Ecophysiology and Food Web Dynamics of Spring Ecotone
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Schriever, Bogan, Boersma, Cañedo-Argüelles, Jaeger, Olden, and Lytle
Schriever, Bogan, Boersma, Cañedo-Argüelles, Jaeger, Olden, and Lytle. Hydrology shapes taxonomic and functional structure of desert stream invertebrate communities. Freshwater Science Vol. 34, No. 2 Appendix S1. References for trait state determination. Order Family Taxon Body Voltinism Dispersal Respiration FFG Diapause Locomotion Source size Amphipoda Crustacea Hyalella 3 3 1 2 2 2 3 1, 2 Annelida Hirudinea Hirudinea 2 2 3 3 6 2 5 3 Anostraca Anostraca Anostraca 2 3 3 2 4 1 5 1, 3 Basommatophora Ancylidae Ferrissia 1 2 1 1 3 3 4 1 Ancylidae Ancylidae 1 2 1 1 3 3 4 3, 4 Class:Arachnida subclass:Acari Acari 1 2 3 1 5 1 3 5,6 Coleoptera Dryopidae Helichus lithophilus 1 2 4 3 3 3 4 1,7, 8 Helichus suturalis 1 2 4 3 3 3 4 1 ,7, 9, 8 Helichus triangularis 1 2 4 3 3 3 4 1 ,7, 9,8 Postelichus confluentus 1 2 4 3 3 3 4 7,9,10, 8 Postelichus immsi 1 2 4 3 3 3 4 7,9, 10,8 Dytiscidae Agabus 1 2 4 3 6 1 5 1,11 Desmopachria portmanni 1 3 4 3 6 3 5 1,7,10,11,12 Hydroporinae 1 3 4 3 6 3 5 1 ,7,9, 11 Hygrotus patruelis 1 3 4 3 6 3 5 1,11 Hygrotus wardi 1 3 4 3 6 3 5 1,11 Laccophilus fasciatus 1 2 4 3 6 3 5 1, 11,13 Laccophilus maculosus 1 3 4 3 6 3 5 1, 11,13 Laccophilus mexicanus 1 2 4 3 6 3 5 1, 11,13 Laccophilus oscillator 1 2 4 3 6 3 5 1, 11,13 Laccophilus pictus 1 2 4 3 6 3 5 1, 11,13 Liodessus obscurellus 1 3 4 3 6 3 5 1 ,7,11 Neoclypeodytes cinctellus 1 3 4 3 7 3 5 14,15,1,10,11 Neoclypeodytes fryi 1 3 4 3 7 3 5 14,15,1,10,11 Neoporus 1 3 4 3 7 3 5 14,15,1,10,11 Rhantus atricolor 2 2 4 3 6 3 5 1,16 Schriever, Bogan, Boersma, Cañedo-Argüelles, Jaeger, Olden, and Lytle. -

Research Report110
~ ~ WISCONSIN DEPARTMENT OF NATURAL RESOURCES A Survey of Rare and Endangered Mayflies of Selected RESEARCH Rivers of Wisconsin by Richard A. Lillie REPORT110 Bureau of Research, Monona December 1995 ~ Abstract The mayfly fauna of 25 rivers and streams in Wisconsin were surveyed during 1991-93 to document the temporal and spatial occurrence patterns of two state endangered mayflies, Acantha metropus pecatonica and Anepeorus simplex. Both species are candidates under review for addition to the federal List of Endang ered and Threatened Wildlife. Based on previous records of occur rence in Wisconsin, sampling was conducted during the period May-July using a combination of sampling methods, including dredges, air-lift pumps, kick-nets, and hand-picking of substrates. No specimens of Anepeorus simplex were collected. Three specimens (nymphs or larvae) of Acanthametropus pecatonica were found in the Black River, one nymph was collected from the lower Wisconsin River, and a partial exuviae was collected from the Chippewa River. Homoeoneuria ammophila was recorded from Wisconsin waters for the first time from the Black River and Sugar River. New site distribution records for the following Wiscon sin special concern species include: Macdunnoa persimplex, Metretopus borealis, Paracloeodes minutus, Parameletus chelifer, Pentagenia vittigera, Cercobrachys sp., and Pseudiron centra/is. Collection of many of the aforementioned species from large rivers appears to be dependent upon sampling sand-bottomed substrates at frequent intervals, as several species were relatively abundant during only very short time spans. Most species were associated with sand substrates in water < 2 m deep. Acantha metropus pecatonica and Anepeorus simplex should continue to be listed as endangered for state purposes and receive a biological rarity ranking of critically imperiled (S1 ranking), and both species should be considered as candidates proposed for listing as endangered or threatened as defined by the Endangered Species Act. -

Pervasive Gene Flow Across Critical Habitat for Four Narrowly Endemic
Freshwater Biology (2016) doi:10.1111/fwb.12758 Pervasive gene flow across critical habitat for four narrowly endemic, sympatric taxa † † ‡ LAUREN K. LUCAS* , ZACHARIAH GOMPERT ,J.RANDYGIBSON , KATHERINE L. BELL*, § C. ALEX BUERKLE AND CHRIS C. NICE* *Department of Biology, Texas State University, San Marcos, TX, U.S.A. † Department of Biology, Utah State University, Logan, UT, U.S.A. ‡ U.S. Fish & Wildlife Service, San Marcos Aquatic Resources Center, San Marcos, TX, U.S.A. § Department of Botany, University of Wyoming, Laramie, WY, U.S.A. SUMMARY 1. We studied genetic variation in four endangered animal taxa in the largest freshwater spring complex in the southwestern U.S.A., Comal Springs (TX): Eurycea salamanders, Heterelmis riffle beetles, Stygobromus amphipods and Stygoparnus dryopid beetles. They inhabit a spring complex with nearly stable conditions, which is threatened by climate change and aquifer withdrawals. The four taxa vary in their habitat affinities and body sizes. 2. We used genotyping-by-sequencing to obtain hundreds to thousands of genetic markers to accurately infer the demographic history of the taxa. We used approximate Bayesian computation to test models of gene flow and compare the results among taxa. We also looked for evidence that would suggest local adaptation within the spring complex. 3. An island model (equal gene flow among all subpopulations) was the most probable of the five models tested, and all four taxa had high migration rate estimates. 4. Small numbers of single nucleotide polymorphisms (SNPs) in each taxon tested were associated with environmental conditions and provide some evidence for potential local adaptation to slightly variable conditions across habitat patches within Comal Springs. -

Edwards Aquifer Authority Study No. 14-14-697-Hcp
ATTACHMENT 3 DETERMINATION OF LIMITATIONS OF COMAL SPRINGS RIFFLE BEETLE PLASTRON USE DURING LOW-FLOW STUDY EDWARDS AQUIFER AUTHORITY STUDY NO. 14-14-697-HCP FINAL REPORT Prepared by Weston H. Nowlin 601 University Drive Department of Biology Aquatic Station Texas State University San Marcos, TX 78666 (512) 245-8794 [email protected] Benjamin Schwartz 601 University Drive Edwards Aquifer Research and Data Center and Department of Biology Aquatic Station Texas State University San Marcos, TX 78666 (512) 245-7608 [email protected] Thom Hardy 601 University Drive The Meadows Center for Water and the Environment Texas State University San Marcos, TX 78666 (512) 245-6729 [email protected] and Randy Gibson United States Fish and Wildlife Service San Marcos Aquatic Resources Center 500 East McCarty Lane San Marcos, TX 78666 (512) 353-0011, ext. 226 [email protected] Riffle Beetle Plastron Study - 1 ATTACHMENT 3 TABLE OF CONTENTS TITLE PAGE………………………………………………………………………………….……1 TABLE OF CONTENTS…...................................................................................................................................2 LIST OF FIGURES AND TABLES……………………………………………………………….……3 EXECUTIVE SUMMARY………………………………………………………………………….…4 BACKGROUND AND SIGNIFICANCE…………………………………………………………….….5 INTRODUCTION AND LITERATURE REVIEW…………………………………………………….…6 CONCEPTUAL FOUNDATION, EXPERIMENTAL DESIGN, AND METHODS…………………….…….7 Collection and Housing of Beetles……………………………………………….………….…..7 Conceptual Foundation for Experiments………………………………. ………………………8 Short-Term -

New State Records of Aquatic Insects for Ohio, U.S.A
Volume 121, Number 1, January and February 2010 75 NEW STATE RECORDS OF AQUATIC INSECTS FOR OHIO, U.S.A. (EPHEMEROPTERA, PLECOPTERA, TRICHOPTERA, COLEOPTERA)1 Michael J. Bolton2 ABSTRACT: Biomonitoring of Ohio streams by the Ohio Environmental Protection Agency has found new state records for the Ephemeroptera (mayflies): Baetis brunneicolor McDunnough, Iswaeon anoka (Daggy), Paracloeodes fleeki McCafferty and Lenat, Plauditus cestus (Provonsha and McCafferty), and Rhithrogena manifesta Eaton; the Plecoptera (stoneflies): Pteronarcys cf. biloba Newman; the Trichop- tera (caddisflies): Brachycentrus numerosus (Say) and Psilotreta rufa (Hagen); and the Coleoptera (bee- tles): Gyretes sinuatus LeConte, Dicranopselaphus variegatus Horn, and Microcylloepus pusillus (Le Conte). Additional records are given for the mayfly Paracloeodes minutus (Daggy). KEY WORDS: Ohio, state record, Ephemeroptera, Plecoptera, Trichoptera, Coleoptera The Ohio Environmental Protection Agency conducts biological and water qual- ity studies of Ohio streams to ascertain the condition of the aquatic resource. One component of these studies is an evaluation of the macroinvertebrate communities. As a result of this sampling, species of aquatic insects in the Ephemeroptera (may- flies), Plecoptera (stoneflies), Trichoptera (caddisflies), and Coleoptera (beetles) orders have been collected that have never been reported from Ohio. Randolph and McCafferty (1998) compiled the first state list of mayflies for Ohio. Gaufin (1956) produced a state list of stoneflies for Ohio with additions by Tkac and Foote (1978), Robertson (1979), and Fishbeck (1987). Listing of species distributions by state in Stewart and Stark (2002) and Stark and Armitage (2000, 2004) incorporated Ohio records found in the various revisionary publications. Huryn and Foote (1983) pro- duced the first comprehensive state list of caddisflies which was amended by Mac Lean and MacLean (1984), Usis and MacLean (1986), Garono and MacLean (1988), Usis and Foote (1989), and Keiper and Bartolotta (2003). -
![Docket No. FWS-R2-ES-2012-0082]](https://docslib.b-cdn.net/cover/5485/docket-no-fws-r2-es-2012-0082-385485.webp)
Docket No. FWS-R2-ES-2012-0082]
This document is scheduled to be published in the Federal Register on 10/19/2012 and available online at http://federalregister.gov/a/2012-25578, and on FDsys.gov 1 DEPARTMENT OF THE INTERIOR Fish and Wildlife Service 50 CFR Part 17 [Docket No. FWS-R2-ES-2012-0082] [4500030114] RIN 1018-AY20 Endangered and Threatened Wildlife and Plants; Proposed Revision of Critical Habitat for the Comal Springs Dryopid Beetle, Comal Springs Riffle Beetle, and Peck’s Cave Amphipod AGENCY: Fish and Wildlife Service, Interior. ACTION: Proposed rule. SUMMARY: We, the U.S. Fish and Wildlife Service (Service), propose to revise 2 designation of critical habitat for the Comal Springs dryopid beetle (Stygoparnus comalensis), Comal Springs riffle beetle (Heterelmis comalensis), and Peck’s cave amphipod (Stygobromus pecki), under the Endangered Species Act of 1973, as amended (Act). In total, approximately 169 acres (68 hectares) are being proposed for revised critical habitat. The proposed revision of critical habitat is located in Comal and Hays Counties, Texas. DATES: We will accept comments received or postmarked on or before [INSERT DATE 60 DAYS AFTER DATE OF PUBLICATION IN THE FEDERAL REGISTER]. Comments submitted electronically using the Federal eRulemaking Portal (see ADDRESSES section, below) must be received by 11:59 p.m. Eastern Time on the closing date. We must receive requests for public hearings, in writing, at the address shown in FOR FURTHER INFORMATION CONTACT by [INSERT DATE 45 DAYS AFTER DATE OF PUBLICATION IN THE FEDERAL REGISTER]. ADDRESSES: You may submit comments by one of the following methods: (1) Electronically: Go to the Federal eRulemaking Portal: http://www.regulations.gov. -

The Safety of SLICE (0.2% Emamectin Benzoate) Administered in Feed to Fingerling Rainbow Trout James D
This article was downloaded by: [Department Of Fisheries] On: 19 August 2013, At: 20:46 Publisher: Taylor & Francis Informa Ltd Registered in England and Wales Registered Number: 1072954 Registered office: Mortimer House, 37-41 Mortimer Street, London W1T 3JH, UK North American Journal of Aquaculture Publication details, including instructions for authors and subscription information: http://www.tandfonline.com/loi/unaj20 The Safety of SLICE (0.2% Emamectin Benzoate) Administered in Feed to Fingerling Rainbow Trout James D. Bowker a , Dan Carty a & Molly P. Bowman a a U.S. Fish and Wildlife Service, Aquatic Animal Drug Approval Partnership Program , 4050 Bridger Canyon Road, Bozeman , Montana , 59715 , USA Published online: 31 Jul 2013. To cite this article: James D. Bowker , Dan Carty & Molly P. Bowman (2013) The Safety of SLICE (0.2% Emamectin Benzoate) Administered in Feed to Fingerling Rainbow Trout, North American Journal of Aquaculture, 75:4, 455-462, DOI: 10.1080/15222055.2013.806383 To link to this article: http://dx.doi.org/10.1080/15222055.2013.806383 PLEASE SCROLL DOWN FOR ARTICLE Taylor & Francis makes every effort to ensure the accuracy of all the information (the “Content”) contained in the publications on our platform. However, Taylor & Francis, our agents, and our licensors make no representations or warranties whatsoever as to the accuracy, completeness, or suitability for any purpose of the Content. Any opinions and views expressed in this publication are the opinions and views of the authors, and are not the views of or endorsed by Taylor & Francis. The accuracy of the Content should not be relied upon and should be independently verified with primary sources of information. -

Biological Monitoring of Surface Waters in New York State, 2019
NYSDEC SOP #208-19 Title: Stream Biomonitoring Rev: 1.2 Date: 03/29/19 Page 1 of 188 New York State Department of Environmental Conservation Division of Water Standard Operating Procedure: Biological Monitoring of Surface Waters in New York State March 2019 Note: Division of Water (DOW) SOP revisions from year 2016 forward will only capture the current year parties involved with drafting/revising/approving the SOP on the cover page. The dated signatures of those parties will be captured here as well. The historical log of all SOP updates and revisions (past & present) will immediately follow the cover page. NYSDEC SOP 208-19 Stream Biomonitoring Rev. 1.2 Date: 03/29/2019 Page 3 of 188 SOP #208 Update Log 1 Prepared/ Revision Revised by Approved by Number Date Summary of Changes DOW Staff Rose Ann Garry 7/25/2007 Alexander J. Smith Rose Ann Garry 11/25/2009 Alexander J. Smith Jason Fagel 1.0 3/29/2012 Alexander J. Smith Jason Fagel 2.0 4/18/2014 • Definition of a reference site clarified (Sect. 8.2.3) • WAVE results added as a factor Alexander J. Smith Jason Fagel 3.0 4/1/2016 in site selection (Sect. 8.2.2 & 8.2.6) • HMA details added (Sect. 8.10) • Nonsubstantive changes 2 • Disinfection procedures (Sect. 8) • Headwater (Sect. 9.4.1 & 10.2.7) assessment methods added • Benthic multiplate method added (Sect, 9.4.3) Brian Duffy Rose Ann Garry 1.0 5/01/2018 • Lake (Sect. 9.4.5 & Sect. 10.) assessment methods added • Detail on biological impairment sampling (Sect. -

Underwater Breathing: the Mechanics of Plastron Respiration
J. Fluid Mech. (2008), vol. 608, pp. 275–296. c 2008 Cambridge University Press 275 doi:10.1017/S0022112008002048 Printed in the United Kingdom Underwater breathing: the mechanics of plastron respiration M. R. FLYNN† AND J O H N W. M. B U S H Department of Mathematics, Massachusetts Institute of Technology, 77 Massachusetts Avenue, Cambridge, MA 02139-4307, USA (Received 11 July 2007 and in revised form 10 April 2008) The rough, hairy surfaces of many insects and spiders serve to render them water-repellent; consequently, when submerged, many are able to survive by virtue of a thin air layer trapped along their exteriors. The diffusion of dissolved oxygen from the ambient water may allow this layer to function as a respiratory bubble or ‘plastron’, and so enable certain species to remain underwater indefinitely. Main- tenance of the plastron requires that the curvature pressure balance the pressure difference between the plastron and ambient. Moreover, viable plastrons must be of sufficient area to accommodate the interfacial exchange of O2 and CO2 necessary to meet metabolic demands. By coupling the bubble mechanics, surface and gas-phase chemistry, we enumerate criteria for plastron viability and thereby deduce the range of environmental conditions and dive depths over which plastron breathers can survive. The influence of an external flow on plastron breathing is also examined. Dynamic pressure may become significant for respiration in fast-flowing, shallow and well-aerated streams. Moreover, flow effects are generally significant because they sharpen chemical gradients and so enhance mass transfer across the plastron interface. Modelling this process provides a rationale for the ventilation movements documented in the biology literature, whereby arthropods enhance plastron respiration by flapping their limbs or antennae. -
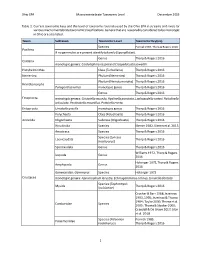
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al. -

Habitat and Phenology of the Endangered Riffle Eetle, Heterelmis
Arch. Hydrobiol. 156 3 361-383 Stuttgart, February 2003 Habitat and phenology of the endangered riffle beetle Heterelmis comalensis and a coexisting species, Microcylloepus pusillus, (Coleoptera: Elmidae) at Comal Springs, Texas, USA David E. Bowles1 *, Cheryl B. Barr2 and Ruth Stanford3 Texas Parks and Wildlife Department, University of California, Berkeley, and United States Fish and Wildlife Service With 5 figures and 4 tables Ab tract: Habitat characteristics and seasonal distribution of the riffle beetles Herere/ mis comalensis and Microcylloepu pusillus were studied at Comal Springs, Texas, during 1993-1994, to aid in developing sound reconunendations for sustaining their natural popu1atioas. Comal Springs consists of four major spring cutlers and spring runs. The four spring-runs are dissimilar in size, appearance, canopy and riparian cover, substrate composition, and aquatic macrophyte composition. Habitat conditions associated with the respective popuJatioos of riffle beetles, including physical-chemi cal measurements, water depth, and currenc velocity, were relatively unifom1 and var ied lHUe among sampling dates and spring-runs. However, the locations of the beetles in the respective spri ng-runs were not well correlated to current velocity, water depth, or distance from primary spring orifices. Factors such as substrate size and availability and competition are proposed as possibly influencing lheir respective distributions. Maintaining high-quality spring-flows and protection of Lhe physical habitat of Here· re/mis comalensis presently are the only means by which to ensure the survival of this endemic species. Key words: Conservation, habitat conditions, substrate availability, competition. 1 Authors' addresses: Texas Parks and Wildlife Department, 4200 Smith School Road, Austin, Texas 78744, USA. -

Empirically Derived Indices of Biotic Integrity for Forested Wetlands, Coastal Salt Marshes and Wadable Freshwater Streams in Massachusetts
Empirically Derived Indices of Biotic Integrity for Forested Wetlands, Coastal Salt Marshes and Wadable Freshwater Streams in Massachusetts September 15, 2013 This report is the result of several years of field data collection, analyses and IBI development, and consideration of the opportunities for wetland program and policy development in relation to IBIs and CAPS Index of Ecological Integrity (IEI). Contributors include: University of Massachusetts Amherst Kevin McGarigal, Ethan Plunkett, Joanna Grand, Brad Compton, Theresa Portante, Kasey Rolih, and Scott Jackson Massachusetts Office of Coastal Zone Management Jan Smith, Marc Carullo, and Adrienne Pappal Massachusetts Department of Environmental Protection Lisa Rhodes, Lealdon Langley, and Michael Stroman Empirically Derived Indices of Biotic Integrity for Forested Wetlands, Coastal Salt Marshes and Wadable Freshwater Streams in Massachusetts Abstract The purpose of this study was to develop a fully empirically-based method for developing Indices of Biotic Integrity (IBIs) that does not rely on expert opinion or the arbitrary designation of reference sites and pilot its application in forested wetlands, coastal salt marshes and wadable freshwater streams in Massachusetts. The method we developed involves: 1) using a suite of regression models to estimate the abundance of each taxon across a gradient of stressor levels, 2) using statistical calibration based on the fitted regression models and maximum likelihood methods to predict the value of the stressor metric based on the abundance of the taxon at each site, 3) selecting taxa in a forward stepwise procedure that conditionally improves the concordance between the observed stressor value and the predicted value the most and a stopping rule for selecting taxa based on a conditional alpha derived from comparison to pseudotaxa data, and 4) comparing the coefficient of concordance for the final IBI to the expected distribution derived from randomly permuted data.