Technical Commentary
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Npp2013223.Pdf
Neuropsychopharmacology (2014) 39, 499–506 & 2014 American College of Neuropsychopharmacology. All rights reserved 0893-133X/14 www.neuropsychopharmacology.org Chronic Administration of the Methylxanthine Propentofylline Impairs Reinstatement to Cocaine by a GLT-1-Dependent Mechanism ,1 1,2 1 1 1 Kathryn J Reissner* , Robyn M Brown , Sade Spencer , Phuong K Tran , Charles A Thomas and 1 Peter W Kalivas 1 2 Department of Neurosciences, Medical University of South Carolina, Charleston, SC, USA; Florey Institute of Neuroscience and Mental Health, University of Melbourne, Parkville, Australia In recent years, interactions between neurons and glia have been evaluated as mediators of neuropsychiatric diseases, including drug addiction. In particular, compounds that increase expression of the astroglial glutamate transporter GLT-1 (N-acetylcysteine and ceftriaxone) can decrease measures of drug seeking. However, it is unknown whether the compounds that influence broad measures of glial physiology can influence behavioral measures of drug relapse, nor is it clear whether the upregulated GLT-1 is functionally important for suppressing of drug seeking. To address these questions, we sought to determine whether the glial modulator and neuroprotective agent propentofylline (PPF) modifies drug seeking in rats using a reinstatement model of cocaine relapse. We found that 7 days of chronic (but not acute) administration of PPF significantly decreased both cue- and cocaine-induced reinstatement of cocaine seeking. We next determined whether the effect of systemic PPF on reinstatement depended upon its ability to restore expression of GLT-1 in the nucleus accumbens. PPF restored the cocaine-induced decrease in GLT-1 in the accumbens core; then, using an antisense strategy against glutamate transporter GLT-1, we found that restored transporter expression was necessary for PPF to inhibit cue-primed cocaine seeking. -

Structure-Activity Relationship and Mechanistic Studies on The
University of Tennessee Health Science Center UTHSC Digital Commons Theses and Dissertations (ETD) College of Graduate Health Sciences 12-2008 Structure-Activity Relationship and Mechanistic Studies on the Chemopreventive Activity of Dipyridamole and Its Analogues Ja’Wanda Shavon Grant University of Tennessee Health Science Center Follow this and additional works at: https://dc.uthsc.edu/dissertations Part of the Pharmacy and Pharmaceutical Sciences Commons Recommended Citation Grant, Ja’Wanda Shavon , "Structure-Activity Relationship and Mechanistic Studies on the Chemopreventive Activity of Dipyridamole and Its Analogues" (2008). Theses and Dissertations (ETD). Paper 100. http://dx.doi.org/10.21007/ etd.cghs.2008.0114. This Dissertation is brought to you for free and open access by the College of Graduate Health Sciences at UTHSC Digital Commons. It has been accepted for inclusion in Theses and Dissertations (ETD) by an authorized administrator of UTHSC Digital Commons. For more information, please contact [email protected]. Structure-Activity Relationship and Mechanistic Studies on the Chemopreventive Activity of Dipyridamole and Its Analogues Document Type Dissertation Degree Name Doctor of Philosophy (PhD) Program Pharmaceutical Sciences Research Advisor John K. Buolamwini, Ph.D. Committee Richard E. Lee, Ph.D. Duane Miller, Ph.D. David Nelson, Ph.D. Jie Zheng, Ph.D. DOI 10.21007/etd.cghs.2008.0114 This dissertation is available at UTHSC Digital Commons: https://dc.uthsc.edu/dissertations/100 STRUCTURE-ACTIVITY RELATIONSHIP AND -

Drugs and Medication Guidelines Brochure
Equine Medication Monitoring Program Drugs and Medication Guidelines January 2021 1 Introduction The California Equine Medication Monitoring Program (EMMP) is an industry funded program to ensure the integrity of public equine events and sales in California through the control of performance and disposition enhancing drugs and permitting limited therapeutic use of drugs and medications. The EMMP and the industry is dedicated and committed to promote the health, welfare and safety of the equine athlete. Owners, trainers, exhibitors, veterinarians and consignors of equines to public sales have a responsibility to be familiar with the California EMMP and the California Equine Medication Rule. California law (Food and Agricultural Code Sections 24000-24018) outlines the equine medication rule for public equine events in California. The owner, trainer and consignor have responsibility to ensure full compliance with all elements of the California Equine Medication Rule. Owners, trainers, exhibitors, veterinarians and consignors of equines to public sales must comply with both the California Equine Medication Rule and any sponsoring organization drug and medication rule for an event. The more stringent medication rule applies for the event. The California Equine Medication Rule is posted on the website: http://www.cdfa.ca.gov/ahfss/Animal_Health/emmp/ The information contained in this document provides advice regarding the California Equine Medication Rule and application of the rule to practical situations. The EMMP recognizes that situations arise where there is an indication for legitimate therapeutic treatment near the time of competition at equine events. The EMMP regulations permit the use of therapeutic medication in certain circumstances to accommodate legitimate therapy in compliance with the requirements of the rule. -

Exploring the Glial Mechanisms Underlying Central Neuropathic Pain in a Novel Rat Model of Spinal Cord Injury
i Exploring the Glial Mechanisms Underlying Central Neuropathic Pain in a Novel Rat Model of Spinal Cord Injury by Amanda Lynne Ellis B.A., University of Colorado, 2006 M.A., University of Colorado, 2011 A thesis submitted to the Faculty of the Graduate School of the University of Colorado in partial fulfillment of the requirement of the degree of Doctor of Philosophy Department of Psychology and Neuroscience 2014 ii This thesis entitled: Exploring the Glial Mechanisms Underlying Central Neuropathic Pain in a Novel Rat Model of Spinal Cord Injury written by Amanda Lynne Ellis has been approved for the Department of Psychology and Neuroscience ___________________________________ Dr. Linda Watkins ______________________________________________ Dr. Steven Maier ___________________________________________ Dr. Serge Campeau _______________________________________________ Dr. Benjamin Greenwood _______________________________________________ Dr. Michael Stallings Date____________ The final copy of this thesis has been examined by the signatories, and we Find that both the content and the form meet acceptable presentation standards Of scholarly work in the above mentioned discipline iii Abstract Ellis, Amanda Lynne (Ph.D., Psychology and Neuroscience) Glial Mechanisms of Central Neuropathic Pain Thesis directed by Distinguished Professor Linda Watkins Neuropathic pain is a debilitating condition that is usually intractable to treatment. Spinal cord injury (SCI) is the leading cause of central neuropathic pain (CNP), and patients often describe their pain as severe and intolerable. Although most of the focus in the pain literature has been on neurons, it is now well known that non-neuronal cells of the central nervous system called microglia and astrocytes (herein referred to as “glial cells” or “glia”) play a key role in the induction and maintenance of neuropathic pain. -

Methylxanthines and Neurodegenerative Diseases: an Update
nutrients Review Methylxanthines and Neurodegenerative Diseases: An Update Daniel Janitschke 1,† , Anna A. Lauer 1,†, Cornel M. Bachmann 1, Heike S. Grimm 1, Tobias Hartmann 1,2 and Marcus O. W. Grimm 1,2,* 1 Experimental Neurology, Saarland University, 66421 Homburg/Saar, Germany; [email protected] (D.J.); [email protected] (A.A.L.); [email protected] (C.M.B.); [email protected] (H.S.G.); [email protected] (T.H.) 2 Deutsches Institut für DemenzPrävention (DIDP), Saarland University, 66421 Homburg/Saar, Germany * Correspondence: [email protected] † These authors contributed equally to this work. Abstract: Methylxanthines (MTX) are purine derived xanthine derivatives. Whereas naturally occurring methylxanthines like caffeine, theophylline or theobromine are widely consumed in food, several synthetic but also non-synthetic methylxanthines are used as pharmaceuticals, in particular in treating airway constrictions. Besides the well-established bronchoprotective effects, methylxanthines are also known to have anti-inflammatory and anti-oxidative properties, mediate changes in lipid homeostasis and have neuroprotective effects. Known molecular mechanisms include adenosine receptor antagonism, phosphodiesterase inhibition, effects on the cholinergic system, wnt signaling, histone deacetylase activation and gene regulation. By affecting several pathways associated with neurodegenerative diseases via different pleiotropic mechanisms and due to its moderate side effects, intake of methylxanthines have been suggested to be an interesting approach in dealing with neurodegeneration. Especially in the past years, the impact of methylxanthines in neurodegenerative diseases has been extensively studied and several new aspects have been elucidated. In this review Citation: Janitschke, D.; Lauer, A.A.; Bachmann, C.M.; Grimm, H.S.; we summarize the findings of methylxanthines linked to Alzheimer´s disease, Parkinson’s disease Hartmann, T.; Grimm, M.O.W. -

Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DIX to the HTSUS—Continued
20558 Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DEPARMENT OF THE TREASURY Services, U.S. Customs Service, 1301 TABLE 1.ÐPHARMACEUTICAL APPEN- Constitution Avenue NW, Washington, DIX TO THE HTSUSÐContinued Customs Service D.C. 20229 at (202) 927±1060. CAS No. Pharmaceutical [T.D. 95±33] Dated: April 14, 1995. 52±78±8 ..................... NORETHANDROLONE. A. W. Tennant, 52±86±8 ..................... HALOPERIDOL. Pharmaceutical Tables 1 and 3 of the Director, Office of Laboratories and Scientific 52±88±0 ..................... ATROPINE METHONITRATE. HTSUS 52±90±4 ..................... CYSTEINE. Services. 53±03±2 ..................... PREDNISONE. 53±06±5 ..................... CORTISONE. AGENCY: Customs Service, Department TABLE 1.ÐPHARMACEUTICAL 53±10±1 ..................... HYDROXYDIONE SODIUM SUCCI- of the Treasury. NATE. APPENDIX TO THE HTSUS 53±16±7 ..................... ESTRONE. ACTION: Listing of the products found in 53±18±9 ..................... BIETASERPINE. Table 1 and Table 3 of the CAS No. Pharmaceutical 53±19±0 ..................... MITOTANE. 53±31±6 ..................... MEDIBAZINE. Pharmaceutical Appendix to the N/A ............................. ACTAGARDIN. 53±33±8 ..................... PARAMETHASONE. Harmonized Tariff Schedule of the N/A ............................. ARDACIN. 53±34±9 ..................... FLUPREDNISOLONE. N/A ............................. BICIROMAB. 53±39±4 ..................... OXANDROLONE. United States of America in Chemical N/A ............................. CELUCLORAL. 53±43±0 -

(12) Patent Application Publication (10) Pub. No.: US 2003/0068365A1 Suvanprakorn Et Al
US 2003.0068365A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2003/0068365A1 Suvanprakorn et al. (43) Pub. Date: Apr. 10, 2003 (54) COMPOSITIONS AND METHODS FOR Related U.S. Application Data ADMINISTRATION OF ACTIVE AGENTS USING LIPOSOME BEADS (60) Provisional application No. 60/327,643, filed on Oct. 5, 2001. (76) Inventors: Pichit Suvanprakorn, Bangkok (TH); Tanusin Ploysangam, Bangkok (TH); Publication Classification Lerson Tanasugarn, Bangkok (TH); Suwalee Chandrkrachang, Bangkok (51) Int. Cl." .......................... A61K 9/127; A61K 35/78 (TH); Nardo Zaias, Miami Beach, FL (52) U.S. Cl. ............................................ 424/450; 424/725 (US) (57) ABSTRACT Correspondence Address: Law Office of Eric G. Masamori Compositions and methods for administration of active 6520 Ridgewood Drive agents encapsulated within liposome beads to enable a wider Castro Valley, CA 94.552 (US) range of delivery vehicles, to provide longer product shelf life, to allow multiple active agents within the composition, (21) Appl. No.: 10/264,205 to allow the controlled use of the active agents, to provide protected and designable release features and to provide (22) Filed: Oct. 3, 2002 Visual inspection for damage and inconsistency. US 2003/0068365A1 Apr. 10, 2003 COMPOSITIONS AND METHODS FOR toxic degradation of the products, leakage of the drug from ADMINISTRATION OF ACTIVE AGENTS USING the liposome and the modifications of the Size and morphol LPOSOME BEADS ogy of the phospholipid liposome vesicles through aggre gation and fusion. Liposome vesicles are known to be CROSS REFERENCE TO OTHER thermodynamically relatively unstable at room temperature APPLICATIONS and can Spontaneously fuse into larger, leSS Stable altered liposome forms. -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -

A Placebo Controlled Study of the Propentofylline Added to Risperidone in Chronic Schizophrenia
Available online at www.sciencedirect.com Progress in Neuro-Psychopharmacology & Biological Psychiatry 32 (2008) 726–732 www.elsevier.com/locate/pnpbp A placebo controlled study of the propentofylline added to risperidone in chronic schizophrenia Samarand Salimi a, Akbar Fotouhi b, Abolfazl Ghoreishi c, Mohammad-Kamran Derakhshan d, Mohammad-Reza Khodaie-Ardakani d, Mohammad-Reza Mohammadi a, Ahmad-Ali Noorbala a, Seyed-Ali Ahmadi-Abhari a, Mohammad Hajiazim e, ⁎ Seyed-Hesameddin Abbasi f, Shahin Akhondzadeh a, a Psychiatric Research Center, Roozbeh Psychiatric Hospital, Tehran University of Medical Sciences, Tehran, Iran b Department of Epidemiology and Biostatistics, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran c Departement of Psychiatry, Zanjan University of Medical Sciences, Zanjan, Iran d Razi Psychiatric Hospital, Welfare Sciences University, Iran e Semnan University of Medical Sciences, Garmsar, Iran f National Iranian Oil Company, Central Hospital, Tehran, Iran Received 10 August 2007; received in revised form 9 November 2007; accepted 17 November 2007 Available online 23 November 2007 Abstract Impaired activity of the purinergic system is a plausible common factor that could be responsible for many aspects of schizophrenia. Based on purinegic hypothesis of schizophrenia, pharmacological treatments enhancing adenosine activity could be effective treatment in schizophrenia. Propentofylline is a novel xantine derivative which is being developed for treatment of degenerative and vascular dementia. It enhances extracellular adenosine level via inhibition of adenosine uptake. The purpose of the present investigation was to assess the efficacy of propentofylline as an adjuvant agent in the treatment of chronic schizophrenia in an 8-week double blind and placebo controlled trial. -
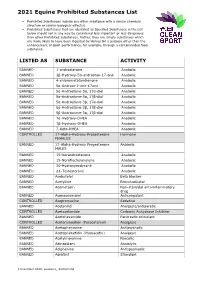
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

Wo 2007/120485 A2
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (43) International Publication Date PCT (10) International Publication Number 25 October 2007 (25.10.2007) WO 2007/120485 A2 (51) International Patent Classification: IS, JP, KE, KG, KM, KN, KP, KR, KZ, LA, LC, LK, LR, A61K 31/195 (2006.01) LS, LT, LU, LY,MA, MD, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PG, PH, PL, PT, RO, RS, (21) International Application Number: RU, SC, SD, SE, SG, SK, SL, SM, SV, SY, TJ, TM, TN, PCT/US2007/0081 14 TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW (22) International Filing Date: 30 March 2007 (30.03.2007) (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (25) Filing Language: English GM, KE, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, TM), (26) Publication Language: English European (AT,BE, BG, CH, CY, CZ, DE, DK, EE, ES, FI, FR, GB, GR, HU, IE, IS, IT, LT,LU, LV,MC, MT, NL, PL, PT, RO, SE, SI, SK, TR), OAPI (BF, BJ, CF, CG, CI, CM, (30) Priority Data: 60/787,150 30 March 2006 (30.03.2006) US GA, GN, GQ, GW, ML, MR, NE, SN, TD, TG). Declarations under Rule 4.17: (71) Applicant (for all designated States except US): CINER- — as to applicant's entitlement to apply for and be granted a GEN, LLC [US/US]; 146 Medinah Drive, Blue Bell, PA patent (Rule 4.17(U)) 19422-3212 (US). -

Customs Tariff - Schedule
CUSTOMS TARIFF - SCHEDULE 99 - i Chapter 99 SPECIAL CLASSIFICATION PROVISIONS - COMMERCIAL Notes. 1. The provisions of this Chapter are not subject to the rule of specificity in General Interpretative Rule 3 (a). 2. Goods which may be classified under the provisions of Chapter 99, if also eligible for classification under the provisions of Chapter 98, shall be classified in Chapter 98. 3. Goods may be classified under a tariff item in this Chapter and be entitled to the Most-Favoured-Nation Tariff or a preferential tariff rate of customs duty under this Chapter that applies to those goods according to the tariff treatment applicable to their country of origin only after classification under a tariff item in Chapters 1 to 97 has been determined and the conditions of any Chapter 99 provision and any applicable regulations or orders in relation thereto have been met. 4. The words and expressions used in this Chapter have the same meaning as in Chapters 1 to 97. Issued January 1, 2020 99 - 1 CUSTOMS TARIFF - SCHEDULE Tariff Unit of MFN Applicable SS Description of Goods Item Meas. Tariff Preferential Tariffs 9901.00.00 Articles and materials for use in the manufacture or repair of the Free CCCT, LDCT, GPT, following to be employed in commercial fishing or the commercial UST, MXT, CIAT, CT, harvesting of marine plants: CRT, IT, NT, SLT, PT, COLT, JT, PAT, HNT, Artificial bait; KRT, CEUT, UAT, CPTPT: Free Carapace measures; Cordage, fishing lines (including marlines), rope and twine, of a circumference not exceeding 38 mm; Devices for keeping nets open; Fish hooks; Fishing nets and netting; Jiggers; Line floats; Lobster traps; Lures; Marker buoys of any material excluding wood; Net floats; Scallop drag nets; Spat collectors and collector holders; Swivels.