Dysregulation of Micro-143-3P and BALBP1 Contributes to the Pathogenesis of the Development of Ovarian Carcinoma
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Capacity of Long-Term in Vitro Proliferation of Acute Myeloid
The Capacity of Long-Term in Vitro Proliferation of Acute Myeloid Leukemia Cells Supported Only by Exogenous Cytokines Is Associated with a Patient Subset with Adverse Outcome Annette K. Brenner, Elise Aasebø, Maria Hernandez-Valladares, Frode Selheim, Frode Berven, Ida-Sofie Grønningsæter, Sushma Bartaula-Brevik and Øystein Bruserud Supplementary Material S2 of S31 Table S1. Detailed information about the 68 AML patients included in the study. # of blasts Viability Proliferation Cytokine Viable cells Change in ID Gender Age Etiology FAB Cytogenetics Mutations CD34 Colonies (109/L) (%) 48 h (cpm) secretion (106) 5 weeks phenotype 1 M 42 de novo 241 M2 normal Flt3 pos 31.0 3848 low 0.24 7 yes 2 M 82 MF 12.4 M2 t(9;22) wt pos 81.6 74,686 low 1.43 969 yes 3 F 49 CML/relapse 149 M2 complex n.d. pos 26.2 3472 low 0.08 n.d. no 4 M 33 de novo 62.0 M2 normal wt pos 67.5 6206 low 0.08 6.5 no 5 M 71 relapse 91.0 M4 normal NPM1 pos 63.5 21,331 low 0.17 n.d. yes 6 M 83 de novo 109 M1 n.d. wt pos 19.1 8764 low 1.65 693 no 7 F 77 MDS 26.4 M1 normal wt pos 89.4 53,799 high 3.43 2746 no 8 M 46 de novo 26.9 M1 normal NPM1 n.d. n.d. 3472 low 1.56 n.d. no 9 M 68 MF 50.8 M4 normal D835 pos 69.4 1640 low 0.08 n.d. -

1 Supporting Information for a Microrna Network Regulates
Supporting Information for A microRNA Network Regulates Expression and Biosynthesis of CFTR and CFTR-ΔF508 Shyam Ramachandrana,b, Philip H. Karpc, Peng Jiangc, Lynda S. Ostedgaardc, Amy E. Walza, John T. Fishere, Shaf Keshavjeeh, Kim A. Lennoxi, Ashley M. Jacobii, Scott D. Rosei, Mark A. Behlkei, Michael J. Welshb,c,d,g, Yi Xingb,c,f, Paul B. McCray Jr.a,b,c Author Affiliations: Department of Pediatricsa, Interdisciplinary Program in Geneticsb, Departments of Internal Medicinec, Molecular Physiology and Biophysicsd, Anatomy and Cell Biologye, Biomedical Engineeringf, Howard Hughes Medical Instituteg, Carver College of Medicine, University of Iowa, Iowa City, IA-52242 Division of Thoracic Surgeryh, Toronto General Hospital, University Health Network, University of Toronto, Toronto, Canada-M5G 2C4 Integrated DNA Technologiesi, Coralville, IA-52241 To whom correspondence should be addressed: Email: [email protected] (M.J.W.); yi- [email protected] (Y.X.); Email: [email protected] (P.B.M.) This PDF file includes: Materials and Methods References Fig. S1. miR-138 regulates SIN3A in a dose-dependent and site-specific manner. Fig. S2. miR-138 regulates endogenous SIN3A protein expression. Fig. S3. miR-138 regulates endogenous CFTR protein expression in Calu-3 cells. Fig. S4. miR-138 regulates endogenous CFTR protein expression in primary human airway epithelia. Fig. S5. miR-138 regulates CFTR expression in HeLa cells. Fig. S6. miR-138 regulates CFTR expression in HEK293T cells. Fig. S7. HeLa cells exhibit CFTR channel activity. Fig. S8. miR-138 improves CFTR processing. Fig. S9. miR-138 improves CFTR-ΔF508 processing. Fig. S10. SIN3A inhibition yields partial rescue of Cl- transport in CF epithelia. -

Role and Regulation of the P53-Homolog P73 in the Transformation of Normal Human Fibroblasts
Role and regulation of the p53-homolog p73 in the transformation of normal human fibroblasts Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen Julius-Maximilians-Universität Würzburg vorgelegt von Lars Hofmann aus Aschaffenburg Würzburg 2007 Eingereicht am Mitglieder der Promotionskommission: Vorsitzender: Prof. Dr. Dr. Martin J. Müller Gutachter: Prof. Dr. Michael P. Schön Gutachter : Prof. Dr. Georg Krohne Tag des Promotionskolloquiums: Doktorurkunde ausgehändigt am Erklärung Hiermit erkläre ich, dass ich die vorliegende Arbeit selbständig angefertigt und keine anderen als die angegebenen Hilfsmittel und Quellen verwendet habe. Diese Arbeit wurde weder in gleicher noch in ähnlicher Form in einem anderen Prüfungsverfahren vorgelegt. Ich habe früher, außer den mit dem Zulassungsgesuch urkundlichen Graden, keine weiteren akademischen Grade erworben und zu erwerben gesucht. Würzburg, Lars Hofmann Content SUMMARY ................................................................................................................ IV ZUSAMMENFASSUNG ............................................................................................. V 1. INTRODUCTION ................................................................................................. 1 1.1. Molecular basics of cancer .......................................................................................... 1 1.2. Early research on tumorigenesis ................................................................................. 3 1.3. Developing -

Human RALBP1 Peptide (DAG-P1058) This Product Is for Research Use Only and Is Not Intended for Diagnostic Use
Human RALBP1 peptide (DAG-P1058) This product is for research use only and is not intended for diagnostic use. PRODUCT INFORMATION Antigen Description RALBP1 plays a role in receptor-mediated endocytosis and is a downstream effector of the small GTP-binding protein RAL (see RALA; MIM 179550). Small G proteins, such as RAL, have GDP- bound inactive and GTP-bound active forms, which shift from the inactive to the active state through the action of RALGDS (MIM 601619), which in turn is activated by RAS (see HRAS; MIM 190020) (summary by Feig, 2003 [PubMed 12888294]).[supplied by OMIM, Nov 2010] Specificity Expressed ubiquitously but at low levels. Shows a strong expression in the erythrocytes. Nature Synthetic Expression System N/A Conjugate Unconjugated Sequence Similarities Contains 1 Rho-GAP domain. Cellular Localization Membrane. Procedure None Format Liquid Preservative None Storage Shipped at 4°C. Upon delivery aliquot and store at -20°C or -80°C. Avoid repeated freeze / thaw cycles. Information available upon request. ANTIGEN GENE INFORMATION Gene Name RALBP1 ralA binding protein 1 [ Homo sapiens (human) ] Official Symbol RALBP1 Synonyms RALBP1; ralA binding protein 1; RIP1; RLIP1; RLIP76; ralA-binding protein 1; DNP-SG ATPase; ral-interacting protein 1; 76 kDa Ral-interacting protein; dinitrophenyl S-glutathione ATPase; Entrez Gene ID 10928 mRNA Refseq NM_006788.3 45-1 Ramsey Road, Shirley, NY 11967, USA Email: [email protected] Tel: 1-631-624-4882 Fax: 1-631-938-8221 1 © Creative Diagnostics All Rights Reserved Protein -

Bioinformatics: a Practical Guide to the Analysis of Genes and Proteins, Second Edition Andreas D
BIOINFORMATICS A Practical Guide to the Analysis of Genes and Proteins SECOND EDITION Andreas D. Baxevanis Genome Technology Branch National Human Genome Research Institute National Institutes of Health Bethesda, Maryland USA B. F. Francis Ouellette Centre for Molecular Medicine and Therapeutics Children’s and Women’s Health Centre of British Columbia University of British Columbia Vancouver, British Columbia Canada A JOHN WILEY & SONS, INC., PUBLICATION New York • Chichester • Weinheim • Brisbane • Singapore • Toronto BIOINFORMATICS SECOND EDITION METHODS OF BIOCHEMICAL ANALYSIS Volume 43 BIOINFORMATICS A Practical Guide to the Analysis of Genes and Proteins SECOND EDITION Andreas D. Baxevanis Genome Technology Branch National Human Genome Research Institute National Institutes of Health Bethesda, Maryland USA B. F. Francis Ouellette Centre for Molecular Medicine and Therapeutics Children’s and Women’s Health Centre of British Columbia University of British Columbia Vancouver, British Columbia Canada A JOHN WILEY & SONS, INC., PUBLICATION New York • Chichester • Weinheim • Brisbane • Singapore • Toronto Designations used by companies to distinguish their products are often claimed as trademarks. In all instances where John Wiley & Sons, Inc., is aware of a claim, the product names appear in initial capital or ALL CAPITAL LETTERS. Readers, however, should contact the appropriate companies for more complete information regarding trademarks and registration. Copyright ᭧ 2001 by John Wiley & Sons, Inc. All rights reserved. No part of this publication may be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic or mechanical, including uploading, downloading, printing, decompiling, recording or otherwise, except as permitted under Sections 107 or 108 of the 1976 United States Copyright Act, without the prior written permission of the Publisher. -

RALBP1 Purified Maxpab Rabbit Polyclonal Antibody (D01P)
RALBP1 purified MaxPab rabbit polyclonal antibody (D01P) Catalog # : H00010928-D01P 規格 : [ 100 ug ] List All Specification Application Image Product Rabbit polyclonal antibody raised against a full-length human RALBP1 Western Blot (Tissue lysate) Description: protein. Immunogen: RALBP1 (NP_006779.1, 1 a.a. ~ 655 a.a) full-length human protein. Sequence: MTECFLPPTSSPSEHRRVEHGSGLTRTPSSEEISPTKFPGLYRTGEPSP PHDILHEPPDVVSDDEKDHGKKKGKFKKKEKRTEGYAAFQEDSSGDEAE enlarge SPSKMKRSKGIHVFKKPSFSKKKEKDFKIKEKPKEEKHKEEKHKEEKHKEK KSKDLTAADVVKQWKEKKKKKKPIQEPEVPQIDVPNLKPIFGIPLADAVER Western Blot (Cell lysate) TMMYDGIRLPAVFRECIDYVEKYGMKCEGIYRVSGIKSKVDELKAAYDRE ESTNLEDYEPNTVASLLKQYLRDLPENLLTKELMPRFEEACGRTTETEKV QEFQRLLKELPECNYLLISWLIVHMDHVIAKELETKMNIQNISIVLSPTVQIS NRVLYVFFTHVQELFGNVVLKQVMKPLRWSNMATMPTLPETQAGIKEEI RRQEFLLNCLHRDLQGGIKDLSKEERLWEVQRILTALKRKLREAKRQEC ETKIAQEIASLSKEDVSKEEMNENEEVINILLAQENEILTEQEELLAMEQFLR RQIASEKEEIERLRAEIAEIQSRQQHGRSETEEYSSESESESEDEEELQIIL EDLQRQNEELEIKNNHLNQAIHEEREAIIELRVQLRLLQMQRAKAEQQAQE enlarge DEEPEWRGGAVQPPRDGVLEPKAAKEQPKAGKEPAKPSPSRDRKETSI Western Blot (Transfected lysate) Host: Rabbit Reactivity: Human, Rat Quality Control Antibody reactive against mammalian transfected lysate. Testing: enlarge Storage Buffer: In 1x PBS, pH 7.4 In situ Proximity Ligation Assay Storage Store at -20°C or lower. Aliquot to avoid repeated freezing and thawing. (Cell) Instruction: MSDS: Download Datasheet: Download enlarge Applications Western Blot (Tissue lysate) RALBP1 MaxPab rabbit polyclonal antibody. Western Blot analysis of RALBP1 expression -

REPS2 Antibody (N-Term) Affinity Purified Rabbit Polyclonal Antibody (Pab) Catalog # Ap13131a
10320 Camino Santa Fe, Suite G San Diego, CA 92121 Tel: 858.875.1900 Fax: 858.622.0609 REPS2 Antibody (N-term) Affinity Purified Rabbit Polyclonal Antibody (Pab) Catalog # AP13131a Specification REPS2 Antibody (N-term) - Product Information Application WB, IHC-P,E Primary Accession Q8NFH8 Other Accession NP_004717.2, NP_001074444.1 Reactivity Human, Mouse Host Rabbit Clonality Polyclonal Isotype Rabbit Ig Calculated MW 71534 Antigen Region 153-182 REPS2 Antibody (N-term) - Additional Information REPS2 Antibody (N-term) (Cat. #AP13131a) Gene ID 9185 western blot analysis in mouse brain tissue lysates (35ug/lane).This demonstrates the Other Names REPS2 antibody detected the REPS2 protein RalBP1-associated Eps domain-containing (arrow). protein 2, Partner of RalBP1, RalBP1-interacting protein 2, REPS2, POB1 Target/Specificity This REPS2 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 153-182 amino acids from the N-terminal region of human REPS2. Dilution WB~~1:1000 IHC-P~~1:10~50 Format Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. REPS2 Antibody (N-term) (Cat. #AP13131a)immunohistochemistry analysis Storage in formalin fixed and paraffin embedded Maintain refrigerated at 2-8°C for up to 2 human cerebellum tissue followed by weeks. For long term storage store at -20°C peroxidase conjugation of the secondary in small aliquots to prevent freeze-thaw antibody and DAB staining.This data cycles. demonstrates the use of REPS2 Antibody (N-term) for immunohistochemistry. Clinical Precautions relevance has not been evaluated. -
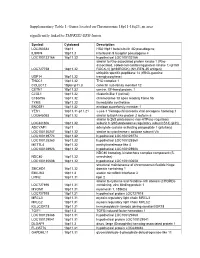
Supplementary Table 1: Genes Located on Chromosome 18P11-18Q23, an Area Significantly Linked to TMPRSS2-ERG Fusion
Supplementary Table 1: Genes located on Chromosome 18p11-18q23, an area significantly linked to TMPRSS2-ERG fusion Symbol Cytoband Description LOC260334 18p11 HSA18p11 beta-tubulin 4Q pseudogene IL9RP4 18p11.3 interleukin 9 receptor pseudogene 4 LOC100132166 18p11.32 hypothetical LOC100132166 similar to Rho-associated protein kinase 1 (Rho- associated, coiled-coil-containing protein kinase 1) (p160 LOC727758 18p11.32 ROCK-1) (p160ROCK) (NY-REN-35 antigen) ubiquitin specific peptidase 14 (tRNA-guanine USP14 18p11.32 transglycosylase) THOC1 18p11.32 THO complex 1 COLEC12 18pter-p11.3 collectin sub-family member 12 CETN1 18p11.32 centrin, EF-hand protein, 1 CLUL1 18p11.32 clusterin-like 1 (retinal) C18orf56 18p11.32 chromosome 18 open reading frame 56 TYMS 18p11.32 thymidylate synthetase ENOSF1 18p11.32 enolase superfamily member 1 YES1 18p11.31-p11.21 v-yes-1 Yamaguchi sarcoma viral oncogene homolog 1 LOC645053 18p11.32 similar to BolA-like protein 2 isoform a similar to 26S proteasome non-ATPase regulatory LOC441806 18p11.32 subunit 8 (26S proteasome regulatory subunit S14) (p31) ADCYAP1 18p11 adenylate cyclase activating polypeptide 1 (pituitary) LOC100130247 18p11.32 similar to cytochrome c oxidase subunit VIc LOC100129774 18p11.32 hypothetical LOC100129774 LOC100128360 18p11.32 hypothetical LOC100128360 METTL4 18p11.32 methyltransferase like 4 LOC100128926 18p11.32 hypothetical LOC100128926 NDC80 homolog, kinetochore complex component (S. NDC80 18p11.32 cerevisiae) LOC100130608 18p11.32 hypothetical LOC100130608 structural maintenance -

Quantitative Trait Loci Mapping of Macrophage Atherogenic Phenotypes
QUANTITATIVE TRAIT LOCI MAPPING OF MACROPHAGE ATHEROGENIC PHENOTYPES BRIAN RITCHEY Bachelor of Science Biochemistry John Carroll University May 2009 submitted in partial fulfillment of requirements for the degree DOCTOR OF PHILOSOPHY IN CLINICAL AND BIOANALYTICAL CHEMISTRY at the CLEVELAND STATE UNIVERSITY December 2017 We hereby approve this thesis/dissertation for Brian Ritchey Candidate for the Doctor of Philosophy in Clinical-Bioanalytical Chemistry degree for the Department of Chemistry and the CLEVELAND STATE UNIVERSITY College of Graduate Studies by ______________________________ Date: _________ Dissertation Chairperson, Johnathan D. Smith, PhD Department of Cellular and Molecular Medicine, Cleveland Clinic ______________________________ Date: _________ Dissertation Committee member, David J. Anderson, PhD Department of Chemistry, Cleveland State University ______________________________ Date: _________ Dissertation Committee member, Baochuan Guo, PhD Department of Chemistry, Cleveland State University ______________________________ Date: _________ Dissertation Committee member, Stanley L. Hazen, MD PhD Department of Cellular and Molecular Medicine, Cleveland Clinic ______________________________ Date: _________ Dissertation Committee member, Renliang Zhang, MD PhD Department of Cellular and Molecular Medicine, Cleveland Clinic ______________________________ Date: _________ Dissertation Committee member, Aimin Zhou, PhD Department of Chemistry, Cleveland State University Date of Defense: October 23, 2017 DEDICATION I dedicate this work to my entire family. In particular, my brother Greg Ritchey, and most especially my father Dr. Michael Ritchey, without whose support none of this work would be possible. I am forever grateful to you for your devotion to me and our family. You are an eternal inspiration that will fuel me for the remainder of my life. I am extraordinarily lucky to have grown up in the family I did, which I will never forget. -

Rlip Depletion Prevents Spontaneous Neoplasia in TP53 Null Mice
Rlip depletion prevents spontaneous neoplasia in TP53 null mice Sanjay Awasthia,b,1, Joshua Tompkinsb, Jyotsana Singhalb, Arthur D. Riggsb,1, Sushma Yadavb, Xiwei Wuc, Sharda Singha, Charles Wardenc, Zheng Liud, Jinhui Wangc, Thomas P. Slavine, Jeffrey N. Weitzele, Yate-Ching Yuand, Meenakshi Awasthib, Satish K. Srivastavaf, Yogesh C. Awasthif, and Sharad S. Singhalb aDivision of Hematology & Oncology, Department of Internal Medicine, Texas Tech Health Sciences Center, Lubbock, TX 79430-9410; bDiabetes and Metabolism Research Institute, City of Hope Comprehensive Cancer Center, Duarte, CA 91010; cIntegrative Genomics Core, City of Hope Comprehensive Cancer Center, Duarte, CA 91010; dBioinformatics Core, City of Hope Comprehensive Cancer Center, Duarte, CA 91010; eDivision of Clinical Cancer Genomics, City of Hope Comprehensive Cancer Center, Duarte, CA 91010; and fDepartment of Biochemistry and Molecular Biology, The University of Texas Medical Branch, Galveston, TX 77555-0647 Contributed by Arthur D. Riggs, February 7, 2018 (sent for review November 9, 2017; reviewed by Shivendra Singh and Dan Theodorescu) TP53 (p53) is a tumor suppressor whose functions are lost or electrophilic toxins (9–30). Because Rlip-catalyzed efflux of GS-E altered in most malignancies. p53 homozygous knockout (p53−/−) prevents product/feedback inhibition of several mercapturic acid mice uniformly die of spontaneous malignancy, typically T-cell pathway enzymes, its loss promotes apoptosis exerted by xenobiotics lymphoma. RALBP1 (RLIP76, Rlip) is a stress-protective, mercapturic and GS-E, derived from oxidative degradation of ω-6 fatty acids acid pathway transporter protein that also functions as a Ral effec- (31). Its ATPase activity is coupled with clathrin-dependent endo- tor involved in clathrin-dependent endocytosis. -

RLIP76: a Structural and Functional Triumvirate
cancers Review RLIP76: A Structural and Functional Triumvirate Jasmine Cornish, Darerca Owen and Helen R. Mott * Department of Biochemistry, University of Cambridge, 80 Tennis Court Road, Cambridge CB2 1GA, UK; [email protected] (J.C.); [email protected] (D.O.) * Correspondence: [email protected] Simple Summary: The RLIP76 protein is present at high levels in multiple cancers, compared with levels in normal cells. In cancer cells it is thought to be important for removal of chemotherapeutics, while in normal cells it has been implicated in endocytosis, stress response and mitochondrial fission. Although RLIP76 is a potential target for ovary, breast, lung, colon, prostate and kidney cancers, only the middle third of the protein has been structurally characterized. Understanding the full structure of RLIP76 will help us to better understand the signalling pathways in which it is involved and by extension its role in cancer. Abstract: RLIP76/RalBP1 is an ATP-dependent transporter of glutathione conjugates, which is overexpressed in various human cancers, but its diverse functions in normal cells, which include endocytosis, stress response and mitochondrial dynamics, are still not fully understood. The protein can be divided into three distinct regions, each with its own structural properties. At the centre of the protein are two well-defined domains, a GTPase activating protein domain targeting Rho family small G proteins and a small coiled-coil that binds to the Ras family small GTPases RalA and RalB. In engaging with Rho and Ral proteins, RLIP76 bridges these two distinct G protein families. The N-terminal region is predicted to be disordered and is rich in basic amino acids, which may Citation: Cornish, J.; Owen, D.; Mott, mediate membrane association, consistent with its role in transport. -

Expression of Ral Gtpases,Their Effectors, and Activators in Human
Human Cancer Biology Expression of Ral GTPases, Their Effectors, and Activators in Human Bladder Cancer Steven Christopher Smith,1, 2 Gary Oxford,1, 2 Alexander S. Baras,3 Charles Owens,1, 2 Dmytro Havaleshko,1, 2 David L. Brautigan,4 Martin K. Safo,6 and DanTheodorescu1, 2 , 5 Abstract Purpose: The Ral family of small G proteins has been implicated in tumorigenesis, invasion, and metastasis in in vitro and animal model systems; however, a systematic evaluation of the state of activation, mutation, or expression of these GTPases has not been reported in any tumor type. Experimental Design: We determined the activation state of the RalA and RalB paralogs in 10 bladder cancer cell lines with varying Ras mutation status. We sequenced RalA and RalB cDNAs from 20 bladder cancer cell lines and functionally evaluated the mutations found. We determined the expression of Ral, Ral activators, and Ral effectors on the level of mRNA or protein in human bladder cancer cell lines and tissues. Results: We uncovered one E97Q substitution mutation of RalA in 1 of 20 cell lines tested and higher Ral activation in cells harboring mutant HRAS. We found overexpression of mRNAs for RalA and Aurora-A, a mitotic kinase that activates RalA, in bladder cancer (both P < 0.001), and in association with tumors of higher stage and grade. RalBP1,a canonical Ral effector, mRNA and protein was overexpressed in bladder cancer (P < 0.001), whereas Filamin A was underex- pressed (P = 0.004).We determined that RalA mRNA levels correlated significantly with protein levels (P < 0.001) and found protein overexpression of both GTPases in homogenized invasive cancers.