com. 397 Key words: Goodyera repens; Chęcińsko-Kielecki Landacape Park; floristic diversity; rare species; protected species; new locality 1. Introduction (Kucharczyk and Wójciak, 1995), while according to Goodyera repens (L.) R. Br. (Creeping Lady's Bróż and Przemyski (2009) in western Poland it is Tresses) is a circumboreal species that mainly occurs regarded as vulnerable (V) and in northern Poland in Central, North and East Europe, Caucasus, North (Gdańsk Pomerania) is considered as near threatened and East Asia, the Himalayas and North America (NT) (Markowski and Buliński, 2004). -

Response of Cypripedium and Goodyera to Disturbance in the Thunder Bay Area
Lakehead University Knowledge Commons,http://knowledgecommons.lakeheadu.ca Electronic Theses and Dissertations Undergraduate theses 2018 Response of Cypripedium and Goodyera to disturbance in the Thunder Bay area Davis, Danielle http://knowledgecommons.lakeheadu.ca/handle/2453/4426 Downloaded from Lakehead University, KnowledgeCommons 5(63216(2)&<35,3(',80$1'*22'<(5$72',6785%$1&(,1 7+(7+81'(5%$<$5($ E\ 'DQLHOOH'DYLV )$&8/7<2)1$785$/5(6285&(60$1$*(0(17 /$.(+($'81,9(56,7< 7+81'(5%$<217$5,2 0D\ RESPONSE OF CYPRIPEDIUM AND GOODYERA TO DISTURBANCE IN THE THUNDER BAY AREA by Danielle Davis An Undergraduate Thesis Submitted in Partial Fulfillment of the Requirements for the Degree of Honours Bachelor of Environmental Management Faculty of Natural Resources Management Lakehead University May 2018 Major Advisor Second Reader ii LIBRARY RIGHTS STATEMENT In presenting this thesis in partial fulfillment of the requirements for the HBEM degree at Lakehead University in Thunder Bay, I agree that the University will make it freely available for inspection. This thesis is made available by my authority solely for the purpose of private study and research and may not be copied or reproduced in whole or in part (except as permitted by the Copyright Laws) without my written authority. Signature: Date: iii A CAUTION TO THE READER This HBEM thesis has been through a semi-formal process of review and comment by at least two faculty members. It is made available for loan by the Faculty of Natural Resources Management for the purpose of advancing the practice of professional and scientific forestry. -

Conservation Assessment for White Adder's Mouth Orchid (Malaxis B Brachypoda)
Conservation Assessment for White Adder’s Mouth Orchid (Malaxis B Brachypoda) (A. Gray) Fernald Photo: Kenneth J. Sytsma USDA Forest Service, Eastern Region April 2003 Jan Schultz 2727 N Lincoln Road Escanaba, MI 49829 906-786-4062 This Conservation Assessment was prepared to compile the published and unpublished information on Malaxis brachypoda (A. Gray) Fernald. This is an administrative study only and does not represent a management decision or direction by the U.S. Forest Service. Though the best scientific information available was gathered and reported in preparation for this document and subsequently reviewed by subject experts, it is expected that new information will arise. In the spirit of continuous learning and adaptive management, if the reader has information that will assist in conserving the subject taxon, please contact: Eastern Region, USDA Forest Service, Threatened and Endangered Species Program, 310 Wisconsin Avenue, Milwaukee, Wisconsin 53203. Conservation Assessment for White Adder’s Mouth Orchid (Malaxis Brachypoda) (A. Gray) Fernald 2 TABLE OF CONTENTS TABLE OF CONTENTS .................................................................................................................1 ACKNOWLEDGEMENTS..............................................................................................................2 EXECUTIVE SUMMARY ..............................................................................................................3 INTRODUCTION/OBJECTIVES ...................................................................................................3 -

Factors Determining the Distribution of Orchids – a Review with Examples from the Czech Republic
European Journal of Environmental Sciences 21 FACTORS DETERMINING THE DISTRIBUTION OF ORCHIDS – A REVIEW WITH EXAMPLES FROM THE CZECH REPUBLIC ZUZANA ŠTÍPKOVÁ1,2,* and PAVEL KINDLMANN1,2 1 Global Change Research Institute, Czech Academy of Science, Bělidla 986/4a, 60300 Brno, Czech Republic 2 Institute for Environmental Studies, Faculty of Science, Charles University, Benátská 2, 12801 Prague 2, Czech Republic * Corresponding author: [email protected] ABSTRACT The natural environment has been significantly altered by human activity over the past few decades. There is evidence we are now experiencing the sixth mass extinction, as many species of plants and animals are declining in abundance. We focused on the Orchidaceae because this plant family has experienced one of the biggest reductions in distribution. We investigated patterns in species richness and distribution of orchids, the rate and causes of their decrease and extinction, and factors influencing their occurrence in the Czech Republic and Greece. The key findings are: (i) Method of pollination and type of rooting system are associated with their distributions and they are different in the two countries. We assume that these differences might be due to the difference in the orography, distribution of suitable habitats and types of bedrock in these two countries. (ii) The greatest reduction in distribution was recorded for critically endangered taxa of orchids. The number of sites suitable for orchids in the Czech Republic declined by 8–92%. The most threatened orchid species are Spiranthes spiralis, Anacamptis palustris, Epipogium aphyllum and Goodyera repens. The distribution of orchids in the Czech Republic is mainly determined by the distribution of their habitats. -

Vegetation Community Monitoring at Congaree National Park: 2014 Data Summary
National Park Service U.S. Department of the Interior Natural Resource Stewardship and Science Vegetation Community Monitoring at Congaree National Park 2014 Data Summary Natural Resource Data Series NPS/SECN/NRDS—2016/1016 ON THIS PAGE Tiny, bright yellow blossoms of Hypoxis hirsuta grace the forest floor at Congaree National Park. Photograph courtesy of Sarah C. Heath, Southeast Coast Network. ON THE COVER Spiraling compound leaf of green dragon (Arisaema dracontium) at Congaree National Park. Photograph courtesy of Sarah C. Heath, Southeast Coast Network Vegetation Community Monitoring at Congaree National Park 2014 Data Summary Natural Resource Data Series NPS/SECN/NRDS—2016/1016 Sarah Corbett Heath1 and Michael W. Byrne2 1National Park Service Southeast Coast Inventory and Monitoring Network Cumberland Island National Seashore 101 Wheeler Street Saint Marys, GA 31558 2National Park Service Southeast Coast Inventory and Monitoring Network 135 Phoenix Drive Athens, GA 30605 May 2016 U.S. Department of the Interior National Park Service Natural Resource Stewardship and Science Fort Collins, Colorado The National Park Service, Natural Resource Stewardship and Science office in Fort Collins, Colorado, publishes a range of reports that address natural resource topics. These reports are of interest and applicability to a broad audience in the National Park Service and others in natural resource management, including scientists, conservation and environmental constituencies, and the public. The Natural Resource Data Series is intended for the timely release of basic data sets and data summaries. Care has been taken to assure accuracy of raw data values, but a thorough analysis and interpretation of the data has not been completed. -

FOREST FLORA Woodland Conservation News • Spring 2018
Wood Wise FOREST FLORA Woodland Conservation News • Spring 2018 QUESTIONING SURVEYING TIME FOR WOODLAND ANCIENT & RESTORING A BUNCE FLORA WOODLAND WOODLAND WOODLAND FOLKLORE INDICATORS FLORA RESURVEY & REMEDIES CONTENTS Smith / Georgina WTML 3 4 8 11 16 20 22 24 Introduction Spring is probably the best time to appreciate woodland The Woodland Trust’s Alastair Hotchkiss focuses on the ground flora. The warming temperatures encourage restoration of plantations on ancient woodland sites. 25 shoots to burst from bulbs, breaking through the soil Gradual thinning and natural regeneration of native trees Introduction to reach the sunlight not yet blocked out by leaves that can allow sunlight to reach the soil again and enable the 3 will soon fill the tree canopy. Their flowers bring colour return of wildflowers surviving in the seed bank. to break the stark winter and inspire hope in many that Questioning ancient woodland indicators The Woodland Survey of Great Britain 1971-2001 4 see them. recorded a change and loss in broadleaf woodland The species of flora covered in this issue are vascular ground flora species. The driver was thought to be a plants found on the woodland floor layer. Vascular plants 8 Trouble for bluebells? reduction in the use and management of woods. This have specialised tissues within their form that carry finding encouraged an increase in management. Several The world beneath your feet water and nutrients around their stems, leaves and other experts involved look at this and the potential for a future 11 structures, which allow them to grow taller than non- resurvey to analyse the current situation. -

Pseudorchis Albida (L.) Á
Pseudorchis albida (L.) Á. and D. Löve Small White Orchid As the common name suggests, Pseudorchis albida is a diminutive orchid with tiny, creamy- white, scented flowers that attract a variety of day-flying insects. It is able to tolerate a wide range of ecological conditions, having a pH range of between 4 and 7, but is most often found in low fertility mesotrophic grasslands and acid heathlands, and more rarely in base- rich mires, limestone heath, and cliff ledges. It is most widespread in the north and west of Scotland but has experienced substantial declines across its range, particularly in Ireland, England and Wales. Pseudorchis albida is assessed as Vulnerable in GB and Critically Endangered in Wales. ©Joan Canals IDENTIFICATION having stomata above (only on the underside of the leaf in P. albida), G. repens by the presence of petioles and C. viride by Pseudorchis albida is a diminutive orchid with a dense spike the absence of longitudinal streaking (Poland & Clements, of very small, creamy-white flowers (2-4 mm across) with a 2009). In Britain, hybrids with Gymnadenia borealis have deeply three-lobed lip. Its leaves have translucent longitudinal been widely reported from Scotland and northern England streaks, are glossy green above, silvery below, and have a and with Dactylorhiza maculata in Skye and Orkney. hooded tip (Poland & Clements, 2009). Throughout its range Reported hybrids with Platanthera chlorantha are now P. albida is probably overlooked due to its small stature, short thought to be aberrant forms of the latter. flowering period, erratic appearance and tendency to form small populations (Duffey et al., 2008). -

Chapter 8 DEMOGRAPHIC STUDIES and LIFE-HISTORY STRATEGIES
K.w. Dixon, S.P. Kell, R.L. Barrett and P.J. Cribb (eds) 2003. Orchid Conservation. pp. 137-158. © Natural History Publications (Borneo), Kota Kinabalu, Sabah. Chapter 8 DEMOGRAPHIC STUDIES AND LIFE-HISTORY STRATEGIES OF TEMPERATE TERRESTRIAL ORCIDDS AS A BASIS FOR CONSERVATION Dennis F Whigham Smithsonian Environmental Research Center, Box. 28, Edgewater, MD 21037, USA. Jo H. Willems Plant Ecology Group, Utrecht University, PO Box 800.84, NL 3508 Utrecht, The Netherlands. "Our knowledge about the lives ofindividual plants and oftheir persistence in plant communities is very incomplete, however, especially when perennial herbs are concerned" - C. 0. Tamm, 1948. Terrestrial orchids represent a wide diversity ofspecies that are characterised by an equally diverse range of life history attributes. Threatened and endangered species of terrestrial orchids have been identifed on all continents where they occur and conservation plans have been developed for some species. Even though there is a considerable amount of information on the ecology of terrestrial orchids, few species have been studied in detail and most management plans focus on habitat conservation. In this paper, we consider the diversity of terrestrial orchids and summarise information on threatened and endangered species from a global perspective. We also describe approaches to the conservation and restoration ofterrestrial orchids and develop the argument that much information is needed ifwe are to successfully conserve this diverse group ofplant species. 1. Introduction Terrestrial orchids represent a wide variety of life history types, from autotrophic evergreen to completely myco-heterotrophic species that obtain most oftheir resources from a mycobiont. Life history characteristics ofterrestrial orchids are generally well known (e.g. -
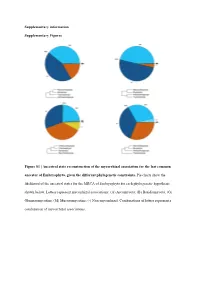
Ancestral State Reconstruction of the Mycorrhizal Association for the Last Common Ancestor of Embryophyta, Given the Different Phylogenetic Constraints
Supplementary information Supplementary Figures Figure S1 | Ancestral state reconstruction of the mycorrhizal association for the last common ancestor of Embryophyta, given the different phylogenetic constraints. Pie charts show the likelihood of the ancestral states for the MRCA of Embryophyta for each phylogenetic hypothesis shown below. Letters represent mycorrhizal associations: (A) Ascomycota; (B) Basidiomycota; (G) Glomeromycotina; (M) Mucoromycotina; (-) Non-mycorrhizal. Combinations of letters represent a combination of mycorrhizal associations. Austrocedrus chilensis Chamaecyparis obtusa Sequoiadendron giganteum Prumnopitys taxifolia Prumnopitys Prumnopitys montana Prumnopitys Prumnopitys ferruginea Prumnopitys Araucaria angustifolia Araucaria Dacrycarpus dacrydioides Dacrycarpus Taxus baccata Podocarpus oleifolius Podocarpus Afrocarpus falcatus Afrocarpus Ephedra fragilis Nymphaea alba Nymphaea Gnetum gnemon Abies alba Abies balsamea Austrobaileya scandens Austrobaileya Abies nordmanniana Thalictrum minus Thalictrum Abies homolepis Caltha palustris Caltha Abies magnifica ia repens Ranunculus Abies religiosa Ranunculus montanus Ranunculus Clematis vitalba Clematis Keteleeria davidiana Anemone patens Anemone Tsuga canadensis Vitis vinifera Vitis Tsuga mertensiana Saxifraga oppositifolia Saxifraga Larix decidua Hypericum maculatum Hypericum Larix gmelinii Phyllanthus calycinus Phyllanthus Larix kaempferi Hieronyma oblonga Hieronyma Pseudotsuga menziesii Salix reinii Salix Picea abies Salix polaris Salix Picea crassifolia Salix herbacea -

Ecological Survey of the Native Pinewoods of Scotland 1971
Earth Syst. Sci. Data, 8, 177–189, 2016 www.earth-syst-sci-data.net/8/177/2016/ doi:10.5194/essd-8-177-2016 © Author(s) 2016. CC Attribution 3.0 License. Ecological survey of the native pinewoods of Scotland 1971 Claire M. Wood1 and Robert G. H. Bunce2 1Centre for Ecology and Hydrology, Lancaster Environment Centre, Bailrigg, Lancaster LA1 4AP, UK 2Estonian University of Life Sciences, Kreuzwaldi 5, 51014 Tartu, Estonia Correspondence to: Claire M. Wood ([email protected]) Received: 3 February 2016 – Published in Earth Syst. Sci. Data Discuss.: 10 February 2016 Revised: 8 April 2016 – Accepted: 20 April 2016 – Published: 10 May 2016 Abstract. In 1971, a comprehensive ecological survey of the native pinewoods of Scotland was carried out by the Institute of Terrestrial Ecology. The survey was initiated as a consequence of growing concern about the status of the pinewood resource. Since the twentieth century, this unique habitat is widely recognised, not only by ecologists for its inherent biodiversity but also by the general public for its cultural and amenity value. The survey, utilising demonstrably repeatable methods, collected information on ground flora, soils, forest structure and also general site information from the major 27 sites of the 35 sites identified as truly native pinewoods in Scotland. The results from the survey prompted the organisation of an international symposium in 1975, which set the conservation agenda for the old Caledonian pinewoods. The data collected during the 1971 survey are now publicly available via the following DOI: doi:10/7xb (“Habitat, vegetation, tree and soil data from Native Pinewoods in Scotland, 1971”).